Form 8-K Foundation Medicine, For: Jan 08
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant
to Section 13 or 15(d)
of
The Securities Exchange Act of 1934
Date
of Report (Date of Earliest Event Reported): January 8, 2018
Foundation Medicine, Inc.
(Exact
name of registrant as specified in its charter)
|
Delaware |
001-36086 |
27-1316416 |
||
|
(State or other jurisdiction |
(Commission |
(I.R.S. Employer Identification No.) |
|
150 Second Street Cambridge, MA |
02141 |
|
|
(Address of principal executive offices) |
(Zip Code) |
Registrant’s telephone number, including area code (617) 418-2200
Not Applicable
(Former
name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
⃞
Written
communications pursuant to Rule 425 under the Securities Act (17 CFR
230.425)
⃞
Soliciting
material pursuant to Rule 14a-12 under the Exchange Act (17 CFR
240.14a-12)
⃞
Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR
240.14d-2(b))
⃞
Pre-commencement
communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR
240.13e-4(c))
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ⊠
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ⊠
Item 2.02. Results of Operations and Financial Condition.
On January 8, 2018, Foundation Medicine, Inc. (the “Company”) issued a press release announcing preliminary financial results for the quarter and year ended December 31, 2017. A copy of the press release is being furnished as Exhibit 99.1 to this Report on Form 8-K.
Item 7.01. Other Events.
Troy Cox, the Company’s Chief Executive Officer, will present at the 36th Annual J.P. Morgan Healthcare Conference on Monday, January 8, 2018 at 3:30 p.m. PST in San Francisco. A copy of the presentation slide deck is being furnished as Exhibit 99.2 to this Report on Form 8-K. Additionally, Mr. Cox will participate in a panel discussion focused on the Food & Drug Administration and Centers for Medicare and Medicaid Services Parallel Review process on Monday, January 8, 2018 at 5:15 p.m. PST. The presentation and panel discussion will each be webcast live through the Company’s website at investors.foundationmedicine.com.
Reference is made to, and there is hereby incorporated by reference into this Item 7.01, the information set forth above under “Item 2.02. Results of Operations and Financial Condition.” relating to the press release announcing preliminary financial results.
The information in this Report on Form 8-K and Exhibits 99.1 and 99.2 attached hereto is intended to be furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as expressly set forth by specific reference in such filing.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits
|
Exhibit No. |
Description |
| 99.1 | Press release issued by Foundation Medicine, Inc. dated January 8, 2018, furnished hereto. |
| 99.2 | Presentation Slide Deck, furnished hereto. |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
Date: January 8, 2018 |
FOUNDATION MEDICINE, INC. |
|
|
|
By: |
/s/ Robert W. Hesslein |
|
Robert W. Hesslein |
||
|
Senior Vice President and General Counsel |
||
EXHIBIT INDEX
|
Exhibit No. |
Description |
|
Press release issued by Foundation Medicine, Inc. dated January 8, 2018, furnished hereto. |
|
Exhibit 99.1
Foundation Medicine Reports Preliminary 2017 Results
Reports Total Revenue of Approximately $152.9 Million, a 31% Year Over Year Increase; Reports 67,375 Clinical Tests in 2017, a 54% Year Over Year Increase
Increases Biopharma Revenue Approximately 27% Year-Over-Year with New and Expanded Collaborations
Achieves Significant Milestone with FDA Approval and Preliminary National Coverage Determination (NCD) for FoundationOne CDx™
CAMBRIDGE, Mass.--(BUSINESS WIRE)--January 8, 2018--Foundation Medicine (NASDAQ: FMI) today announced preliminary unaudited total revenue of approximately $48.9 million for the fourth quarter of 2017 and approximately $152.9 million for the full year ended December 31, 2017, representing a 70% and 31% increase from the $28.8 million and $116.9 million recorded in the fourth quarter and full year ended December 31, 2016, respectively.
“2017 was a year of significant milestones for Foundation Medicine, including posting record revenue and achieving our largest annual increase in recorded clinical volume,” stated Troy Cox, chief executive officer of Foundation Medicine. “Our most notable achievement was the successful completion of the parallel review process during the fourth quarter which culminated with FDA approval and a preliminary National Coverage Determination (NCD) for FoundationOne CDx™ from CMS. This approval and preliminary NCD are transformational for our company, clinicians, biopharma partners and most importantly, for patients with advanced cancer, as we continue to meaningfully improve access to personalized cancer care.”
Revenue from biopharmaceutical companies is expected to be approximately $33.4 million in the fourth quarter of 2017 and approximately $99.7 million for the full year ended December 31, 2017, compared to $19.0 million and $78.8 million in the fourth quarter and full year ended December 31, 2016, respectively. The company reported 6,206 tests to biopharmaceutical customers in this year’s fourth quarter.
Revenue from clinical testing is expected to be approximately $15.5 million in the fourth quarter of 2017 and approximately $53.1 million for the full year ended December 31, 2017, compared to $9.8 million and $38.1 million in the fourth quarter and full year ended December 31, 2016, respectively. The company reported 20,044 clinical tests to ordering physicians in the fourth quarter of 2017, compared to a total of 12,788 tests reported during the fourth quarter of 2016, an increase of 57%. A total of 67,375 clinical tests were reported to ordering physicians for the full year ended December 31, 2017, compared to 43,686 clinical tests reported in 2016, an increase of 54%.
Based on the new revenue reporting the company initiated during 2017, Molecular Information Services revenue is expected to be approximately $37.4 million in the fourth quarter of 2017 and approximately $117.1 million for the full year ended December 31, 2017, compared to $20.4 million and $81.8 million in the fourth quarter and full year ended December 31, 2016, respectively. Pharma Research and Development Services revenue is expected to be approximately $11.5 million in the fourth quarter of 2017 and approximately $35.8 million for the full year ended December 31, 2017, compared to $8.4 million and $35.1 million in the fourth quarter and full year ended December 31, 2016, respectively.
Cash and cash equivalents at December 31, 2017 was approximately $71.4 million, including $30 million in new borrowings received during the fourth quarter under the company’s Credit Facility Agreement with Roche Finance.
2017 Enterprise Highlights:
- Received approval from the U.S. Food & Drug Administration (FDA) under the parallel review process for FoundationOne CDx, the first broad genomic profiling test incorporating multiple companion diagnostics. Simultaneously, the Centers for Medicare and Medicaid Services (CMS) issued a preliminary NCD for FoundationOne CDx, which is expected to improve access to molecular information for personalized healthcare. The NCD is expected to be finalized during the first quarter of 2018.
- Grew the biopharma business with new and expanded collaborations for molecular information solutions including companion diagnostics, molecular profiling, data insights, and biomarker discovery.
- Presented validation data for a novel assay measuring tumor mutational burden in blood (bTMB) providing evidence that response to immunotherapy can be predicted using a blood sample. Based on these findings, Foundation Medicine’s bTMB assay is being integrated as part of Roche/Genentech's prospective, randomized Phase III Blood First Assay Screening Trial (BFAST) as a companion diagnostic assay investigating the use of bTMB as a non-invasive biomarker of response to first-line atezolizumab in advanced NSCLC patients.
- Received approval from the State of New York Department of Health for FoundationACT®, the company’s liquid biopsy assay.
- Announced a collaboration with the American Society of Clinical Oncology (ASCO) to identify patients for its Targeted Agent and Profiling Utilization Registry (TAPUR) study.
- Announced a collaboration with the National Cancer Institute (NCI) and ECOG-ACRIN Cancer Research Group to identify patients for the NCI-Match (Molecular Analysis for Therapy Choice) study.
- Increased FoundationCORE™, the company’s molecular information database, to nearly 180,000 clinical cases.
- Expanded the company’s global footprint and patient access to personalized healthcare by commencing operations at the company’s laboratory in Penzberg, Germany. The Penzberg location is supporting continued growth and expansion in Europe through the company’s commercial collaboration with Roche.
- Published 95 peer-reviewed manuscripts in top medical and scientific journals and presented 141 podium talks and posters at scientific and medical meetings.
Complete 2017 fourth quarter and full year financial results will be announced during the company's fourth quarter and fiscal year 2017 financial results conference call. The company also anticipates providing 2018 financial guidance at that time. This press release contains certain unaudited financial results for the company. These unaudited results could change as a result of further review by the company's management and its independent auditors.
Mr. Cox is scheduled to present at the 36th Annual J.P. Morgan Healthcare Conference on Monday, January 8, 2017, at 3:30 p.m. PST, in San Francisco. Additionally, Mr. Cox will participate in a panel discussion focused on the FDA and CMS parallel review process on Monday, January 8, 2018 at 5:15 p.m. PST. Live, listen-only webcasts of these presentations and the breakout session may be accessed by visiting the investors section of the company's website at investors.foundationmedicine.com. A replay of these webcasts will be available shortly after the conclusion of the presentations and will be archived on the company's website for two weeks.
About Foundation Medicine
Foundation Medicine (NASDAQ: FMI) is a molecular information company dedicated to a transformation in cancer care in which treatment is informed by a deep understanding of the genomic changes that contribute to each patient’s unique cancer. The company offers a full suite of comprehensive genomic profiling assays to identify the molecular alterations in a patient’s cancer and match them with relevant targeted therapies, immunotherapies and clinical trials. Foundation Medicine's molecular information platform aims to improve day-to-day care for patients by serving the needs of clinicians, academic researchers and drug developers to help advance the science of molecular medicine in cancer. For more information, please visit http://www.FoundationMedicine.com or follow Foundation Medicine on Twitter (@FoundationATCG).
Foundation Medicine® and FoundationACT® are registered trademarks, and FoundationOne CDx™ and FoundationCORE™ are trademarks, of Foundation Medicine, Inc.
Cautionary Note Regarding Forward-Looking Statements
This press release contains "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995, including, but not limited to, statements regarding the benefits of FDA approval and a preliminary NCD for FoundationOne CDx to Foundation Medicine, physicians, biopharmaceutical companies, and patients in the treatment of cancer; the scope and timing of any finalization of the NCD; and the ability of the company’s laboratories to support any growth or expansion plans. All such forward-looking statements are based on management's current expectations of future events and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements. These risks and uncertainties include the risks that any final NCD is not issued by CMS or if CMS is delayed in finalizing such NCD; the company’s new facilities do not facilitate the company’s ability to achieve it business objectives; the company’s distribution partner outside the United States is not able to achieve market penetration in new and existing markets as quickly or as extensively as projected; and the risks described under the caption "Risk Factors" in Foundation Medicine's Annual Report on Form 10-K for the year ended December 31, 2016, which is on file with the Securities and Exchange Commission, as well as other risks detailed in Foundation Medicine's subsequent filings with the Securities and Exchange Commission. With respect to Foundation Medicine's estimated cash, total revenue, clinical testing revenue, biopharma revenue, Molecular Information Services revenue, Pharma Research and Development Services revenue, clinical tests and other financial and business results as of and for the year ended December 31, 2017, it should be noted that this information is unaudited and that the company has not finalized its financial and business results for the three and twelve months ended December 31, 2017. All information in this press release is as of the date of the release, and Foundation Medicine undertakes no duty to update this information unless required by law.
CONTACT:
Foundation Medicine
Media Contact:
Lee-Ann
Murphy, 617-245-3077
[email protected]
or
Investor
Contact:
Kimberly Brown, 617-418-2215
[email protected]
Exhibit 99.2

Personalized Cancer Care. Delivered. JP Morgan Healthcare Conference 2018 January 8, 2018 CONFIDENTIAL
Forward Looking Statement January 8, 2018 This presentation contains forward-looking statements about our business. These statements may be identified by words such as “may,” “will,” “should,” “could,” “expect,” “intend,” “plan,” “anticipate,” “believe,” “estimate,” “predict,” “potential,” “forecast,” “continue” or the negative of these terms or other words or terms of similar meaning. All forward-looking statements are based upon estimates, forecasts, assumptions and expectations that, although we believe them to be reasonable, are inherently uncertain. All forward-looking statements are subject to risks and uncertainties that may cause actual results to differ materially from those that we expected. Risks and uncertainties that could cause our actual results to differ materially from those set forth in any forward-looking statements include, but are not limited to, the matters listed under “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2016, which is on file with the Securities and Exchange Commission, as well as other risks, uncertainties and other important factors detailed in our subsequent filings with the Securities and Exchange Commission. These filings are available at www.sec.gov or by contacting our investor relations department at [email protected]. Statements and information, including forward-looking statements, speak only to the date they are provided (unless an earlier date is indicated), and we do not undertake any obligation to publicly update any statements or information, including forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
January 8, 2018 CONFIDENTIAL

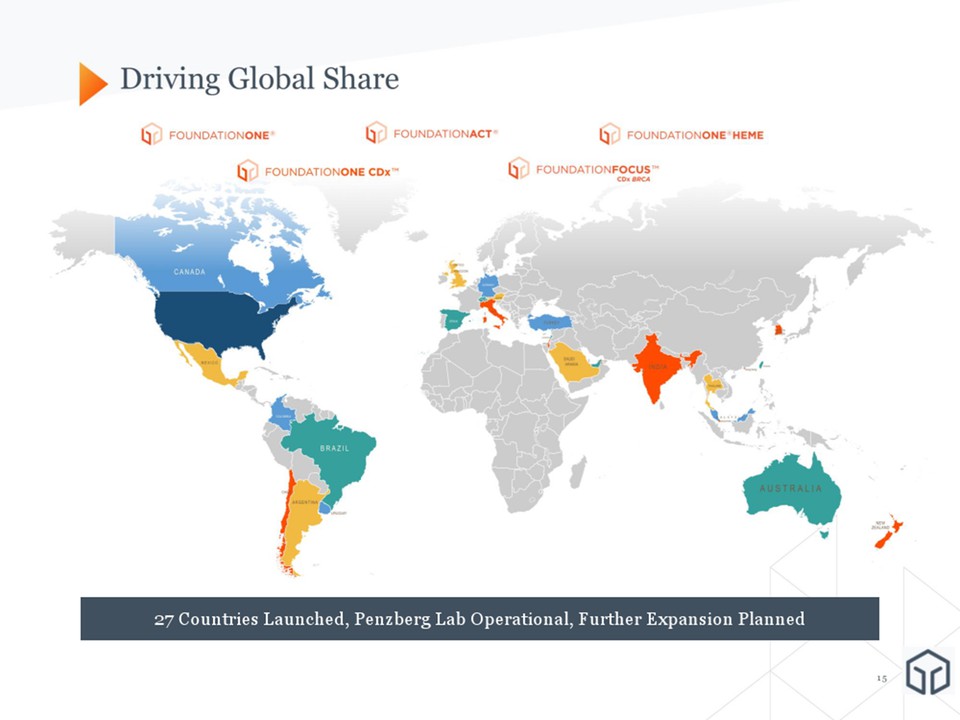
Achieved FDA approval and a preliminary National Coverage Determination (NCD) from CMS 2017: A Year of Transformational Milestones and Accomplishments January 8, 2018 Posted record clinical volume Posted record revenue Signed significant new biopharma partnerships, including in the area of Immunotherapy To date, launched in 27 countries, expanding our global footprint 67,375 tests, 54% growth $152.9M, 31% growth

The Cancer Care Gap is Enormous January 8, 2018
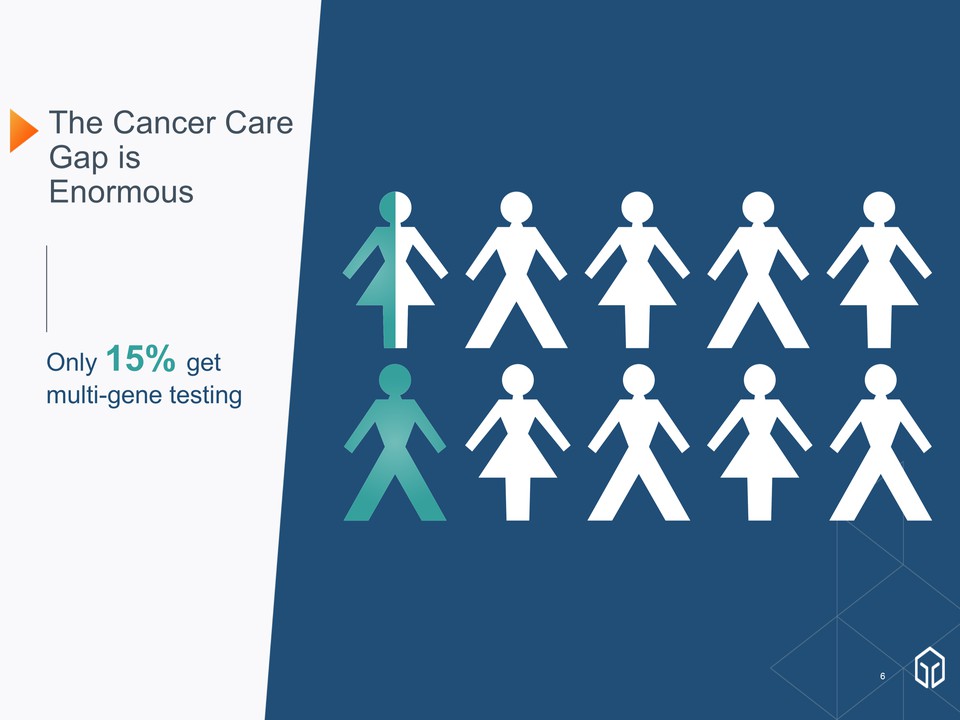
The Cancer Care Gap is Enormous January 8, 2018 Only 15% get multi-gene testing
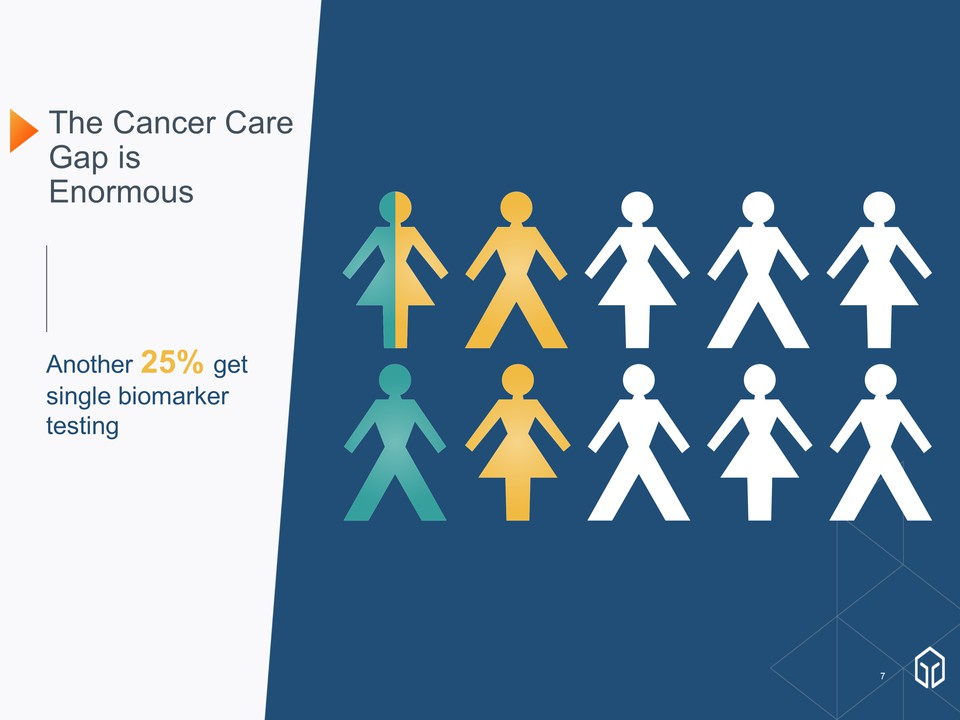
Another 25% get single biomarker testing The Cancer Care Gap is Enormous January 8, 2018

60% of patients do not receive genomic testing The Cancer Care Gap is Enormous January 8, 2018

Foundation Medicine’s Opportunity: 10 of 10 All Advanced Cancer Patients Deserve Personalized Care January 8, 2018

Foundation Medicine’s Opportunity: 10 of 10 January 8, 2018 CDx TM ONE TEST The FirstFDA Approved broad genomic profiling test with multiple companion diagnostics 4 out of 5Matched to a clinical trial* 35% Matched to a FDA approved therapy* Universal Platform for CDx Development and Access to Targeted and Immunotherapies *Estimates based on historical data of patients who received CGP testing from Foundation Medicine
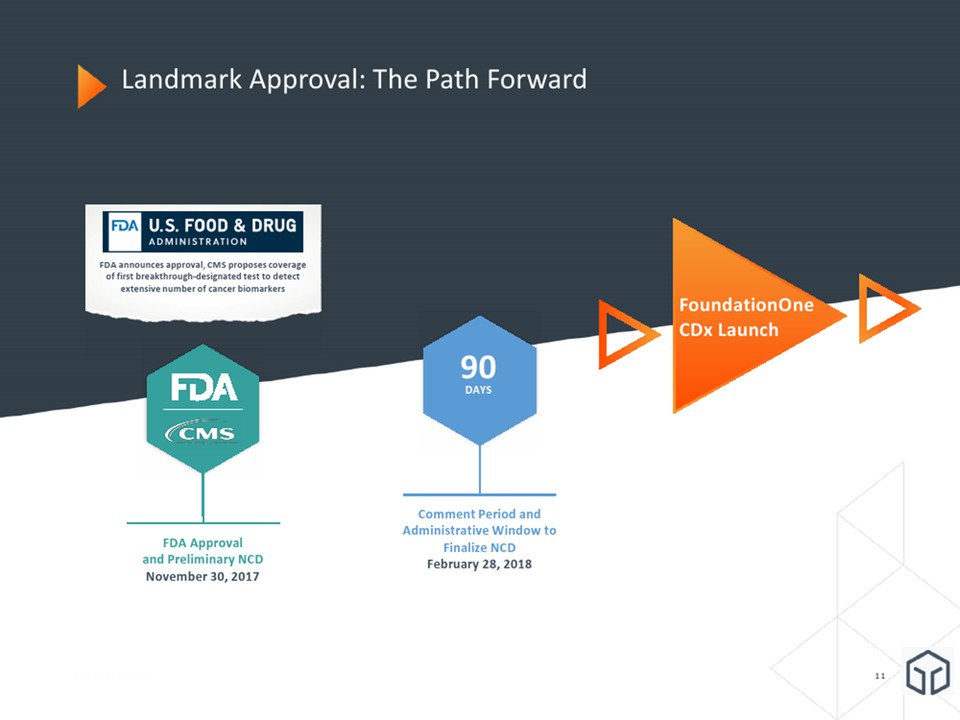
Landmark Approval: The Path Forward January 8, 2018 90DAYS Comment Period and Administrative Window to Finalize NCDFebruary 28, 2018 FDA Approvaland Preliminary NCDNovember 30, 2017 FDA announces approval, CMS proposes coverage of first breakthrough-designated test to detect extensive number of cancer biomarkers FoundationOne CDx Launch

50% of FMI’s Solid Tumor Testing*40% Medicare & Medicare Advantage* Ovarian Lung Breast Colorectal Melanoma New FDA approved CDx for additional tumor types Coverage with Evidence Development FoundationOne CDx: Meaningful Medicare Coverage through a National Coverage Determination (NCD) *Estimates are projections based on historical data contained in a genomic database of patients with NSCLC, melanoma, colorectal cancer, ovarian cancer or breast cancer who received CGP testing from Foundation Medicine
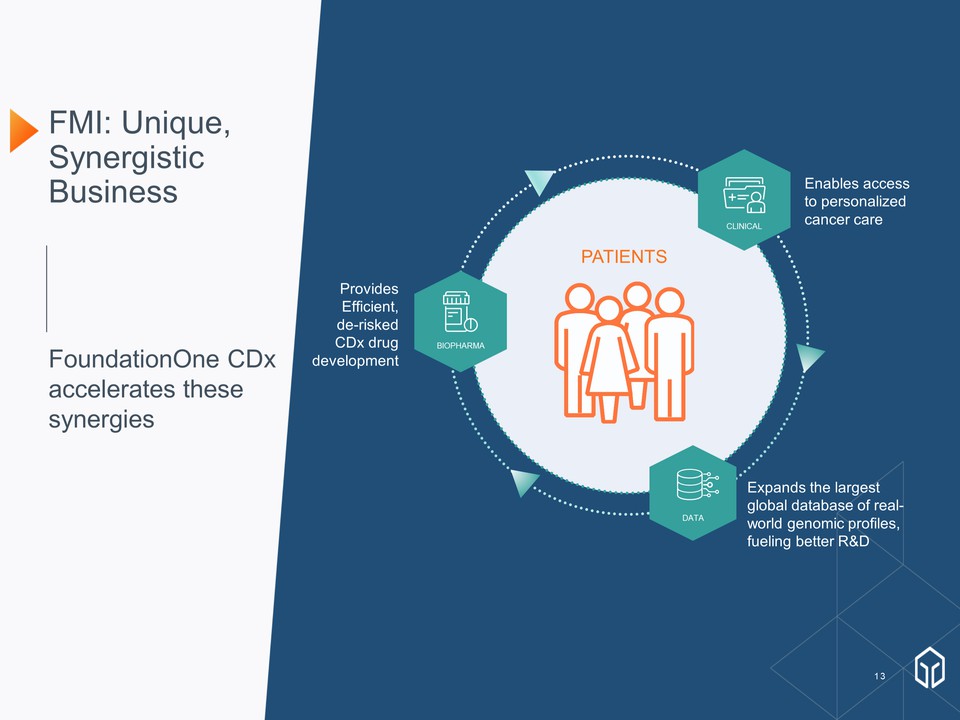
FMI: Unique, Synergistic Business FoundationOne CDx accelerates these synergies PATIENTS CLINICAL Enables access to personalized cancer care BIOPHARMA Provides Efficient, de-risked CDx drug development DATA Expands the largest global database of real-world genomic profiles, fueling better R&D

The Next Chapter of Growth DRIVE CLINICAL ADOPTION AND GLOBAL REACH CONTINUED REIMBURSEMENT PROGRESS EXPAND BIOPHARMAGROWTH 30+PARTNERS BROADEN SUITE OF TARGETED AND IMMUNOTHERAPY OFFERINGS $
January 8, 2018 CONFIDENTIAL
January 8, 2018 CONFIDENTIAL

Reimbursement Strategy January 8, 2018 Removing Barriers for Commercial Payer Expansion FDA Approval/ Preliminary NCD from CMS Professional Guidelines Momentum in Personalized Cancer Care Strengthened Commercial Capabilities
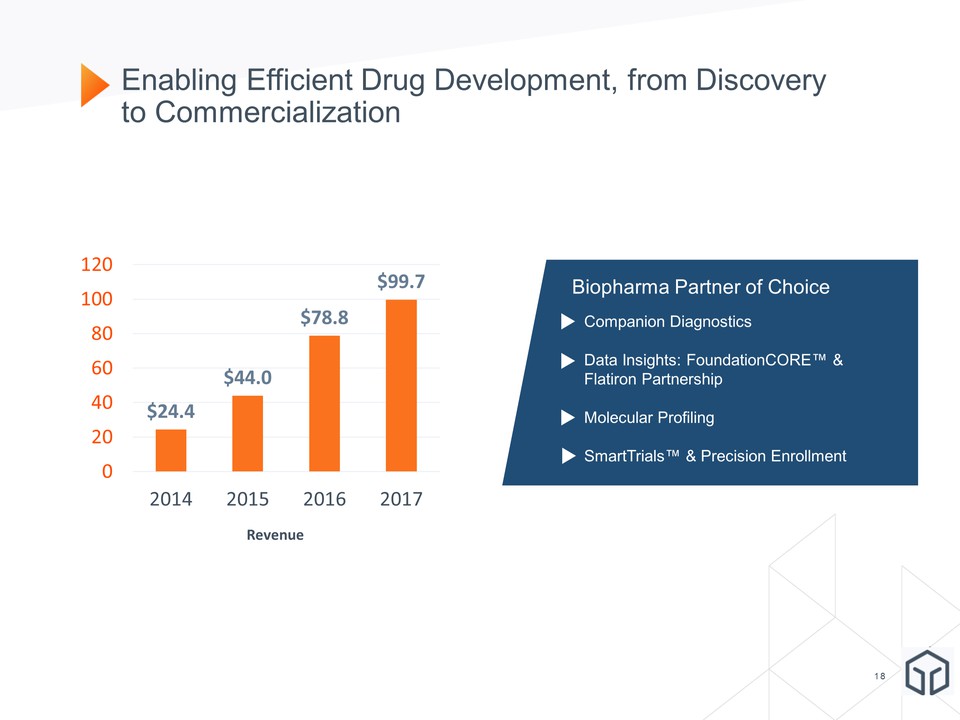
Enabling Efficient Drug Development, from Discovery to Commercialization January 8, 2018 January 8, 2018 Biopharma Partner of Choice Companion DiagnosticsData Insights: FoundationCORE™ & Flatiron PartnershipMolecular ProfilingSmartTrials™ & Precision EnrollmentCompanion Diagnostics Revenue
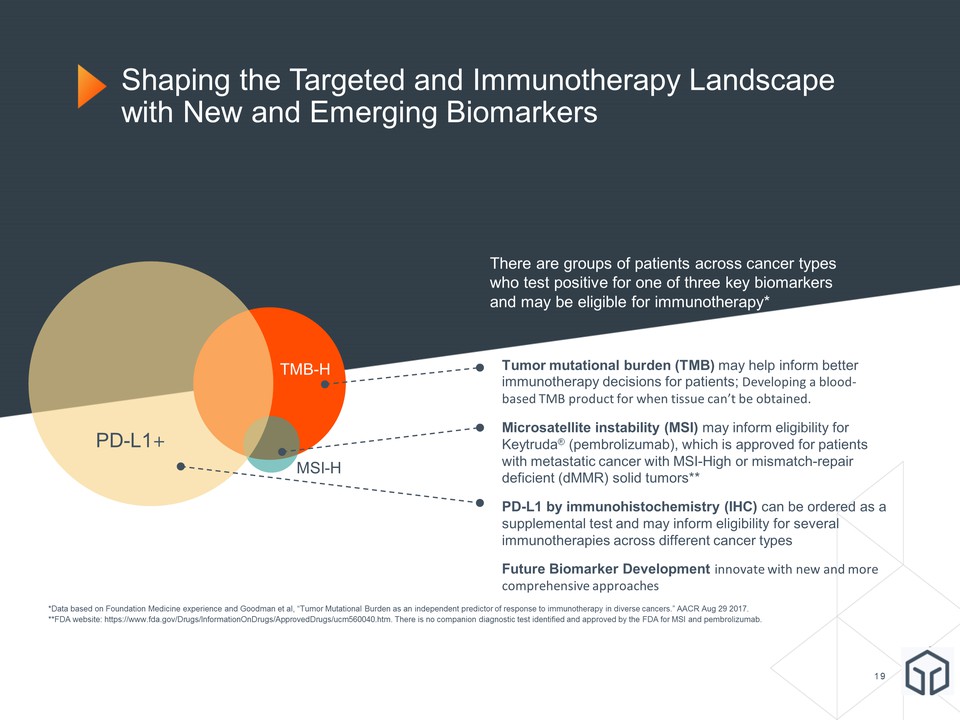
PD-L1+ TMB-H MSI-H Shaping the Targeted and Immunotherapy Landscape with New and Emerging Biomarkers *Data based on Foundation Medicine experience and Goodman et al, “Tumor Mutational Burden as an independent predictor of response to immunotherapy in diverse cancers.” AACR Aug 29 2017.**FDA website: https://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm560040.htm. There is no companion diagnostic test identified and approved by the FDA for MSI and pembrolizumab. Tumor mutational burden (TMB) may help inform better immunotherapy decisions for patients; Developing a blood-based TMB product for when tissue can’t be obtained.Microsatellite instability (MSI) may inform eligibility for Keytruda® (pembrolizumab), which is approved for patients with metastatic cancer with MSI-High or mismatch-repair deficient (dMMR) solid tumors**PD-L1 by immunohistochemistry (IHC) can be ordered as a supplemental test and may inform eligibility for several immunotherapies across different cancer typesFuture Biomarker Development innovate with new and more comprehensive approaches There are groups of patients across cancer types who test positive for one of three key biomarkers and may be eligible for immunotherapy*

Our Mission: 10 of 10
