Form 10-K CANTEL MEDICAL CORP For: Jul 31
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
☒ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended July 31, 2017
Or
☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File No. 001-31337
CANTEL MEDICAL CORP.
(Exact name of registrant as specified in its charter)
Delaware | 22-1760285 | |
(State or other jurisdiction of | (I.R.S. employer | |
incorporation or organization) | identification no.) | |
150 Clove Road, Little Falls, New Jersey | 07424 | |
(Address of principal executive offices) | (Zip code) | |
Registrant’s telephone number, including area code: (973) 890-7220
Securities registered pursuant to Section 12(b) of the Act:
Name of each exchange | ||
Title of each class | on which registered | |
Common Stock, $0.10 par value | New York Stock Exchange | |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ☒ No ☐
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. Yes ☐ No ☒
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer”, “accelerated filer”, “small reporting company,” and "emerging growth company" in Rule 12b-2 of the Exchange Act.
Large accelerated filer ☒ | Accelerated filer ☐ | Non-accelerated filer ☐ | Smaller reporting company ☐ | Emerging growth company ☐ | ||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ☒
State the aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price at which the common equity was last sold, or the average bid and asked price of such common equity, as of the last business day of the registrant’s most recently completed second fiscal quarter, as quoted by the New York Stock Exchange on that date: $2,767,926,147.
Indicate the number of shares outstanding of each of the registrant’s classes of common stock as of the close of business on August 31, 2017: 41,727,461
Documents incorporate by reference: Portions of the definitive proxy statement to be filed pursuant to Regulation 14A promulgated under the Securities Exchange Act of 1934 in connection with the 2017 Annual Meeting of Stockholders of Registrant are hereby incorporated by reference into Part III of this Form 10-K and certain documents are incorporated by reference into Part IV.
Cantel Medical Corp. 2017 Annual Report on Form 10-K
TABLE OF CONTENTS
Page No. | ||
Item 1. | ||
Item 1A. | ||
Item 1B. | ||
Item 2. | ||
Item 3. | ||
Item 4. | ||
Item 5. | ||
Item 6. | ||
Item 7. | ||
Item 7A. | ||
Item 8. | ||
Item 9. | ||
Item 9A. | ||
Item 9B. | ||
Item 10. | ||
Item 11. | ||
Item 12. | ||
Item 13. | ||
Item 14. | ||
Item 15. | ||
Item 16. | ||
Signatures. | ||
2
Cantel Medical Corp. 2017 Annual Report on Form 10-K
PART I
Item 1. Business.
Overview:
Throughout this document, references to “Cantel,” “us,” “we,” “our” and the “Company” are references to Cantel Medical Corp. and its subsidiaries, except where the context makes it clear the reference is to Cantel Medical Corp. itself and not its subsidiaries. Unless otherwise indicated, references in this Form 10-K to 2017, 2016, 2015 or “fiscal” 2017, 2016, 2015 or other years refer to our fiscal year ended July 31, of that respective year, and references to “fiscal” 2018 refer to our fiscal year ending July 31, 2018.
We are a leading provider of infection prevention products and services in the healthcare market, specializing in the following reportable segments: Endoscopy, Water Purification and Filtration, Healthcare Disposables, Dialysis and Other. Most of our products are used to help prevent the occurrence or spread of infections. We operate our four operating segments through wholly-owned subsidiaries in the United States and internationally.
Net Sales by Reportable Segment | Year Ended July 31, | |||||||||||||||||||
2017 | 2016 | 2015 | ||||||||||||||||||
Endoscopy | $ | 398,773 | 51.8 | % | $ | 341,752 | 51.4 | % | $ | 248,654 | 44.0 | % | ||||||||
Water Purification and Filtration | 196,446 | 25.5 | % | 177,669 | 26.7 | % | 173,834 | 30.8 | % | |||||||||||
Healthcare Disposables | 144,457 | 18.7 | % | 112,584 | 17.0 | % | 106,920 | 18.9 | % | |||||||||||
Dialysis | 30,481 | 4.0 | % | 32,750 | 4.9 | % | 31,240 | 5.5 | % | |||||||||||
Other(1) | — | — | % | — | — | % | 4,356 | 0.8 | % | |||||||||||
$ | 770,157 | 100.0 | % | $ | 664,755 | 100.0 | % | $ | 565,004 | 100.0 | % | |||||||||
________________________________________________
(1) | Through April 7, 2015, we had another reportable segment, known as Specialty Packaging, which included specialty packaging and thermal control products, as well as related compliance training, for the transport of infectious and biological specimens and thermally sensitive pharmaceutical, medical and other products. We divested this business during fiscal 2015. |
Information Related to Reportable Segments:
Endoscopy
General. Our Endoscopy segment designs, develops, manufactures, sells and installs a comprehensive offering of products and services comprising a complete circle of infection prevention solutions. Our products include endoscope reprocessing and endoscopy procedure products. Our endoscope reprocessing products and services include:
• | a full range of automated endoscope reprocessing systems, |
• | high-level disinfectants and sterilants, |
• | detergents, |
• | leak testing and manual cleaning products, |
• | storage cabinets, transport systems and mobile medical carts, |
• | manual cleaning products, |
• | endoscope process tracking products, |
• | other consumables, accessories and supplies used to high-level disinfect rigid endoscopes, flexible endoscopes and other instrumentation and |
• | technical maintenance service on our products. |
Our endoscopy procedure products are designed to eliminate the challenges associated with proper cleaning and high-level disinfection of numerous reusable components used in GI endoscopy procedures. Our procedure products include:
• | CO2 and water irrigation pumps and disposable procedure kits, |
• | sterile irrigation tubing and |
• | single-use valves. |
Our endoscopy products, most of which are proprietary medical devices subject to rigorous standards and regulations, contribute to the safe and effective use of endoscopes in healthcare facilities throughout the world and improve the quality of
(dollar amounts in thousands except share and per share data or as otherwise specified) 3
Cantel Medical Corp. 2017 Annual Report on Form 10-K
healthcare delivery by reducing the threat of nosocomial (hospital/healthcare facility acquired) infections. In addition, our disposable procedure products provide greater patient safety and infection prevention, through the replacement of reusable devices requiring disinfection with our single-use sterile products. In particular, such products are intended to reduce the challenges associated with proper cleaning and high-level disinfection of numerous reusable components used in gastrointestinal (GI) endoscopy procedures.
We design, develop and manufacture most of our endoscopy products. Our Endoscopy segment offers various preventative maintenance programs, repair services and user training programs to support the effective operation of reprocessing systems over their lifetime. Our field service personnel and international third-party distributors install, maintain, upgrade and repair equipment.
Sales, Marketing and Distribution. We sell and service our full line of endoscopy products through our direct field sales and clinical support service organizations in the United States, Canada, the United Kingdom, Italy, the Netherlands, Germany, France, Singapore, Malaysia and Australia. Elsewhere in Europe, Asia Pacific and Latin America, we sell primarily through independent distribution partners. In China, we sell both direct and through distributors, based on regional market demands.
Competition. We compete with a number of large companies that have significant product portfolios and global reach, as well as a number of small companies with very limited product offerings and operations in one or a limited number of countries. On a product basis, our principal competitors are Steris, Olympus, Boston Scientific, ASP (a division of Johnson & Johnson), Metrex, Ruhof, Ecolab, ERBE, Getinge, SteelCo and Wassenburg. We believe that our principal competitive advantages include the strength of our dedicated sales teams, our comprehensive product line of differentiated automated endoscope reprocessors, disposable procedure products and proprietary chemistries, and our reputation for providing high-quality and reliable products supported by our highly responsive clinical support and service teams.
Acquisitions. On August 23, 2017, we purchased all of the issued and outstanding stock of BHT Hygienetechnik Holding GmbH (“BHT Group”), a leader in the German market in automated endoscope reprocessing and related equipment and services. BHT Group consists of a portfolio of high-quality automatic endoscope reprocessors, advanced endoscope storage and drying cabinets (products globally distributed by our Company prior to the acquisition under an agreement with BHT Group), washer-disinfectors for central sterile applications, associated technical service and parts as well as flexible endoscope repair services.
On April 1, 2017, we purchased the net assets of CR Kennedy & Company Pty Ltd. (“CR Kennedy”) related to its distribution and sale of our Medivators-branded automated endoscope reprocessors, chemistries, endoscopy procedure products and other consumables in Australia, including a full sales and service organization.
On September 26, 2016, we acquired certain net assets of Vantage Endoscopy Inc. ("Vantage") related to its distribution and sale of our Medivators endoscopy products in Canada. Vantage was our exclusive distributor of Medivators' automated endoscope reprocessors and related consumables and accessories in Canada.
Water Purification and Filtration
General. Our Water Purification and Filtration segment designs, develops, manufactures, sells, and installs water purification systems for medical, pharmaceutical and other bacteria controlled applications. We also provide filtration/separation and disinfectant technologies to the medical and life science markets through a worldwide distributor network. Our products and services include:
• | central dialysis water purification systems, |
• | portable dialysis water purification systems, |
• | bicarbonate systems, |
• | hollow fiber filters and other filtration and separation products, |
• | liquid disinfectants and cold sterilization products, |
• | “dry fog” products and |
• | room temperature sterilizers. |
Our products are generally designed for dialysis and other specific healthcare applications, research laboratories and pharmaceutical, food and beverage, and commercial industrial customers. Our water systems provide biologically pure water specific to our customers’ needs and site conditions, ranging from low-volume, reverse osmosis (“RO”) and deionization systems, to high-volume, complete turnkey purification systems. We provide service and maintenance for water purification systems through an extensive network of regional offices in the United States and, to a smaller degree, in Canada.
(dollar amounts in thousands except share and per share data or as otherwise specified) 4
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Our expertise includes designing systems capable of delivering water for hemodialysis that meets the water quality standards and good manufacturing standards of the Association for the Advancement of Medical Instrumentation (“AAMI”) and all grades of U.S. Pharmacopeia (“USP”) water (i.e., water meeting the U.S. Food and Drug Administration (“FDA”) enforced standards of the USP including “USP Purified Water,” which is a FDA requirement for the labeling of “purified” bottled water). We also package these same technologies and expertise in industrial designs to meet the requirements for high-purity water in the commercial industrial markets, such as boiler feedwater production or high quality rinse water production.
We also offer a full line of proprietary and third party filters utilizing hollow fiber membrane technology to remove impurities from liquid streams for a wide range of applications. Such applications include the filtering of ultrapure water to remove endotoxins, bacteria and other contaminants in medical environments to provide protection for patients undergoing treatments that use ultrapure water. Our therapeutic filtration products include hemoconcentrators, hemofilters and specialty filters utilized for therapeutic medical applications.
Our liquid disinfectant and cold sterilant products are used in the dialysis, medical, pharmaceutical and other industries. These products include surface disinfectants as well as chemistries used to disinfect ultrapure water systems as part of overall procedures to control the contamination of systems by microorganisms and spores. Our “Dry Fog” equipment dispenses our cold sterilant products in a mist form into rooms and certain structures with complex geometries in order to achieve validated surface disinfection.
Our REVOX® sterilization solutions business provides an innovative true room-temperature vapor sterilization method for the medical device, pharmaceutical and biomedical industries. It provides customers the capability to sterilize their products at room temperature, through either contract service or on-site agreements, while reducing overall processing times and inventory and capital requirements associated with other industrial sterilization methods.
Sales, Marketing and Distribution. We generally sell our equipment on a direct basis in the United States and Canada and through third-party distributors in other international markets. We are the market leader in the supply of FDA 510(k) cleared water purification systems to the dialysis industry in North America. During fiscal 2017, a significant portion of our sales in this segment were derived from sales of products and service to dialysis clinics and hospitals in North America.
Competition. We compete with a number of large companies that have significant product portfolios and global reach, as well as a number of small companies with very limited product offerings and operations in one or a limited number of countries. On a product basis, competitors include Evoqua, IsoPure, Baxter and Steris. We believe that the ability of our Water Purification and Filtration segment to successfully compete in the water purification, filtration and disinfectant market derives from our expertise in a FDA regulated environment, our broad product offerings and the high value and quality of our products and our national service coverage.
Outside of the United States, we believe there is a trend in formal or informal bundling partnerships and arrangements between kidney dialysis machine suppliers and companies offering medical water purification systems that compete with our systems. The ability to bundle these products offers a competitive advantage to such suppliers, which include Baxter (dialysis machine)/Gambro (water system), B. Braun (dialysis machine)/Lauer (water system), and Fresenius (dialysis machine)/Vivonic (water system). The bundling approach is now being used in the United States by B. Braun/Lauer and represents a competitive threat to our dialysis water business. See Item 1A, “Risk Factors.”
Healthcare Disposables
General. We design, manufacture, sell, supply and distribute a broad selection of infection prevention healthcare products, the majority of which are single-use products used by dental practitioners. Our products include the following:
• | sterility assurance products such as biological indicators, chemical integrators and sterilization pouches, |
• | consumables such as towels, bibs, tray liners and sponges, |
• | nitrous oxide/oxygen sedation equipment and related single-use disposable nasal masks, |
• | personal barrier products such as face masks, shields, and hand protection products such as hand sanitizers and germicidal wipes, |
• | cleaning solutions, high level disinfectants and surface disinfectants, |
• | waterline treatment products for maintaining safe dental unit waterlines, |
• | treatment accessories such as saliva ejectors, evacuator tips and plastic cups and |
• | preventatives such as prophy angles and prophy paste. |
(dollar amounts in thousands except share and per share data or as otherwise specified) 5
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Significant brand names for our healthcare disposable products include SECURE FIT®, ISOFLUID®, RAPICIDE® and DentaPure® Cartridge.
Our most significant business in this segment derives from our sterility assurance business. We offer both mail-in services and in-office biological monitoring (spore test) systems enabling healthcare professionals to verify the performance of their sterilizers in accordance with the U.S. Centers for Disease Control and Prevention (“CDC”) and industry guidelines for daily or weekly testing. Our products include a wide-array of biological indicators, chemical integrators and related products and services that enable hospitals, surgical centers, office-based practitioners and dental facilities to safely and accurately monitor and verify their sterilization practices and protocols.
We maintain a leading market position in the United States for face masks and dental unit waterline treatments as well as several of our other products used in the dental market.
Sales, Marketing, and Distribution. Our healthcare disposable products are sold globally to approximately 350 wholesale customers in over 100 countries, with a significant majority located in the United States. Our distribution partners generally include major healthcare distributors, group purchasing organizations and buying co-operatives that sell our products to dental practices, medical facilities, veterinary clinics, and government and educational institutions. The majority of our healthcare disposable products are sold under the Crosstex brand name. We also produce private label products for several of our distribution partners.
Competition. We compete with a number of large companies that have significant product portfolios and global reach, as well as a number of small companies with very limited product offerings. On a product basis, competitors include Halyard Health, 3M, ASP, Steris, Danaher/Sybron, Dentsply/Sultan Healthcare, Amcor, Porter Instrument, Sterisil, ProEdge and more less expensive imported generic products from Asia and other lower cost manufacturing locations. We believe that our long-standing brands, product quality, superior customer service and breadth of portfolio are competitive advantages and are the basis for our success in this segment.
Acquisitions. On August 1, 2016, we acquired all of the issued and outstanding stock of Accutron Inc. ("Accutron"), a Phoenix-based company. Accutron designs, manufactures and sells nitrous oxide/oxygen sedation systems including flowmeters, manifold systems and related equipment and accessories as well as single-use nasal masks for use in dental procedures.
Dialysis
General. We design, develop, manufacture, sell and service reprocessing systems and sterilants for dialyzers (a device serving as an artificial kidney), as well as dialysate concentrates and supplies utilized for renal dialysis. Our renal dialysis products include:
• | medical device reprocessing systems, |
• | sterilants/disinfectants and |
• | dialysate concentrates and other supplies. |
Our renal dialysis treatment products include a line of acid and bicarbonate concentrates, referred to as dialysate concentrates, used by kidney dialysis centers to prepare dialysate, a chemical solution that draws waste products from the patient’s blood through a dialyzer membrane during the hemodialysis treatment. Dialysate concentrates are used in the dialysis process, whether single-use or reuse dialyzers (described below) are being utilized.
Our dialyzer reprocessing products are limited to use by centers that choose to clean, disinfect and reuse dialyzers for the same patient, known as “dialyzer reuse,” rather than discard the dialyzers after a single use. There has been a significant downward trend in dialyzer reuse since 2001, which has significantly decreased sales of our dialysis products tied to reuse during that period. Although the reduction of our dialysis reuse business had a material adverse effect on our Dialysis segment business during fiscal 2017, this was mitigated by increased sales of dialysate concentrate.
Today, only a small number of all dialysis procedures in the United States reuse dialyzers. The downward trend in reuse dialyzers in the United States accelerated during fiscal 2016 and 2017. The most significant manufacturers of reuse dialyzers have indicated that they will be ceasing their manufacture of such products in the near term. As such, the downward trend will likely accelerate in fiscal 2018 and significantly reduce or eliminate our sale of dialyzer reprocessors and related single-use products in the United States by the end of the fiscal year. However, we are exploring dialysis-related opportunities with the potential to mitigate the loss of such business. Likewise, we are expanding marketing efforts of reuse products in emerging markets in Asia,
(dollar amounts in thousands except share and per share data or as otherwise specified) 6
Cantel Medical Corp. 2017 Annual Report on Form 10-K
South America and elsewhere. However, no assurance can be given that such opportunities and efforts will prove successful. See Item 1A, “Risk Factors.”
Sales, Marketing and Distribution. Our products are sold in the United States and, to a significantly lesser extent, throughout the world. Our customer base is comprised of large and small dialysis chains as well as independent dialysis clinics. We sell products in the United States primarily through our own direct distribution network, and in many international markets either directly or under various third-party distribution agreements.
Competition. In our Dialysis segment, our most significant competition comes from manufacturers of single-use dialyzers, particularly Fresenius, the largest dialysis chain in the United States and a manufacturer of single-use dialyzers. All or substantially all Fresenius dialysis clinics exclusively use single-use dialyzers and therefore have no need for dialyzer reprocessing equipment.
Information with Respect to Our Business Generally:
Government Regulation
Our business and products are subject to various degrees of governmental regulation in the countries in which we operate. In the United States, the FDA, U.S. Environmental Protection Agency (“EPA”) and other governmental authorities regulate the development, manufacture, labeling, sale, storage and distribution of our products and services. Our international operations also are subject to a significant amount of government regulation, including country-specific rules and regulations and U.S. regulations applicable to our international operations. Compliance with applicable government regulations is a significant expense for us.
Numerous aspects of our business are subject to government regulations including, among other things, research and development, product approvals, product manufacturing, labeling, marketing and promotion, distribution, record-keeping, storage and disposal practices. For example, the FDA inspects medical device manufacturers for compliance with the current Quality Systems Regulations (“QSRs”), which govern the methods used in, and the facilities and controls used for, the design, manufacture, packaging and servicing of all finished medical devices intended for human use. In addition, introductions of new medical devices are generally subject to regulatory clearance or approval. Failure to receive or maintain, or delays in receiving, such clearance or approvals may hurt our competitiveness and have other material adverse consequences on our business and results of operations.
We cannot predict the effect on our operations resulting from current or future governmental regulations or the interpretation or application of these regulations. However, such governmental regulations could prevent, delay, or result in the revocation or rejection of regulatory clearance of our products. In addition, if we fail to comply with any applicable regulatory requirements, fines, sanctions, regulatory actions and other penalties could be imposed on us.
We believe that we are currently compliant in all material respects with applicable regulatory requirements. However, there can be no assurance that future or current regulatory, governmental, or private action will not have a material adverse effect on us or on our performance, results, or financial condition. See Item 1A, “Risk Factors.”
Sources and Availability of Raw Materials
We purchase raw materials, sub-assemblies, components and other supplies from numerous suppliers in the United States and abroad. The principal raw materials and supplies that we use to conduct operations include chemicals, paper, resin, stainless steel and plastic components. These raw materials are generally obtainable from several sources and in sufficient quantities within the lead times specified to vendors. As such, we do not currently foresee extraordinary difficulty in fiscal 2018 in obtaining the materials, sub-assemblies, components, or other supplies necessary for our business operations.
Pursuant to the Dodd-Frank Wall Street Reform and Consumer Protection Act (Dodd-Frank), the United States Securities and Exchange Commission (the "SEC") has implemented reporting and disclosure requirements related to public companies that use certain minerals and metals, known as “conflict minerals,” in their products. The rules require us to undertake measures to understand the origin and, as need be, source of conflict minerals within our supply chain and to report those measures and whether or not any such conflict minerals originated from the Democratic Republic of the Congo and adjoining countries. As such, the implementation of these rules could adversely affect the sourcing, availability, and pricing of materials used in our products if they are found to be sourced from that region. See "Risk Factors"
Intellectual Property
We protect our technology and products by, among other means, filing U.S. and foreign patent applications. There can be no assurance, however, that any patent will provide adequate protection for the technology, system, product, service or process
(dollar amounts in thousands except share and per share data or as otherwise specified) 7
Cantel Medical Corp. 2017 Annual Report on Form 10-K
it covers. In addition, the process of obtaining and protecting patents can be long and expensive. We also rely upon trade secrets, technical know-how and continuing technological innovation to develop and maintain our proprietary position.
As of July 31, 2017, we held 62 U.S. patents and 211 foreign patents, and had 41 U.S. patents pending and 83 foreign patents pending. The majority of our U.S. and foreign patents, for individual products, are effective for twenty years from the filing date. The actual protection afforded by a patent, which can vary from country to country, depends upon the type of patent, the scope of its coverage and the availability of legal remedies in the country. In addition, we license from independent third parties under patents, trade secrets and other intellectual property, the right to manufacture and sell certain of our products. In the aggregate, these intellectual property assets and licenses (each of which is long-term) are of material importance to our business.
Our products and services are sold around the world under various trade names, trademarks and brand names. We consider our trade names, trademarks and brand names to be valuable in the marketing of our products in each segment. As of July 31, 2017, we had a total of 2,099 trademark registrations in the United States and in various foreign countries in which we conduct business, as well as 44 trademark applications pending worldwide.
Seasonality
Our businesses generally are not seasonal in nature.
Principal Customers
None of our customers accounted for 10% or more of our consolidated net sales during fiscal 2017, 2016 or 2015. Except as described below, none of our segments are reliant upon a single customer, or a few customers, the loss of any one or more of which could have a material adverse effect on the segment.
Our Water Purification and Filtration segment is reliant on two customers, who collectively accounted for approximately 50.2% of our segment net sales during fiscal 2017.
Our Healthcare Disposables segment is reliant on three customers, who collectively accounted for approximately 43.4% of our segment net sales during fiscal 2017.
Our Dialysis segment is reliant on two customers, who collectively accounted for approximately 44.2% of our segment net sales during fiscal 2017.
Backlog
On July 31, 2017, our consolidated backlog was approximately $88,004 compared with approximately $72,415 on August 31, 2016. The majority of the backlog was in our Water Purification and Filtration segment which had backlog of $65,760 and $52,248 at July 31, 2017 and August 31, 2016, respectively. The entire backlog is expected to be recognized as revenue within one year of such date.
Competition
The markets in which our business is conducted are highly competitive. Competition is intense in all of our business segments and includes many large and small competitors. Important competitive factors generally include breadth of product offering, product design and quality, safety, ease of use, brand, product service and support, and price. We expect to face continued intense competition and believe that the long-term competitive position for all of our segments depends principally on our success in developing, manufacturing and marketing innovative, cost-effective products and services.
Many of our competitors have greater financial, technical, and human resources than we do, are well-established with reputations for success in the sale and service of their products, and may have certain other competitive advantages over us. However, we believe that the worldwide reputation for the quality and innovation of our products among customers and our reputation for providing quality product service give us a competitive advantage with respect to many of our products.
In addition, certain companies have developed, or may be expected to develop, new technologies or products that directly or indirectly compete with our products. We anticipate that we may face increased competition in the future as new infection prevention products and services enter the market. Numerous organizations are believed to be working with a variety of technologies and sterilizing agents. In addition, a number of companies have developed or are developing disposable medical instruments and
(dollar amounts in thousands except share and per share data or as otherwise specified) 8
Cantel Medical Corp. 2017 Annual Report on Form 10-K
other devices designed to address the risks of infection and contamination. There can be no assurance that new products or services developed by our competitors will not be more commercially successful than those provided or developed by us in the future.
A relatively new threat to our dialysis water business is an apparent trend, particularly outside of the United States, in formal or informal bundling partnerships and arrangements between kidney dialysis machine suppliers and companies offering medical water purification systems that compete with our systems. The ability to bundle these products offers a competitive advantage to such suppliers. A continuation of this trend could have a material effect on our business and results of operations. See Item 1A, "Risk Factors."
Research and Development
Research and development expenses (which include continuing engineering costs) increased by $2,957 to $18,367 in fiscal 2017 from $15,410 in fiscal 2016. Our research and development expenses primarily relate to development work on new products in our three largest segments, Endoscopy, Water Purification and Filtration, and Healthcare Disposables, as well as continuing engineering costs primarily related to endoscopy products.
Quality Assurance
We manufacture, assemble and package most of our products in the United States and, to a significantly lesser extent in the United Kingdom, Italy and elsewhere. Each of our production facilities is dedicated to particular processes and products. We have implemented quality assurance procedures to support the quality and integrity of our production processes.
Environmental Matters
We anticipate that our compliance with federal, state, and local laws and regulations, relating to the discharge of materials into the environment, or otherwise relating to the protection of the environment, will not have any material effect on our capital expenditures, earnings or competitive position.
Employees
As of July 31, 2017, we employed 2,337 persons, of whom 1,712 are located in the United States, 409 are located in Europe, the Middle East and Africa, 122 are located in Southeast Asia and Australia, and 94 are located in Canada. None of our employees are represented by labor unions. We consider our relations with our employees to be satisfactory.
Financial Information about Geographic Areas
Although the majority of our manufacturing is performed in the United States, we conduct manufacturing, sales, and distribution operations on a worldwide basis and are subject to a variety of risk associated with doing business internationally. These operations involve the same business segments as our domestic operations. U.S. net sales represented 77.9% of our fiscal 2017 net sales. Net sales from Europe, Middle East and Africa ("EMEA"), Asia Pacific and Canada were 12.4%, 5.3%, and 3.5%, respectively, of our fiscal 2017 net sales. The remaining 0.9% was generated in Latin American and South American regions. For a geographic presentation of net sales and other financial data for the three years ended July 31, 2017, see Note 17 to our consolidated financial statements in Part II, Item 8 of this report.
We ship certain of our products to Iran, and conduct related activities, in accordance with general or specific licenses issued by the Office of Foreign Assets Control ("OFAC") of the U.S. Department of the Treasury. The Iranian sales were generally conducted through distributors, some of whose customers may include public hospitals owned or controlled directly or indirectly by the Iranian government.
Available Information
Under the Securities Exchange Act of 1934, as amended, we are required to file with or furnish to the SEC annual, quarterly and current reports, proxy and information statements and other information. You may read and copy any document we file with the SEC at the SEC’s Public Reference Room at 100 F Street, NE, Washington, D.C. 20549. Please call the SEC at 1.800.SEC.0330 for further information about the public reference room. The SEC maintains a website at www.sec.gov that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC. We file electronically with the SEC.
(dollar amounts in thousands except share and per share data or as otherwise specified) 9
Cantel Medical Corp. 2017 Annual Report on Form 10-K
We make available, free of charge through the investor relations section of our website, our reports on Forms 10‑K, 10‑Q and 8‑K, and amendments to those reports, filed with or furnished to the SEC as soon as reasonably practicable after they are filed or furnished to the SEC. The address for our website is www.cantelmedical.com.
Also available on our website are our Corporate Governance Guidelines, Charters of the Nominating and Governance Committee, Compensation Committee and Audit Committee, and Code of Business Conduct and Ethics. Information contained on our website is not part of, and is not incorporated in, this or any other report we file or furnish to the SEC.
Forward Looking Statements
This Annual Report on Form 10-K contains “forward-looking statements” as that term is defined under the Private Securities Litigation Reform Act of 1995. These statements are based on current expectations, estimates, or forecasts about our businesses, the industries in which we operate, and the current beliefs and assumptions of management; they do not relate strictly to historical or current facts. Without limiting the foregoing, words or phrases such as “expect,” “anticipate,” “goal,” “will continue,” “project,” “intend,” “plan,” “believe,” “seek,” “may,” “could” and variations of such words and similar expressions generally identify forward-looking statements. In addition, any statements that refer to predictions or projections of our future financial performance, anticipated growth and trends in our businesses, and other characterizations of future events or circumstances are forward-looking statements. Readers are cautioned that these forward-looking statements are only predictions about future events, activities or developments and are subject to numerous risks, uncertainties, and assumptions that are difficult to predict. We caution that undue reliance should not be placed on such forward-looking statements, which speak only as of the date made. Some of the factors which could cause results to differ from those expressed in any forward-looking statement are set forth under Item 1A of this Form 10-K, entitled Risk Factors. We expressly disclaim any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in our expectations with regard thereto or any change in events, conditions or circumstances on which any such statement is based.
Item 1A. Risk Factors.
We are subject to various risks and uncertainties relating to or arising out of the nature of our businesses and general business, economic, financing, legal and other factors or conditions that may affect us. We provide the following cautionary discussion of risks and uncertainties relevant to our businesses, which we believe are factors that, individually or in the aggregate, could have a material and adverse impact on our business, results of operations and financial condition, or could cause our actual results to differ materially from expected or historical results. We note these factors for investors as permitted by the Private Securities Litigation Reform Act of 1995. You should understand that it is not possible to predict or identify all such factors. Consequently, you should not consider the following to be a complete discussion of all potential risks or uncertainties.
We face intense competition and may not be able to keep pace with the rapid technological changes in the medical device industry, which could have a material adverse effect on our business, financial condition or results of operations. The medical device markets in which we primarily participate are highly competitive. We encounter significant competition across our product lines and in each market in which our products are sold from various medical device companies, many of which may have greater financial, technical and marketing resources than we do and are well-established. Some competitors have developed or may be expected to develop technologies or products that could compete with our products or that would render our products obsolete or noncompetitive. In addition, our competitors may achieve patent protection, regulatory approval or product commercialization that would limit our ability to compete with them. Additionally, the medical device markets in which we primarily participate are characterized by extensive research and development, new product introductions and product enhancements, rapid technological change and evolving industry standards. Developments by other companies of new or improved products, processes or technologies may make our products or proposed products obsolete or less competitive and may negatively impact our net sales. Accordingly, our ability to compete is in part dependent on our ability to continually offer enhanced and improved products that meet the changing requirements of our customers. As such, we are required to devote continued efforts and financial resources to develop or acquire scientifically advanced technologies and products, apply our technologies cost-effectively across product lines and markets, obtain patent and other protection for our technologies and products, obtain required regulatory and reimbursement approvals and successfully manufacture and market our products consistent with our quality standards. If we fail to develop new products or enhance existing products, it could have a material adverse effect on our business, financial condition or results of operations.
We face increased competition in the water purification system market due to the alliance of kidney dialysis machine suppliers and water purification system suppliers. Outside of the United States, we believe there is a trend in formal or informal bundling partnerships and arrangements between kidney dialysis machine suppliers and companies offering medical water purification systems that compete with our systems. The ability to bundle these products offers a competitive advantage to such suppliers, which include Baxter (dialysis machine)/Gambro (water system), B. Braun (dialysis machine)/Lauer (water system), and Fresenius
(dollar amounts in thousands except share and per share data or as otherwise specified) 10
Cantel Medical Corp. 2017 Annual Report on Form 10-K
(dialysis machine)/Vivonic (water system). The bundling approach is now being used in the United States by B. Braun/Lauer and represents a competitive threat to our dialysis water business. If such approach expands in the United States and we do not succeed in forming an alliance with a high-quality supplier of kidney dialysis machines, we can lose our current competitive advantages and experience a material loss of net sales and a decrease in margins in our water purification system business.
The market for our dialysis reprocessing products is limited to dialysis centers that reuse dialyzers. The decrease in the reuse portion of the dialysis market in the United States accelerated significantly during fiscal 2016 and 2017 and such decrease is expected to continue. Our dialyzer reprocessing products are limited to use by clinics that choose to clean, sterilize and reuse dialyzers, rather than discard the dialyzers after a single-use. Today, only a small number of all dialysis procedures in the United States reuse dialyzers. The downward trend in reuse dialyzers in the United States accelerated during fiscal 2016 and 2017, which resulted in the sale of no reuse dialyzers in the United States during such fiscal years. Further, the most significant manufacturers of reuse dialyzers recently indicated that they will be ceasing their manufacture of such products over the next approximately twelve months. As such, clinics that currently utilize reuse dialyzers will be forced to convert to single use dialyzers, which will accelerate the downward trend in fiscal 2018 and likely eliminate our sale of dialyzer reprocessors and related single-use products in the United States by the end of the fiscal year. The reduction of our dialysis reuse business has had an adverse effect on our Dialysis segment business, which has reduced our margins and net income in this segment.
We face significant challenges in growing our dialysate concentrate sales. The reduced sales of our dialysis reuse products was significantly mitigated by increased sales in our dialysate concentrate sales during fiscal 2017, which sales are anticipated to remain at similar levels during fiscal 2018. However, no assurance can be given that we will succeed at increasing sales in the near or long term. Fresenius, the largest dialysis chain in the United States, manufactures dialysate concentrate itself and therefore provides dialysate concentrate to its own dialysis clinics. DaVita and certain international customers have also continued their reduction of dialysate concentrate purchases from us as a result of the highly competitive and price sensitive market for such product. In addition, there is increased demand in the market for powdered dialysate products principally due to the lower costs associated with shipping such products. However, we do not manufacture powdered dialysate products.
Because a significant portion of our Water Purification and Filtration and Healthcare Disposables segments net sales comes from a few large customers, any significant decrease in sales to these customers, due to industry consolidation or otherwise, could harm our operating results. In our Water Purification and Filtration segment, two customers collectively accounted for 50.2% of our fiscal 2017 net sales for this segment. The loss of a significant amount of business from either of these two customers would have a material adverse effect on our Water Purification and Filtration segment. The distribution network in the United States dental industry is concentrated, with relatively few distributors of consumable products accounting for a significant share of the sales volume to dentists. Accordingly, net sales and profitability of our Healthcare Disposables segment are highly dependent on our relationships with a limited number of large distributors. During fiscal 2017, the top three customers of our Healthcare Disposables segment accounted for 43.4% of its net sales. The loss of a significant amount of business from any of these three customers would have a material adverse effect on our Healthcare Disposables segment. In addition, because our Healthcare Disposables segment products are primarily sold through third-party distributors and not directly to end users, we cannot control the amount and timing of resources that our distributors devote to our products. There can be no assurance that there will not be a loss or reduction in business from one or more of our major customers. In addition, we cannot assure that net sales from customers that have accounted for significant net sales in the past, either individually or as a group, will reach or exceed historical levels in any future period.
Our industry is experiencing significant scrutiny and regulation by governmental authorities, which may lead to greater regulation in the future. Our medical devices and our business activities are subject to rigorous regulation, including by the FDA, EPA, Department of Justice (“DOJ”), and numerous other federal, state, and foreign governmental authorities. These authorities and members of Congress have been increasing their scrutiny of our industry. In addition, certain state governments and the federal government have enacted legislation aimed at increasing transparency of our interactions with healthcare providers. As a result, we are required by law to disclose payments and other transfers of value to healthcare providers licensed by certain states and to all U.S. physicians and U.S. teaching hospitals at the federal level. Any failure to comply with these legal and regulatory requirements could adversely impact our business. In addition, we may continue to devote substantial additional time and financial resources to further develop and implement policies, systems, and processes to comply with enhanced legal and regulatory requirements, which may also impact our business. We anticipate that governmental authorities will continue to scrutinize our industry closely, and that additional regulation may increase compliance and legal costs, exposure to litigation, and other adverse effects to our operations. Moreover, as directed by the Dodd-Frank Wall Street Reform and Consumer Protection Act (Dodd-Frank), the SEC has implemented reporting and disclosure requirements related to the use of certain minerals, known as “conflict minerals” (specifically, tantalum, tin, tungsten (or their ores), and gold) which are mined from the Democratic Republic of the Congo and adjoining countries. Pursuant to these requirements, we are required to publicly report on Form SD the procedures we employ to determine the sourcing of such minerals and metals produced from those minerals and whether we can determine if conflict minerals are used in products that we manufacture. There are costs associated with complying with these disclosure
(dollar amounts in thousands except share and per share data or as otherwise specified) 11
Cantel Medical Corp. 2017 Annual Report on Form 10-K
requirements, including for diligence in regards to the sources of any conflict minerals used in our products, in addition to the cost of remediation and other changes to products, processes, or sources of supply as a consequence of such verification activities, if appropriate. In addition, the implementation of these rules could adversely affect the sourcing, supply, and pricing of materials used in our products. As of the date of our conflict minerals report for the 2016 calendar year, although we fully complied with the regulation, we were unable to obtain the necessary information on conflict minerals from all of our suppliers and were unable to determine that all of our products are conflict free. We may continue to face difficulties in gathering this information in the future. We may face reputational challenges if we determine that certain of our products contain minerals not determined to be conflict free or if we are unable to sufficiently verify the origins for all conflict minerals used in our products through the procedures we implement.
Our planned implementation of an Enterprise Resource Planning ("ERP") software solution and other information technology systems could result in significant disruptions to our operations. We plan to implement an enterprise resource planning, or ERP, and other complementary information technology systems over the next several years. Implementation of these solutions and systems is highly dependent on coordination of numerous software and system providers and internal business teams. The interdependence of these solutions and systems is a significant risk to the successful completion of the initiatives and the failure of any one system could have a material adverse effect on the implementation of our overall information technology infrastructure. We may experience difficulties as we transition to these new or upgraded systems and processes, including loss or corruption of data, delayed shipments, decreases in productivity as our personnel and third party providers implement and become familiar with new systems, increased costs and lost revenues. In addition, transitioning to these new systems requires significant capital investments and personnel resources. Difficulties in implementing new or upgraded information systems or significant system failures could disrupt our operations and have a material adverse effect on our capital resources, financial condition, results of operations or cash flows. Implementation of this new information technology infrastructure has a significant impact on our business processes and information systems across a significant portion of our operations. As a result, we will be undergoing significant changes in our operational processes and internal controls as our implementation progresses, which in turn will require significant change management, including recruiting and training of qualified personnel. If we are unable to successfully manage these changes as we implement these systems, including harmonizing our systems, data, processes and reporting analytics, our ability to conduct, manage and control routine business functions could be negatively affected and significant disruptions to our business could occur. In addition, we could incur material unanticipated expenses, including additional costs of implementation or costs of conducting business. These risks could result in significant business disruptions or divert management’s attention from key strategic initiatives and have a material adverse effect on our capital resources, financial condition, results of operations or cash flows.
Our businesses are heavily reliant on certain raw materials and can be adversely impacted by rising prices. We purchase raw materials, sub-assemblies, components and other supplies essential to our operations from numerous suppliers in the United States and abroad. The principal raw materials that we use to conduct operations include chemicals, paper, resin, stainless steel and plastic components. From time to time we experience price increases for raw materials, with no guarantee that such increases can be passed along to our customers. In addition, although fuel and oil prices have been at relatively low levels, an increase in prices can also have a significant adverse impact on transportation costs related to both the purchasing and delivery of products and services. If costs materially increase in the future, we may not be able to implement price increases to our customers, which would adversely impact our gross margins.
The acquisition of new businesses and product lines, which has inherent risks, is an important part of our growth strategy.
We intend to grow, in part, by acquiring new products and businesses. The success of this strategy depends upon several factors, including our ability to:
• | identify and acquire appropriate products and businesses, |
• | obtain financing for acquisitions on terms that are favorable or acceptable, |
• | integrate acquired operations, personnel, products, technologies and regulatory procedures into our organization effectively, |
• | retain and motivate key personnel and retain the customers and suppliers of acquired companies, |
• | realize perceived synergies and |
• | successfully promote and increase sales and profits of acquired product lines. |
Even if acceptable financing is obtained, such financing may result in charges associated with the potential write-off of existing deferred financing costs. We also may not be able to sustain the rates of growth that we have experienced in the past, whether by acquiring businesses or otherwise. In addition, we often experience competition from third parties interested in the same acquisition candidate. This may result in increases in the price paid for acquisition candidates. In addition, assumptions regarding the growth of businesses we acquire may differ from actual results.
(dollar amounts in thousands except share and per share data or as otherwise specified) 12
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Other risks and uncertainties related to acquisitions include:
• | delays in realizing the benefits of the transactions, including achievement of anticipated operating efficiencies and synergies and other transaction benefits as well as forecasted sales and earnings, |
• | diversion of management’s time and attention, |
• | difficulties in implementing and maintaining uniform standards, controls, procedures and policies, and |
• | risks associated with the assumption of contingent or undisclosed liabilities of acquired companies. |
Given the subjective nature of the assumptions used in the determination of fair value calculations, we may potentially have significant earnings volatility in our future results of operations. In addition, we have occasionally used our stock as partial consideration for acquisitions. Our common stock may not remain at a price at which it can be used as consideration for acquisitions without diluting our existing stockholders, and potential acquisition candidates may not view our stock attractively. We have a significant amount of goodwill and intangible assets on our balance sheet related to acquisitions. If future operating results of the acquired businesses are significantly less than the results anticipated at the time of the acquisitions, we may be required to incur impairment charges.
The indemnification provisions of acquisition agreements by which we have acquired companies may not fully protect us and as a result we may face unexpected liabilities. Certain of the acquisition agreements by which we have acquired companies require the former owners to indemnify us against certain liabilities related to the operation of the company before we acquired it. In most of these agreements, however, the liability of the former owners is limited and certain former owners may be unable to meet their indemnification responsibilities. We cannot assure you that these indemnification provisions will protect us fully or at all, and as a result we may face unexpected liabilities that adversely affect our financial condition or results of operations.
Our international business subjects us to a number of risks and our limited operating experience and market recognition in new international markets may limit our international expansion strategy and cause our international return on investments and growth to suffer. Our international business subjects us to a number of risks and complications associated with manufacturing, sales, services, and other operations outside of the United States. These include: risks associated with foreign currency exchange rate fluctuations; difficulties in enforcing agreements and collecting receivables through some foreign legal systems; enhanced credit risks in certain European countries as well as emerging market regions; foreign customers with longer payment cycles than customers in the United States; tax laws that restrict our ability to use tax credits, offset gains, or repatriate funds; tariffs, exchange controls or other trade restrictions including transfer pricing restrictions when products produced in one country are sold to an affiliated entity in another country. Our future growth depends in part on our international expansion efforts, including efforts in emerging markets such as China. We have limited experience with regulatory environments and market practices internationally, and we may not be able to penetrate or successfully operate in locations and environments unfamiliar to us. Additionally, global operations are subject to risks and uncertainties, including political and economic instability, general economic conditions, imposition of government controls, the need to comply with a wide variety of foreign and U.S. export laws and trade restrictions. In connection with our expansion efforts we may encounter obstacles we did not face in North America, including cultural and linguistic differences, differences in regulatory environments, labor and market practices, difficulties in keeping abreast of market, business and technical developments, foreign customers’ requirements and preferences, and the difficulty of administering business overseas. Further, sales practices in certain international markets may be inconsistent with our desired business practices and U.S. and other legal requirements, which may impact our ability to expand as planned. We may also encounter difficulty expanding in new international markets because of competitors already entrenched in the market, and our limited brand recognition leading to delayed acceptance of our products in these new international markets. Our failure to develop new markets or disappointing growth outside of existing markets may negatively affect our return on investments relating to our international expansion efforts. In addition, we may experience difficulties in enforcing intellectual property rights or weaker intellectual property right protections in some countries.
On June 23, 2016, the United Kingdom held a referendum in which voters approved an exit from the European Union (“E.U.”), commonly referred to as “Brexit.” As a result of the referendum, it is expected that the British government will begin negotiating the terms of the U.K.’s future relationship with the E.U. Although it is unknown what those terms will be, it is possible that there will be greater restrictions on imports and exports between the U.K. and E.U. countries and increased regulatory complexities. These changes may adversely affect our operations and financial results since we have a significant presence in the U.K. Further, international markets are increasingly being affected by economic pressure to contain reimbursement levels and healthcare costs, and certain international markets may also be impacted by foreign government efforts to understand healthcare practices and pricing in other countries, which could result in increased pricing transparency across geographies and pressure to harmonize reimbursement and ultimately reduce the selling prices of our products. Most international jurisdictions have regulatory approval and periodic renewal requirements for medical devices, and countries that previously did not have regulatory requirements for medical devices may adopt such requirements; we must comply with these requirements in order to market our products in these jurisdictions. In addition, the trend in countries around the world toward more stringent regulatory requirements for product
(dollar amounts in thousands except share and per share data or as otherwise specified) 13
Cantel Medical Corp. 2017 Annual Report on Form 10-K
clearance, changing reimbursement models, and more rigorous inspection and enforcement activities has generally caused or may cause us and other medical device manufacturers to experience more uncertainty, delay, risk and expense. We expect that the international regulatory environment will continue to evolve, which could impact our ability to obtain approvals for our products in those jurisdictions, and thereby have a material impact on our business. Further, any significant changes in the competitive, political, legal, regulatory, reimbursement or economic environment where we conduct international operations may have a material impact on our business, financial condition or results of operations.
Health care policy changes on both the federal and state levels may have a material adverse effect on us. In response to perceived increases in health care costs in recent years, there have been and continue to be proposals by the federal government, state governments, regulators, and third-party payers to control these costs and, more generally, to reform the U.S. health care system. Certain of these proposals could limit the prices we are able to charge for our products or the amounts of reimbursement available for our products and could limit the acceptance and availability of our products. The adoption of some or all of these proposals could have a material adverse effect on our financial position and results of operations. In addition, the U.S. Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act, contains provisions that could have a material impact on our business. Among other provisions, this legislation imposes a 2.3% excise tax on all U.S. medical device sales. Late in 2015, Congress enacted legislation that suspended the excise tax for calendar years 2016 and 2017. Should the U.S. Congress take no further action with regard to this tax we will begin to incur excise tax in the fourth quarter of fiscal 2018. During fiscals 2016 and 2015, our total excise tax incurred was $2,035 and $4,369, respectively, which decreased our gross profit by such amounts. Furthermore, we have been required to commit significant resources to “Sunshine Act” compliance. In addition, various healthcare reform proposals have also emerged at the state level. We cannot predict with certainty what healthcare initiatives, if any, will be implemented at the state level, or what the ultimate effect of federal healthcare reform or any future legislation or regulation may have on us or on our customers’ purchasing decisions regarding our products and services.
Our stock price and trading volume has been volatile from time to time and has experienced significant fluctuations over the past several months and years as a result of various market factors. We may experience continued fluctuations in price and volume in the future that could negatively impact the value of our outstanding shares. The market for our common stock has, from time to time, experienced significant price and volume fluctuations that may have been unrelated to our operating performance. In addition, the trading market for our common stock relies in part on the research and reports that industry and other financial analysts publish about us, our business and our industry. We do not control these or any other analysts, nor do we control their respective reports. Our future operating results are subject to substantial uncertainty, and our stock price could decline significantly if we fail to meet or exceed analysts’ forecasts and expectations. If any of the analysts who cover us downgrade our stock, lower their price target or issue commentary or observations about us or our stock that are perceived by the market as negative, our stock price would likely decline rapidly. In addition, there are many other large, well-established, publicly traded companies active in our industry and market, which may cause our company to garner less attention from industry analysts. If these analysts decrease coverage or otherwise cease to cover our company, we could lose visibility in the market, which in turn could cause our stock price to decline.
Competition from lower cost manufacturing facilities such as those located in China, Southeast Asia and certain locations within North America could result in a reduction in our net sales of healthcare disposable products due to reduced average selling prices or our customers no longer purchasing certain products from us. Despite expensive shipping costs, quality concerns, sustainability issues and other matters, some of our competitors manufacture certain healthcare disposable products in lower cost locations such as China, Southeast Asia and certain locations within North America. Although we believe the quality of our healthcare disposable products, which are generally produced in the United States, are superior, our sales in the future may be adversely affected by either loss of sales or reductions in the prices of our products as a result of this lower cost competition. Price erosion resulting from lower cost competition did not have a material adverse impact on our business during fiscal year 2017, but no assurance can be given that we will not face increased competition in the future.
We are subject to extensive government regulation, which may delay or prevent new product introduction and subject us to citations, fines and other regulatory actions. Our operations are subject to extensive regulation by governmental and private agencies in both the United States and in other countries where we do business. In the United States, our products and services are regulated by the FDA and other regulatory authorities. In many foreign countries, sales of our products are subject to extensive regulations that may or may not be comparable to those of the FDA. In Europe, our products are regulated primarily by country and community regulations of those countries within the European Economic Area and must conform to the requirements of those authorities. The regulatory agencies regulate the testing, manufacturing, recordkeeping, storage, packaging, labeling, marketing, distribution, marketing, reporting, safety and import and export of medical supplies and devices. Certain international regulatory bodies also impose import restrictions, tariff regulations, duties and tax requirements. In general, unless an exemption applies, a medical device or product or service must receive regulatory approval or clearance before it can be marketed or sold. Delays in agency review can significantly delay new product introduction and may result in a product becoming “dated” or losing its market
(dollar amounts in thousands except share and per share data or as otherwise specified) 14
Cantel Medical Corp. 2017 Annual Report on Form 10-K
opportunity before it can be introduced. In addition, the FDA and other agency clearances generally are required before we can make significant modifications to existing products or market new claims or uses for existing products. The FDA also has the authority to require a recall or modification of products in the event of a defect or other issues. The process of obtaining marketing clearances and approvals from regulatory agencies for new products (or modifications to, or additional claims or uses for, existing products) can be time consuming and expensive. There is no assurance that clearances or approvals will be granted or that agency review will not involve delays that would adversely affect our ability to commercialize our products. During the past several years, the FDA, in accordance with its standard practice, has conducted a number of inspections of our manufacturing facilities to ensure compliance with regulatory standards relating to our testing, manufacturing, storage and packaging of products. On occasion, following an inspection, the FDA has called our attention to certain “Good Manufacturing Practices” compliance deficiencies. If we fail to meet QSRs or violate applicable FDA, EPA or other laws or regulations or if any of our medical devices are found to be ineffective or pose an unreasonable health risk, or if we fail to adequately correct violations or comply with requests by regulatory agencies, we could be subject to reports or warning letters, citations and fines as well as additional regulatory action including an order to recall, replace, repair, or refund non-compliant medical devices. Further, regulatory agencies could detain or seize adulterated or misbranded medical devices, or ban such medical devices. The regulatory agencies may also impose operating restrictions, enjoin and/or restrain certain conduct resulting in violations of applicable law pertaining to medical devices, including a hold on approving new devices until issues are resolved to its satisfaction, and assess civil or criminal penalties against our officers, employees, or us. The regulatory agencies may also recommend prosecution to the DOJ. Federal, state and foreign regulations regarding the manufacture and sale of our products are subject to change. We cannot predict what impact, if any, such changes might have on our business. In addition, there can be no assurance that regulation of our products will not become more restrictive in the future and that any such development would not have a material adverse effect on our business.
Compliance with international laws and regulations, import and export limitations, anti-corruption laws, and exchange controls may be difficult, burdensome and expensive. We are subject to compliance with various laws and regulations, including the U.S. Foreign Corrupt Practices Act, the U.K. Bribery Act, and similar anti-bribery laws, which generally prohibit companies and their intermediaries from making bribes or other improper payments to officials for the purpose of obtaining or retaining business. We are also subject to limitations on trade with persons in sanctioned countries. While our employees, distributors and agents are required to comply with these laws, no assurance can be given that our training and internal policies and procedures will always protect us from violations of these laws, despite our commitment to legal compliance and corporate ethics. The failure to comply with these laws and regulations could subject us to severe fines and penalties material in scope.
Our operations could be adversely impacted by changes in the U.S. political environment. The 2016 presidential and congressional elections in the United States have resulted in significant uncertainty with respect to, and could result in changes in, legislation, regulation and government policy at the federal level, as well as the state and local levels. Any such changes could significantly impact our business as well as the markets in which we compete. Specific legislative and regulatory proposals discussed during election campaigns and more recently that might materially impact us include, but are not limited to, changes to existing trade agreements, import and export regulations, tariffs and customs duties, income tax regulations and the federal tax code, healthcare delivery and spending, public company reporting requirements, environmental regulation and antitrust enforcement.
Our operations, products and services expose us to the risk of environmental, health and safety liabilities, costs and violations that could adversely affect our reputation and financial results. In the ordinary course of certain of our manufacturing processes, we use various chemicals and other regulated substances. Our operations, products and services are subject to environmental laws and regulations, which impose limitations on the discharge of pollutants into the environment and establish standards for the use, generation, treatment, storage and disposal of hazardous and non-hazardous wastes. Although we are not aware of any material claims involving violation of environmental or occupational health and safety laws or regulations, there can be no assurance that such a claim may not arise in the future, which could have a material adverse effect on us. We must also comply with various health and safety regulations in the United States and abroad in connection with our operations. We can give no assurance that our environmental, health and safety compliance programs have been or will at all times be effective. Failure to comply with any of these laws and regulations could result in civil and criminal, monetary and non-monetary penalties and damage to our reputation. In addition, we cannot provide assurance that our costs of complying with current or future environmental protection and health and safety laws and regulations will not exceed our estimates or adversely affect our financial condition or results of operations. In addition, we may incur costs related to remedial efforts or alleged environmental damage associated with past or current waste disposal practices or other hazardous materials handling practices. We are also from time to time party to personal injury or other claims brought by private parties alleging injury due to the presence of or exposure to hazardous substances. We may also become subject to additional remedial, compliance or personal injury costs due to future events such as changes in existing laws or regulations, changes in agency direction or enforcement policies, developments in remediation technologies, changes in the conduct of our operations and changes in accounting rules. We cannot assure you that any liabilities arising from past or future releases of, or exposures to, hazardous substances will not adversely affect our reputation or adversely affect our financial condition or results of operations.
(dollar amounts in thousands except share and per share data or as otherwise specified) 15
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Healthcare cost containment pressures and legislative or administrative reforms resulting in restrictive reimbursement practices of third-party payors or preferences for alternate therapies could decrease the demand for our products, the prices which customers are willing to pay for those products and the number of procedures performed using our devices, which could have an adverse effect on our business, financial condition or results of operations. Many of our products are purchased by hospitals, physicians and other healthcare providers that typically bill various third-party payors, including governmental programs (e.g., Medicare and Medicaid), private insurance plans and managed care programs, for the healthcare services provided to their patients. The ability of customers to obtain appropriate reimbursement for (or associated with) their products and services from private and governmental third-party payors is critical to the success of medical device companies. The availability of reimbursement affects which products customers purchase and the prices they are willing to pay. Reimbursement varies from country to country and can significantly impact the acceptance of new products and services. Even if we offer a promising new product, we may find limited demand for the product unless reimbursement approval is obtained from private and governmental third-party payors for such product (or associated with its use). Further legislative or administrative reforms to the reimbursement systems in the United States and foreign countries in a manner that significantly reduces reimbursement for procedures using our medical devices or denies coverage for those procedures, including price regulation, competitive pricing, coverage and payment policies, comparative effectiveness of therapies, technology assessments and managed-care arrangements, could have a material adverse effect on our business, financial condition or results of operations.
Currency fluctuations and trade barriers could adversely affect our results of operations. A portion of our products in all of our business segments are exported to and imported from a variety of geographic locations, and our business could be materially and adversely affected by the imposition of trade barriers, fluctuations in the rates of exchange of various currencies, tariff increases and import and export restrictions, affecting all of such geographies including but not limited to the United States, Canada, the European Union, the United Kingdom, Australia, and Asia. Changes in the value of the Euro, British Pound, Canadian dollar, Australian dollar, Singapore dollar and Chinese Renminbi against the U.S. dollar affect our results of operations because certain cash bank accounts, accounts receivable and liabilities of Cantel and its subsidiaries are denominated and ultimately settled in U.S. dollars, Euros, British Pounds, Canadian dollars, Australian dollars, Singapore dollars or Chinese Renminbi but must be converted into each entity’s functional currency. Furthermore, the financial statements of our Italy, the Netherlands, United Kingdom, Canada, Australia and China subsidiaries are translated using the accounting policies described in Note 2 to our consolidated financial statements in Part II, Item 8 of this report, and therefore are impacted by changes in the Euro, British Pound, Canadian dollar, Australian dollar and Chinese Renminbi exchange rates relative to the U.S. dollar.
We may be exposed to product liability claims resulting from the use of products we sell and distribute. We may be exposed to product liability claims resulting from the products we sell and distribute. We maintain product liability insurance, which we believe is adequate for our businesses. However, there can be no assurance that insurance coverage for these risks will continue to be available or, if available, that it will be sufficient to cover potential claims or that the present level of coverage will continue to be available at a reasonable cost. A partially or completely uninsured successful claim against us could have a material adverse effect on us. In addition, we may not have insurance covering claims of emotional harm or mental distress related to our products or services when not associated with physical injury. This could result in our incurring significant uninsured damages.
We rely on intellectual property and proprietary rights to maintain our competitive position. We rely heavily on proprietary technology that we protect primarily through licensing arrangements, patents, trade secrets and proprietary know-how. There can be no assurance that any pending or future patent applications will be granted or that any current or future patents, regardless of whether we are an owner or a licensee of the patent, will not be challenged, rendered unenforceable, invalidated or circumvented or that the rights will provide a competitive advantage to us. There can also be no assurance that our trade secrets or non-disclosure agreements will provide meaningful protection of our proprietary information. There can also be no assurance that others will not independently develop similar technologies or duplicate any technology developed by us or that our technology will not infringe upon patents or other rights owned by others.
Breaches of our information technology systems could have a material adverse effect on our operations. We rely on information technology systems to process, transmit, and store electronic information in our day-to-day operations. Our information technology systems have been subjected to computer viruses, or other malicious codes, and cyber or phishing attacks. Although past attacks did not have a significant adverse impact on our business, these types of attacks could result in our intellectual property and other confidential information being lost or stolen, disruption of our operations, or other negative consequences, such as increased costs for security measures or remediation costs, diversion of management attention and adverse impact on our relationships with vendors and customers. Cyber attacks are becoming more sophisticated and frequent and the techniques used in such attacks change rapidly. There can be no assurances that our protective measures will prevent future attacks that could have a significant impact on our business.
(dollar amounts in thousands except share and per share data or as otherwise specified) 16
Cantel Medical Corp. 2017 Annual Report on Form 10-K
If we are unable to retain key personnel, our business could be adversely affected. Our success is dependent to a significant degree upon the efforts of key members of our management. Although none of our key executives has an employment agreement with the Company, each executive, including division CEOs, is party to a severance agreement with the Company. In addition, we have short and long term incentive plans for our key executives that are designed in part to have a retentive effect on the executives. However, there can be no assurance that the terms of the severance agreements or incentive plans will have such an effect. We believe the loss or unavailability of any such individuals could have a material adverse effect on our business. In addition, our success depends in large part on our ability to attract and retain highly qualified scientific, technical, sales, marketing and other personnel. Competition for such personnel is intense and there can be no assurance that we will be able to attract and retain the personnel necessary for the development and operation of our businesses.
Some of our facilities are located near coastal zones, and the occurrence of a hurricane or other natural disasters could damage our facilities and equipment, which could harm our operations. Some of our facilities are vulnerable to damage from hurricanes and from other types of disasters, including fire, floods, power loss, communications failures, terrorism and similar events since any insurance we may maintain may not be adequate to cover our losses. If any disaster were to occur, our ability to operate our business at our facilities could be seriously, or potentially completely, impaired.
Item 1B. Unresolved Staff Comments.
None.
(dollar amounts in thousands except share and per share data or as otherwise specified) 17
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Item 2. Properties.
Our corporate headquarters are located at 150 Clove Road, Little Falls, NJ. Listed below are our manufacturing facilities and the principal warehouses, distribution centers, research facilities and administrative offices that we own or lease. In addition, we maintain administrative and sales offices and warehousing and distribution centers in other locations domestically and globally. We believe that our properties are suitable and adequate for the manufacture and distribution of our products.
Location | Owned/Leased | Purpose | Square Footage | Segment | ||||
Plymouth, MN | Owned | Executive, administrative and sales staff, research, manufacturing and warehousing | 110,000 | Endoscopy, Dialysis, Water Purification and Filtration | ||||
Plymouth, MN | Owned | Manufacturing, warehousing and vacant land | 65,000 | Endoscopy, Dialysis, Water Purification and Filtration | ||||
Hauppauge, NY | Owned | Executive, administrative and sales staff, manufacturing and warehousing | 65,000 | Healthcare Disposables | ||||
Conroe, TX | Owned | Manufacturing, warehousing and administrative, sales and other staff | 60,000 | Endoscopy | ||||
Hauppauge, NY | Leased | Warehousing | 52,000 | Healthcare Disposables | ||||
Sharon, PA | Leased | Manufacturing and warehousing | 50,000 | Healthcare Disposables | ||||
Southend-on-Sea, England | Owned | Manufacturing, warehousing and administrative offices | 49,500 | Endoscopy | ||||
Pomezia, Italy | Owned | Manufacturing, warehousing and administrative offices | 48,000 | Endoscopy | ||||
Plymouth, MN | Owned | Manufacturing, warehousing, administrative and sales staff | 43,000 | Water Purification and Filtration | ||||
Lawrenceville, GA | Leased | Manufacturing and warehousing | 41,000 | Healthcare Disposables | ||||
Rush, NY | Owned | Manufacturing, warehousing and administrative, sales and other staff | 38,000 | Healthcare Disposables | ||||
Phoenix, AZ | Leased | Manufacturing, administrative offices and warehousing | 37,000 | Healthcare Disposables | ||||
Santa Fe Springs, CA | Leased | Manufacturing and warehousing | 32,000 | Healthcare Disposables | ||||
Heerlen, the Netherlands | Leased | Sales and service offices, warehouse and distribution hub | 26,000 | Various | ||||
Lowell, MA | Leased | Sales and administrative offices, manufacturing, warehousing and regeneration plant | 26,000 | Water Purification and Filtration | ||||
Skippack, PA | Leased | Sales and administrative offices, manufacturing, warehousing and regeneration plant | 23,000 | Water Purification and Filtration | ||||
Burlington, Ontario | Leased | Sales and administrative offices, research and engineering, manufacturing and warehousing | 22,000 | Water Purification and Filtration | ||||
Clevedon, England | Leased | Administrative offices, manufacturing and warehousing | 20,000 | Endoscopy | ||||
Cuba, NY | Leased | Administrative offices, manufacturing, warehousing and laboratory | 19,000 | Healthcare Disposables | ||||
Conroe, TX | Leased | Executive, sales and finance offices, research and development, training | 18,000 | Endoscopy | ||||
Markham, Ontario | Leased | Administrative offices, manufacturing, and warehousing | 16,000 | Water Purification and Filtration | ||||
Mebane, NC | Leased | Administrative offices and warehousing | 16,000 | Water Purification and Filtration | ||||
Buena Park, CA | Owned | Warehousing and regeneration plan | 14,000 | Water Purification and Filtration | ||||
Conroe, TX | Owned | Manufacturing and vacant land | 12,000 | Endoscopy | ||||
18
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Item 3. Legal Proceedings.
In the normal course of business, we are subject to pending and threatened legal actions. It is our policy to accrue for amounts related to these legal matters if it is probable that a liability has been incurred and an amount of anticipated exposure can be reasonably estimated. We do not believe that any of these pending claims or legal actions will have a material adverse effect on our business, financial condition, results of operations or cash flows.
Item 4. Mine Safety Disclosures.
Not applicable.
19
Cantel Medical Corp. 2017 Annual Report on Form 10-K
PART II
Item 5. Market for Registrant's Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities.
Our common stock trades on the New York Stock Exchange ("NYSE") under the symbol “CMD.” The following table sets forth, for the periods indicated, the high and low sales prices for the common stock as reported by the NYSE.
HIGH | LOW | ||||||
Fiscal Year Ended July 31, 2017 | |||||||
First Quarter | $ | 81.39 | $ | 68.19 | |||
Second Quarter | 85.85 | 69.37 | |||||
Third Quarter | 85.31 | 71.41 | |||||
Fourth Quarter | 81.02 | 70.19 | |||||
Fiscal Year Ended July 31, 2016 | |||||||
First Quarter | $ | 59.72 | $ | 48.38 | |||
Second Quarter | 66.45 | 56.18 | |||||
Third Quarter | 72.21 | 57.33 | |||||
Fourth Quarter | 73.88 | 64.55 | |||||
On August 31, 2017, we had 409 record holders of common stock. A number of such holders of record are brokers and other institutions holding shares of common stock in “street name” for more than one beneficial owner.
The following table represents information with respect to purchases of common stock made by the Company during the fourth quarter of fiscal 2017:
Period | Total number of shares purchased | Average price paid per share | Total number of shares purchased as part of publicly announced plans or programs | Maximum number of shares that may yet be under the plan or programs | ||||||||
May 1 - May 31 | 2,579 | (1) | $ | 71.49 | — | — | ||||||
June 1 - June 30 | 3,776 | (1) | 75.91 | — | — | |||||||
July 1 - July 31 | 679 | (1) | 76.26 | — | — | |||||||
7,034 | (1) | $ | 74.32 | — | — | |||||||
_______________________________________________
(1) | The Company does not currently have a share repurchase program. All of the shares purchased during the fourth quarter of fiscal 2017 represent shares surrendered to the Company to pay employee withholding taxes due upon the vesting of restricted stock. |
Dividends
During fiscal 2017, we paid semi-annual cash dividends totaling $0.14 per outstanding share of common stock, of which $0.07 per share was paid on each of January 27, 2017 and July 27, 2017. During fiscal 2016, we paid semi-annual cash dividends totaling $0.12 per outstanding share of common stock, of which $0.06 per share was paid on each of January 29, 2016 and July 29, 2016. Future declaration of dividends and the establishment of future record and payment dates are subject to the final determination of the Company’s Board of Directors. However, it is our current expectation that semi-annual cash dividends of at least $0.07 per common share will continue to be paid in the foreseeable future.
(dollar amounts in thousands except share and per share data or as otherwise specified) 20
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Stock Performance Graph
The following graph compares the cumulative total stockholder return on our common stock for the last five fiscal years with the cumulative total returns of the Russell 2000 index and the Dow Jones U.S. Health Care Equipment & Services index over the same period (assuming an investment of $100 in our common stock and in each of the indexes on July 31, 2012, and where applicable, the reinvestment of all dividends).
Comparison of 5 Year Cumulative Total Return
Among Cantel Medical Corp. Common Stock, the Russell 2000 Index
and the Dow Jones U.S. Health Care Equipment & Services Index
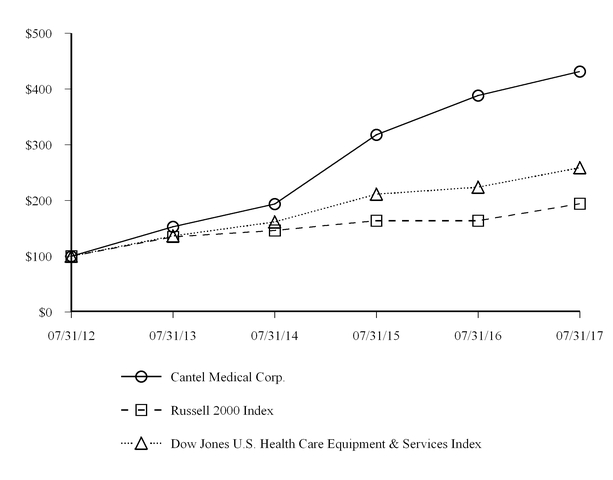
July 31, | |||||||||||||||||||||||
2012 | 2013 | 2014 | 2015 | 2016 | 2017 | ||||||||||||||||||
Cantel Medical Corp.(1) | $ | 100.00 | $ | 153.06 | $ | 193.87 | $ | 317.98 | $ | 388.66 | $ | 431.54 | |||||||||||
Russell 2000 Index | $ | 100.00 | $ | 134.76 | $ | 146.30 | $ | 163.89 | $ | 163.89 | $ | 194.14 | |||||||||||
Dow Jones U.S. Health Care Equipment & Services Index | $ | 100.00 | $ | 136.23 | $ | 161.32 | $ | 211.37 | $ | 223.65 | $ | 258.63 | |||||||||||
________________________________________________
(1) | $100 invested on July 31, 2012 in Cantel Medical Corp.'s common stock or index, including reinvestment of dividends. |
Indexes calculated on month-end basis.
(dollar amounts in thousands except share and per share data or as otherwise specified) 21
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Item 6. Selected Consolidated Financial Data.
July 31, | |||||||||||||||||||
Consolidated Statements of Income Data | 2017 | 2016 | 2015 | 2014 | 2013 | ||||||||||||||
Net sales | $ | 770,157 | $ | 664,755 | $ | 565,004 | $ | 488,749 | $ | 425,026 | |||||||||
Cost of Sales | 402,997 | 355,569 | 311,537 | 275,450 | 241,550 | ||||||||||||||
Gross profit | 367,160 | 309,186 | 253,467 | 213,299 | 183,476 | ||||||||||||||
Income from operations | 110,410 | 97,251 | 80,761 | 70,928 | 63,188 | ||||||||||||||
Interest expense, net | 4,303 | 3,320 | 2,364 | 2,317 | 2,834 | ||||||||||||||
Other income | (126 | ) | — | — | — | — | |||||||||||||
Loss on sale of business | — | — | 2,206 | — | — | ||||||||||||||
Income before income taxes | 106,233 | 93,931 | 76,191 | 68,611 | 60,354 | ||||||||||||||
Income taxes | 34,855 | 33,978 | 28,238 | 25,346 | 21,115 | ||||||||||||||
Net income | $ | 71,378 | $ | 59,953 | $ | 47,953 | $ | 43,265 | $ | 39,239 | |||||||||
Earnings per share data: | |||||||||||||||||||
Weighted average basic shares outstanding | 41,723,214 | 41,684,376 | 41,518,173 | 41,309,881 | 40,907,712 | ||||||||||||||
Weighted average diluted shares outstanding | 41,797,492 | 41,730,557 | 41,581,306 | 41,469,566 | 41,196,719 | ||||||||||||||
Basic earnings per common share | $ | 1.71 | $ | 1.44 | $ | 1.16 | $ | 1.05 | $ | 0.96 | |||||||||
Diluted earnings per common share | $ | 1.71 | $ | 1.44 | $ | 1.15 | $ | 1.04 | $ | 0.95 | |||||||||
Dividends per common share | $ | 0.14 | $ | 0.12 | $ | 0.10 | $ | 0.09 | $ | 0.07 | |||||||||
Other Financial Data | |||||||||||||||||||
Net cash provided by operating activities | $ | 108,193 | 80,268 | $ | 59,070 | $ | 64,272 | $ | 51,494 | ||||||||||
Capital expenditures | 27,065 | 18,889 | 12,760 | 13,541 | 6,745 | ||||||||||||||
Acquisition of businesses, net of cash acquired | 70,044 | 94,528 | 43,567 | 33,547 | 45,071 | ||||||||||||||
Depreciation | 15,045 | 11,989 | 10,692 | 8,245 | 7,202 | ||||||||||||||
Amortization | 18,407 | 13,095 | 13,265 | 10,641 | 10,061 | ||||||||||||||
Consolidated Balance Sheets Data | |||||||||||||||||||
Cash and cash equivalents | $ | 36,584 | $ | 28,367 | $ | 31,720 | $ | 31,781 | $ | 34,076 | |||||||||
Total assets | 786,373 | 694,532 | 584,031 | 536,145 | 487,671 | ||||||||||||||
Working capital | 150,592 | 126,407 | 117,737 | 97,410 | 91,509 | ||||||||||||||
Long-term debt | 126,000 | 116,000 | 78,500 | 80,500 | 85,000 | ||||||||||||||
Stockholders' equity | 523,932 | 454,370 | 406,633 | 365,246 | 321,132 | ||||||||||||||
(dollar amounts in thousands except share and per share data or as otherwise specified) 22
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Item 7. Management's Discussion and Analysis of Financial Condition and Results of Operations.
The following Management’s Discussion and Analysis of Financial Condition and Results of Operations (“MD&A”) is intended to help you understand Cantel and its subsidiaries. The MD&A is provided as a supplement to and should be read in conjunction with the consolidated financial statements and the accompanying notes included elsewhere in this report.
Overview
Cantel is a leading provider of infection prevention products and services in the healthcare market, specializing in the following reportable segments: Endoscopy, Water Purification and Filtration, Healthcare Disposables and Dialysis. Most of our equipment, consumables and supplies are used to help prevent the occurrence or spread of infections. We operate our four operating segments through wholly-owned subsidiaries in the United States and internationally.
Fiscal 2017 Highlights
Some of our key financial results for fiscal 2017 compared with fiscal 2016 were as follows:
• | Net sales increased by 15.9% to $770,157 from $664,755, with organic sales growth of 11.0%, |
• | Net income increased by 19.1% to $71,378 from $59,953, |
• | Non-GAAP net income* increased by 18.9% to $86,740 from $72,938, |
• | Diluted EPS increased by 18.9% to $1.71 from $1.44, |
• | Non-GAAP diluted EPS* increased by 18.7% to $2.08 from $1.75, and |
• | Adjusted EBITDAS* increased by 17.0% to $161,466 from $137,949. |
* See Non-GAAP Financial Measures below.
Recent Developments
On August 23, 2017, we purchased all of the issued and outstanding stock of BHT Hygienetechnik Holding GmbH (“BHT Group”), a leader in the German market in automated endoscope reprocessing and related equipment and services, for total consideration, excluding acquisition related costs, of $61,236. The BHT Group consists of a portfolio of high-quality automatic endoscope reprocessors, advanced endoscope storage and drying cabinets (products globally distributed by our Company prior to the acquisition under an agreement with BHT Group), washer-disinfectors for central sterile applications, associated technical service and parts as well as flexible endoscope repair services. BHT Group will be included in our Endoscopy segment.
Acquisitions
Fiscal 2017
On April 1, 2017, we purchased certain endoscopy-related net assets of CR Kennedy, related to its distribution and sale of our Medivators endoscopy products in Australia for total consideration, excluding acquisition related costs, of $11,999. The CR Kennedy business includes a full sales and service organization and our exclusive distributor of Medivators-branded automated endoscope reprocessors, chemistries, endoscopy procedure products and other consumables in Australia, and is included in our Endoscopy segment.
On September 26, 2016, we acquired certain net assets of Vantage, related to its distribution and sale of our Medivators endoscopy products in Canada for total consideration, excluding acquisition-related costs, of $4,044. Vantage was our exclusive distributor of Medivators capital equipment (e.g., automated endoscope reprocessors) and related consumables and accessories in Canada, and is included in our Endoscopy segment.
On August 1, 2016, we acquired all of the issued and outstanding stock of Accutron, a Phoenix-based company for total consideration, excluding acquisition-related costs, of $53,049. The Accutron business designs, manufactures and sells nitrous oxide conscious sedation equipment and single use nasal masks for use in dental procedures, and is included in our Healthcare Disposables segment.
(dollar amounts in thousands except share and per share data or as otherwise specified) 23
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Fiscal 2016
On March 1, 2016, we acquired certain net assets of North American Science Associates, Inc.’s Sterility Assurance Monitoring Products Division ("NAMSA") for total consideration, excluding acquisition-related costs, of $13,424. The NAMSA business manufactures a broad suite of high-quality biological and chemical indicators which are used to accurately monitor the effectiveness of sterilization processes primarily for manufacturers of medical device, life science and other products, and is included in our Healthcare Disposables segment.
On September 14, 2015, we acquired all of the issued and outstanding stock of Medical Innovations Group Holdings Ltd. ("MI"), a company providing specialized endoscopy medical devices and products primarily in the United Kingdom for total consideration, excluding acquisition-related costs, of $79,597. The MI business includes proprietary short-term and long-term endoscope transport and storage systems, a comprehensive range of endoscopic consumable accessories, OEM mobile medical carts, as well as specialized products for patient warming and patient transfer, and is included in our Endoscopy segment.
See Note 3 to our consolidated financial statements in Part II, Item 8 of this report.
Results of Operations
The following table gives information as to the percentages of net sales represented by selected items reflected in our consolidated statements of income.
Year Ended July 31 | Percentage Change | ||||||||||||||||||||||
Statement of Income Data: | 2017 | 2016 | 2015 | 2017 / 2016 | 2016 / 2015 | ||||||||||||||||||
Net sales | $ | 770,157 | 100.0 | % | $ | 664,755 | 100.0 | % | $ | 565,004 | 100.0 | % | 15.9 | % | 17.7 | % | |||||||
Cost of sales | 402,997 | 52.3 | % | 355,569 | 53.5 | % | 311,537 | 55.1 | % | 13.3 | % | 14.1 | % | ||||||||||
Gross profit | 367,160 | 47.7 | % | 309,186 | 46.5 | % | 253,467 | 44.9 | % | 18.8 | % | 22.0 | % | ||||||||||
Selling | 116,113 | 15.1 | % | 99,062 | 14.9 | % | 80,787 | 14.3 | % | 17.2 | % | 22.6 | % | ||||||||||
General and administrative | 122,270 | 15.9 | % | 97,463 | 14.7 | % | 77,897 | 13.8 | % | 25.5 | % | 25.1 | % | ||||||||||
Research and development | 18,367 | 2.4 | % | 15,410 | 2.3 | % | 14,022 | 2.5 | % | 19.2 | % | 9.9 | % | ||||||||||
256,750 | 33.4 | % | 211,935 | 31.9 | % | 172,706 | 30.6 | % | 21.1 | % | 22.7 | % | |||||||||||
Operating income | 110,410 | 14.3 | % | 97,251 | 14.6 | % | 80,761 | 14.3 | % | 13.5 | % | 20.4 | % | ||||||||||
Interest expense, net | 4,303 | 0.5 | % | 3,320 | 0.5 | % | 2,364 | 0.4 | % | 29.6 | % | 40.4 | % | ||||||||||
Other income | (126 | ) | — | % | — | — | % | — | — | % | — | % | — | % | |||||||||
Loss on sale of business | — | — | % | — | — | % | 2,206 | 0.4 | % | — | % | (100.0 | )% | ||||||||||
Income before income taxes | 106,233 | 13.8 | % | 93,931 | 14.1 | % | 76,191 | 13.5 | % | 13.1 | % | 23.3 | % | ||||||||||
Income taxes | 34,855 | 4.5 | % | 33,978 | 5.1 | % | 28,238 | 5.0 | % | 2.6 | % | 20.3 | % | ||||||||||
Net income | $ | 71,378 | 9.3 | % | $ | 59,953 | 9.0 | % | $ | 47,953 | 8.5 | % | 19.1 | % | 25.0 | % | |||||||
(dollar amounts in thousands except share and per share data or as otherwise specified) 24
Cantel Medical Corp. 2017 Annual Report on Form 10-K
The following table gives information as to the net sales by reporting segment and geography, as well as the related percentage of such sales to the total net sales.
Year Ended July 31, | ||||||||||||||||||||
Net Sales by Segment | 2017 | 2016 | 2015 | |||||||||||||||||
Endoscopy | $ | 398,773 | 51.8 | % | $ | 341,752 | 51.4 | % | $ | 248,654 | 44.0 | % | ||||||||
Water Purification and Filtration | 196,446 | 25.5 | % | 177,669 | 26.7 | % | 173,834 | 30.8 | % | |||||||||||
Healthcare Disposables | 144,457 | 18.7 | % | 112,584 | 17.0 | % | 106,920 | 18.9 | % | |||||||||||
Dialysis | 30,481 | 4.0 | % | 32,750 | 4.9 | % | 31,240 | 5.5 | % | |||||||||||
Other | — | — | — | — | 4,356 | 0.8 | % | |||||||||||||
Total net sales | $ | 770,157 | 100.0 | % | $ | 664,755 | 100.0 | % | $ | 565,004 | 100.0 | % | ||||||||
Net Sales by Geography | ||||||||||||||||||||
United States | $ | 599,657 | 77.9 | % | $ | 515,055 | 77.4 | % | $ | 447,848 | 79.3 | % | ||||||||
International | 170,500 | 22.1 | % | 149,700 | 22.6 | % | 117,156 | 20.7 | % | |||||||||||
Total net sales | $ | 770,157 | 100.0 | % | $ | 664,755 | 100.0 | % | $ | 565,004 | 100.0 | % | ||||||||
The following table gives information as to the amount of operating income, as well as operating income as a percentage of net sales, for each of our reporting segments.
Year Ended July 31, | ||||||||||||||||||||
Operating Income (including percentage of net sales): | 2017 | 2016 | 2015 | |||||||||||||||||
Endoscopy | $ | 73,440 | 18.4 | % | $ | 61,021 | 17.9 | % | $ | 40,863 | 16.4 | % | ||||||||
Water Purification and Filtration | 33,159 | 16.9 | % | 30,620 | 17.2 | % | 30,606 | 17.6 | % | |||||||||||
Healthcare Disposables | 28,000 | 19.4 | % | 24,486 | 21.7 | % | 19,904 | 18.6 | % | |||||||||||
Dialysis | 8,154 | 26.8 | % | 7,907 | 24.1 | % | 6,749 | 21.6 | % | |||||||||||
Other | — | — | % | — | — | % | 1,118 | 25.7 | % | |||||||||||
Operating income by segment | 142,753 | 18.5 | % | 124,034 | 18.7 | % | 99,240 | 17.6 | % | |||||||||||
General corporate expenses | 32,343 | 4.2 | % | 26,783 | 3.9 | % | 18,479 | 3.3 | % | |||||||||||
Income from operations | $ | 110,410 | 14.3 | % | $ | 97,251 | 14.6 | % | $ | 80,761 | 14.3 | % | ||||||||
Fiscal 2017 compared with Fiscal 2016
Net Sales
Total net sales increased by $105,402 or 15.9%, to $770,157 in fiscal 2017 from $664,755 in fiscal 2016. The 15.9% increase in net sales for fiscal 2017 includes an increase of 11.0% in organic sales, an increase of 5.9% in sales due to acquisitions, partially offset by a decrease of 1.0% due to foreign currency translation.
International net sales increased by $20,800, or 13.9%, to $170,500 in fiscal 2017 from $149,700 in fiscal 2016. The 13.9% increase in net sales consist of an increase of 9.2% in organic sales and an increase of 8.9% in net sales due to acquisitions, partially offset by a decrease of 4.2% due to foreign currency translation.
Endoscopy. Net sales of endoscopy products and services increased by $57,021, or 16.7%, in fiscal 2017 compared with fiscal 2016. The 16.7% increase in net sales consist of an increase of 15.0% in organic net sales and an increase of 3.5% in net sales due to acquisitions, partially offset by a decrease of 1.8% due to foreign currency translation. The increase in organic net sales was primarily due to volume increases in the United States and internationally for endoscopy procedure products, storage cabinets and mobile medical carts, and disinfectants and service due to the increased installed base of our endoscope reprocessing equipment. We expect net sales of disinfectants, service, filters and equipment accessories, most of which carry higher margins, to continue to benefit as the installed base of endoscope reprocessing equipment increases.
Water Purification and Filtration. Net sales of water purification and filtration products and services increased by $18,777, or 10.6%, in fiscal 2017 compared with fiscal 2016. The 10.6% increase in net sales was primarily due to an increase in demand for our water purification equipment.
(dollar amounts in thousands except share and per share data or as otherwise specified) 25
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Healthcare Disposables. Net sales of healthcare disposables products increased by $31,873, or 28.3%, in fiscal 2017 when compared with fiscal 2016. The 28.3% increase in net sales consists of an increase of 23.9% in net sales due to acquisitions and an increase of 4.4% in organic net sales. The increase in organic net sales was driven by our higher margin products such as sterility assurance and waterline disinfection products, as well as our branded products.
Dialysis. Net sales of dialysis products and services decreased by $2,269, or 6.9%, in fiscal 2017 when compared with fiscal 2016, principally due to the decrease in demand for our sterilant product and reprocessing equipment, both internationally and in the United States, due to the continued market shift from reusable to single-use dialyzers, as further described below.
Net sales in our Dialysis segment in recent years have been adversely impacted by the decrease in demand for our sterilants and reprocessing equipment principally due to the shift from reusable to single-use dialyzers as a result of the declining cost of single-use dialyzers and the ease of using a dialyzer one time as well as the commitment of Fresenius, the largest dialysis provider chain in the United States and a manufacturer of single-use dialyzers, to convert dialysis clinics performing reuse to single-use facilities.
We expect the downward trend in reuse dialyzers in the United States to continue during fiscal 2018 and thereafter as the most significant manufacturers of reuse dialyzers have indicated that they will cease manufacturing of such products in the near term. A substantial decrease in the market for reprocessing products is likely to result in a significant loss of net sales and a lower level of profitability and operating cash flow in this segment in the future as well as potential future impairments of long-lived assets. Such reduction would also have a material effect on our consolidated results of operations. See "Risk Factors” in Part I, Item 1A in this report.
Gross Profit
Gross profit increased by $57,974 or 18.8%, to $367,160 in fiscal 2017 from $309,186 in fiscal 2016. Gross profit as a percentage of net sales in fiscal 2017 and 2016 was 47.7% and 46.5%, respectively. Excluding the impact of acquisition related items, gross profit as a percentage of net sales in fiscal 2017 and 2016 was 47.7% and 46.6%, respectively.
The higher gross profit as a percentage of net sales in fiscal 2017 and 2016 was primarily attributable to (i) more favorable sales mix due to increases in sales volume of certain higher margin products, such as our endoscopy procedure products and disinfectants in our Endoscopy segment and sterility assurance and waterline disinfection products in our Healthcare Disposables segment, (ii) lower manufacturing costs primarily due to cost control initiatives, (iii) increased plant productivity due to increased sales volume and (iv) the favorable impact of the suspension of the U.S. medical device excise tax, partially offset by an increase in net sales of lower margin capital equipment primarily in our Water Purification and Filtration segment and increased warranty charges primarily relating to our water purification equipment.
In December 2015, the Consolidated Appropriations Act of 2016 was signed into law and included a two-year moratorium effective January 1, 2016 on the medical device excise tax, which was a tax on medical device manufacturers in the form of a 2.3% excise tax on all U.S. medical device sales. A significant portion of our net sales are considered U.S. medical device sales and therefore our gross profit percentage will continue to be favorably impacted until the two-year moratorium expires. However, we are investing a significant portion of the savings from this moratorium into sales and marketing, product development and human resources initiatives.
Operating Expenses
Selling expenses increased by $17,051, or 17.2%, to $116,113 in fiscal 2017 from $99,062 in fiscal 2016. In fiscal 2017, selling expenses increased primarily due to (i) higher commission expense relating to increased net sales in our Endoscopy segment, (ii) increased sales and marketing initiatives to expand into new markets, including international markets, and to gain or maintain market share by hiring and training additional sales and marketing personnel, (iii) the inclusion of selling and marketing expenses of acquisitions, and (iv) an increase in salary and incentive compensation costs. Selling expenses as a percentage of net sales were 15.1% and 14.9% in fiscal 2017 and 2016, respectively.
General and administrative expenses increased by $24,807, or 25.5%, to $122,270 in fiscal 2017 from $97,463 in fiscal 2016. General and administrative expenses increased primarily due to (i) increases in annual salaries and incentive compensation, including stock-based compensation, (ii) the addition of internal and external resources to address various growth initiatives and compliance requirements, (iii) an increase in amortization expense related to recent acquisitions and (iv) severance and other restructuring costs, partially offset by lower acquisition related items such as transaction and integration charges and fair value adjustments. General and administrative expenses as a percentage of net sales were 15.9% and 14.7% in fiscal 2017 and 2016, respectively.
(dollar amounts in thousands except share and per share data or as otherwise specified) 26
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Research and development expenses (which include continuing engineering costs) increased by $2,957, or 19.2%, to $18,367 in fiscal 2017 from $15,410 in fiscal 2016. The increase was primarily due to additional product development initiatives primarily in our Endoscopy segment, including projects relating to recent acquisitions. Research and development expenses as a percentage of net sales were 2.4% and 2.3%% in fiscal 2017 and 2016, respectively.
Operating Income
Endoscopy. The Endoscopy segment’s operating income increased by $12,419, or 20.4%, in fiscal 2017 compared with fiscal 2016, primarily due to favorable product mix and increased sales volume in the United States and internationally for our endoscopy products and services, as further explained above and the impact of our recent acquisitions, partially offset by increased compensation-related costs and investments in our sales team and other selling initiatives.
Water Purification and Filtration. The Water Purification and Filtration segment’s operating income increased by $2,539, or 8.3%, in fiscal 2017 compared with fiscal 2016, primarily as a result of higher sales, partially offset by (i) increased compensation-related costs, (ii) higher commission expenses, and (iii) lower margins due to increased bad debt and warranty expenses.
Healthcare Disposables. The Healthcare Disposables segment’s operating income increased by $3,514, or 14.4%, in fiscal 2017 compared with fiscal 2016, primarily due to the sales impact of our recent acquisition and favorable product mix of both core and acquired products. This was partially offset by increased salary and incentive compensation costs, the hiring of additional sales personnel and commission expense associated with our recent acquisition.
Dialysis. The Dialysis segment’s operating income increased by $247, or 3.1% in fiscal 2017 compared with fiscal 2016, primarily due to cost control initiatives, partially offset by lower net sales, as further explained above.
General Corporate Expenses
General corporate expenses relate to unallocated corporate costs primarily related to executive management personnel as well as costs associated with certain facets of our acquisition program and being a publicly traded company. Such expenses increased by $5,560, or 20.8% in fiscal 2017 compared with fiscal 2016, primarily due to (i) various restructuring and business optimization activities, (ii) the addition of internal and external resources to address various growth initiatives and compliance requirements and (iii) increases in compensation-related costs, including stock-based compensation expense, partially offset by a decrease in costs associated with the retirement of our Chief Executive Officer in fiscal 2016.
Interest
Interest expense, net increased by $983 to $4,303 in fiscal 2017 from $3,320 in fiscal 2016, as a result of an increase in the average outstanding borrowings due to the funding of acquisitions.
Income Taxes
The consolidated effective tax rate decreased by 3.4% to 32.8% in fiscal 2017 from 36.2% in fiscal 2016, due to the favorable impact recording the excess tax benefits relating to stock awards as a result of the adoption of ASU 2016-09 on August 1, 2016 and the favorable impact in the current year from the retroactive application for the research and experimentation tax credit for fiscal 2016, 2015, and 2014 in Minnesota where our principal research & development activities occur, as further described within Non-GAAP Financial Measures elsewhere in this MD&A. Additionally, the current year consolidated effective tax rate was negatively impacted by increased state tax expense due to expanded presence within various U.S. tax jurisdictions.
Fiscal 2016 compared with Fiscal 2015
Net Sales
Total net sales increased by $99,751 or 17.7%, to $664,755 in fiscal 2016 from $565,004 in fiscal 2015. The 17.7% increase in net sales for fiscal 2016 includes an increase of 12.7% in organic sales, an increase of 5.5% in sales due to acquisitions, partially offset by the divestiture of our specialty packaging business and a decrease of 0.5% due to foreign currency translation.
International net sales increased by $32,544, or 27.8%, to $149,700 in fiscal 2016 from $117,156 in fiscal 2015. The 27.8% increase in net sales consist of an increase of 12.5% in organic sales and an increase of 17.5% in net sales due to acquisitions, partially offset by a decrease of 2.2% due to foreign currency translation.
(dollar amounts in thousands except share and per share data or as otherwise specified) 27
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Endoscopy. Net sales of endoscopy products and services increased by $93,098, or 37.4%, in fiscal 2016 compared with fiscal 2015. The 37.4% increase in net sales consist of an increase of 28.0% in organic net sales and an increase of 10.2% in net sales due to acquisitions, partially offset by a decrease of 0.8% due to foreign currency translation. The increase in organic net sales was primarily due to increases in demand in the United States and internationally for our (i) endoscopy procedure products (disposable infection control products used in GI endoscopy procedures) due to sales and marketing efforts, (ii) endoscope reprocessing equipment due to our sales and marketing programs and (iii) disinfectants and service due to the increase in the installed base of endoscope reprocessing equipment. These increases were partially offset by overall lower selling prices principally related to endoscopy reprocessing equipment and endoscopy procedure products as a result of our strategic growth plan and increased competition.
Water Purification and Filtration. Net sales of water purification and filtration products and services increased by $3,835, or 2.2%, in fiscal 2016 compared with fiscal 2015. The 2.2% increase in net sales consist of an increase of 1.0% in organic net sales and an increase of 1.6% in net sales due to acquisitions, partially offset by a decrease of 0.4% due to foreign currency translation. The increase in organic net sales was primarily due to an increase in demand for our sterilants products, service and water purification equipment used for commercial and industrial (large capital) applications.
Healthcare Disposables. Net sales of healthcare disposables products increased by $5,664, or 5.3%, in fiscal 2016 when compared with fiscal 2015. The 5.3% increase in net sales consist of increase of 0.3% in organic net sales and an increase of 5.0% in net sales due to acquisitions. Organic net sales for fiscal 2016 were similar to fiscal 2015 as the increase in sales of sterility assurance and waterline disinfection products in the fiscal 2016 were offset by the elevated demand during the first half of fiscal 2015 for our face masks and certain sterilization products as a result of customers buying products in advance of certain sales price increases and customer response to the Ebola virus.
Dialysis. Net sales of dialysis products and services increased by $1,510, or 4.8% in fiscal 2016 when compared with fiscal 2015, principally due to an increase in demand for our concentrate product by a single customer, partially offset by a decrease in demand for our sterilant products and RENATRON® reprocessing equipment due to the market shift from reusable to single-use dialyzers.
Gross Profit
Gross profit increased by $55,719 or 22.0%, to $309,186 in fiscal 2016 from $253,467 in fiscal 2015. Gross profit as a percentage of net sales in fiscal 2016 and 2015 was 46.5% and 44.9%, respectively. Excluding the impact of acquisition accounting charges, gross profit as a percentage of net sales in fiscal 2016 and 2015 was 46.7% and 45.2%, respectively.
The higher gross profit as a percentage of net sales in fiscal 2016 and 2015 was primarily attributable to (i) more favorable sales mix due to increases in sales volume of certain products that carry higher gross margin percentages such as our endoscopy procedure products and disinfectants in our Endoscopy segment, sterility assurance and waterline disinfection products in our Healthcare Disposables segment and sterilants and filters in our Water Purification and Filtration segment, (ii) the inclusion of higher margin sales in our Endoscopy and Healthcare Disposables segments as a result of the MI and NAMSA acquisitions, respectively, (iii) lower manufacturing costs and (iv) decrease of $2,334 in medical device excise tax due to the recent moratorium, partially offset by an increase in net sales of lower margin capital equipment primarily in our Endoscopy segment and higher charges for warranty primarily relating to our water purification equipment.
Operating Expenses
Selling expenses increased by $18,275, or 22.6%, to $99,062 in fiscal 2016 from $80,787 in fiscal 2015. In fiscal 2016, selling expenses increased primarily due to (i) higher commission expense relating to increased net sales in our Endoscopy segment, (ii) increased sales and marketing initiatives to expand into new markets, including international markets, and to gain or maintain market share by hiring and training additional sales and marketing personnel and increasing travel budgets in our Endoscopy, Water Purification and Filtration and Healthcare Disposables segments, (iii) the inclusion of selling and marketing expenses of acquisitions, and (iv) increases in annual salaries. These increases were partially offset by a decrease of $884 in selling expense for fiscal 2016 relating to our specialty packaging business divested in April 2015. Selling expenses as a percentage of net sales were 14.9% and 14.3% in fiscal 2016 and 2015, respectively.
General and administrative expenses increased by $19,566, or 25.1%, to $97,463 in fiscal 2016 from $77,897 in fiscal 2015. General and administrative expenses increased primarily due to (i) the inclusion of general and administrative expenses of our acquisitions, (ii) the impact of atypical items relating to acquisitions and costs associated with the retirement of our Chief Executive Officer, partially offset by the prior year impairment of an acquired license, as further described below and within Non-GAAP Financial Measures elsewhere in this MD&A, and (iii) increases in annual salaries and incentive compensation including
(dollar amounts in thousands except share and per share data or as otherwise specified) 28
Cantel Medical Corp. 2017 Annual Report on Form 10-K
stock-based compensation. General and administrative expenses as a percentage of net sales were 14.7% and 13.8% in fiscal 2016 and 2015, respectively.
Research and development expenses (which include continuing engineering costs) increased by $1,388, or 9.9%, to $15,410 in fiscal 2016 from $14,022 in fiscal 2015. The increase was primarily due to additional product development initiatives primarily in our Endoscopy segment, including the inclusion of projects relating to acquisitions. Research and development expenses as a percentage of net sales were 2.3% and 2.5% in fiscal 2016 and 2015, respectively.
Operating Income
Endoscopy. The Endoscopy segment’s operating income increased by $20,158, or 49.3%, in fiscal 2016 compared with fiscal 2015, primarily due to increases in sales in the United States and internationally for our endoscopy products and services, as further explained above, and to a much lesser extent, the inclusion of operating income from acquisitions. These items were partially offset by (i) higher commission expense and other incentive compensation, (ii) increased investment in our sales team and other selling initiatives, (iii) an increase in annual salaries and (iii) the net unfavorable impact from certain items, as further described below and within Non-GAAP Financial Measures elsewhere in this MD&A.
Water Purification and Filtration. The Water Purification and Filtration segment’s operating income increased slightly in fiscal 2016 compared with fiscal 2015, primarily as a result of higher sales, partially offset by increases in annual salaries, the hiring of additional sales personnel, increased travel budgets and higher charges for warranty related to our water purification equipment.
Healthcare Disposables. The Healthcare Disposables segment’s operating income increased by $4,582, or 23.0%, in fiscal 2016 compared with fiscal 2015, primarily due to (i) the inclusion of sales relating to acquisitions, (ii) less amortization expense and (iii) lower manufacturing costs, partially offset by increases to annual salaries and the hiring of additional sales personnel.
Dialysis. The Dialysis segment’s operating income increased by $1,158, or 17.2% in fiscal 2016 compared with fiscal 2015, primarily due to an increase in sales for our low margin concentrate product to a single customer and successful cost control initiatives, partially offset by a decrease in demand for our higher margin sterilant products and RENATRON® reprocessing equipment.
General Corporate Expenses
General corporate expenses relate to unallocated corporate costs primarily related to executive management personnel as well as costs associated with certain facets of our acquisition program and being a publicly traded company. Such expenses increased by $8,304, or 44.9% in fiscal 2016 compared with fiscal 2015, primarily due to (i) $3,487 of costs recorded in the second half of fiscal 2016 associated with the retirement of our Chief Executive Officer, (ii) the addition of internal and external resources to address various growth initiatives and compliance requirements, (iii) increases in costs associated with our acquisition program and (iv) increases in annual salaries and incentive compensation, including stock-based compensation expense.
Interest
Interest expense, net increased by $956 to $3,320 in fiscal 2016 from $2,364 in fiscal 2015, as a result of an increase in the average outstanding borrowings due to the funding of the MI and NAMSA acquisitions in September 2015 and March 2016, respectively.
Income Taxes
The consolidated effective tax rate decreased by 0.9% to 36.2% in fiscal 2016, from 37.1% in fiscal 2015, due to the negative impact of the divesture of our specialty packaging business in fiscal 2015 and the favorable impact in fiscal 2016 from the enactment of tax legislation in the United States and internationally, partially offset by higher non-deductible acquisition related items in fiscal 2016, as further described within Non-GAAP Financial Measures elsewhere in this MD&A. Additionally, the fiscal 2016 consolidated effective tax rate was favorably impacted by improved operating results of our international operations, which are located in lower tax rate jurisdictions.
(dollar amounts in thousands except share and per share data or as otherwise specified) 29
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Non-GAAP Financial Measures
In evaluating our operating performance, we supplement the reporting of our financial information determined under generally accepted accounting principles in the United States (“GAAP”) with certain non-GAAP financial measures including (i) non-GAAP net income; (ii) non-GAAP earnings per diluted share ("EPS"); (iii) earnings before interest, taxes, depreciation, amortization, loss on disposal of fixed assets, and stock-based compensation expense (“EBITDAS”); (iv) adjusted EBITDAS; (v) net debt; and (vi) organic sales. These non-GAAP financial measures are indicators of the Company's performance that are not required by, or presented in accordance with, GAAP. They are presented with the intent of providing greater transparency to financial information used by us in our financial analysis and operational decision-making. We believe that these non-GAAP measures provide meaningful information to assist investors, stockholders and other readers of our consolidated financial statements in making comparisons to our historical operating results and analyzing the underlying performance of our results of operations. These non-GAAP financial measures are not intended to be, and should not be, considered separately from, or as an alternative to, the most directly comparable GAAP financial measures.
To measure earnings performance on a consistent and comparable basis, we exclude certain items that affect comparability of operating results and the trend of earnings. These adjustments are irregular in timing, may not be indicative of our past and future performance and are therefore excluded to allow investors to better understand underlying operating trends. The following are examples of the types of adjustments that are excluded: (i) amortization of purchased intangible assets; (ii) acquisition related items; (iii) business optimization and restructuring-related charges; (iv) certain significant and discrete tax matters; and (v) other significant items management deems irregular or non-operating in nature.
Amortization expense of purchased intangible assets is a non-cash expense related to intangibles that were primarily the result of business acquisitions. Our history of acquiring businesses has resulted in significant increases in amortization of intangible assets that reduced the Company’s net income. The removal of amortization from our overall operating performance helps in assessing our cash generated from operations including our return on invested capital, which we believe is an important analysis for measuring our ability to generate cash and invest in our continued growth.
Acquisition related items consist of (i) fair value adjustments to contingent consideration and other contingent liabilities resulting from acquisitions, (ii) due diligence, integration, legal fees and other transaction costs associated with our acquisition program and (iii) acquisition accounting charges for the amortization of the initial fair value adjustments of acquired inventory and deferred revenue. The adjustments of contingent consideration and other contingent liabilities are periodic adjustments to record such amounts at fair value at each balance sheet date. Given the subjective nature of the assumptions used in the determination of fair value calculations, fair value adjustments may potentially cause significant earnings volatility that are not representative of our operating results. Similarly, due diligence, integration, legal and other acquisition costs associated with our acquisition program, including acquisition accounting charges relating to recording acquired inventory and deferred revenue at fair market value, can be significant and also adversely impact our effective tax rate as certain costs are often not tax-deductible. Since these acquisition related items are irregular and often mask underlying operating performance, we excluded these amounts for purposes of calculating these non-GAAP financial measures to facilitate an evaluation of our current operating performance and a comparison to past operating performance.
As a result of the adoption of a new accounting standard on August 1, 2016, as further described in Note 2, Note 11 and Note 15 of the consolidated financial statements in Part II, Item 8 of this report, we no longer record excess tax benefits as an adjustment to additional paid-in capital, but record such excess tax benefits on a prospective basis as a reduction of income tax expense, which amounted to $2,241 in fiscal 2017. The magnitude of the impact of excess tax benefits generated in the future, which may be favorable or unfavorable, are dependent upon our future grants of equity awards, our future share price on the date awards vest in relation to the fair value of awards on grant date and the exercise behavior of our stock option holders. Since these favorable tax benefits are largely unrelated to our results and unrepresentative of our normal effective tax rate, we excluded its impact on net income and diluted EPS for fiscal 2017 to arrive at our non-GAAP financial measures.
In fiscal 2016, we announced the retirement plans of our Chief Executive Officer and recorded the majority of the costs associated with his retirement in our consolidated financial statements. Since these costs are irregular and mask our underlying operating performance, we made an adjustment to our net income and diluted EPS for fiscal 2017 and 2016 to exclude such costs to arrive at our non-GAAP financial measures.
Tax legislation was enacted in the United States and internationally that enabled us to record favorable tax benefits in the second quarter of fiscal 2016 relating to the 2015 calendar year. Since these favorable tax benefits were largely unrelated to fiscal 2016, we excluded its impact on net income and diluted EPS for fiscal 2016 for purposes of calculating these non-GAAP financial measures to facilitate an evaluation of our current performance and a comparison to past performance.
(dollar amounts in thousands except share and per share data or as otherwise specified) 30
Cantel Medical Corp. 2017 Annual Report on Form 10-K
On April 7, 2015, we completed the sale of our specialty packaging business that resulted in a $2,206 loss, or $0.04 in diluted EPS. This was recorded as a loss on sale of business in our consolidated statements of income. Since the divestiture of a business is infrequent and non-operating in nature and the loss on sale masks our underlying operating performance, we excluded the loss on sale of business for purposes of calculating these non-GAAP financial measures for fiscal 2015.
In Fiscal 2015, we recorded an impairment loss of $1,287 associated with an acquired license, which was recorded in general and administrative expenses in the consolidated statements of income. Since the acquisition of the license and subsequent impairment were outside our standard endoscopy business operations, we excluded the impairment of the acquired license for purposes of calculating these non-GAAP financial measures to facilitate an evaluation of our current operating performance and a comparison to past operating performance.
Fiscal 2017
We made adjustments to net income and diluted EPS to exclude (i) amortization expense of purchased intangible assets, (ii) acquisition related items, (iii) costs associated with the retirement of our former Chief Executive Officer and (iv) other business optimization and restructuring-related charges to arrive at our non-GAAP financial measures, non-GAAP net income and non-GAAP diluted EPS.
Fiscal 2016
We made adjustments to net income and diluted EPS to exclude (i) amortization expense of purchased intangible assets, (ii) acquisition related items, (iii) costs associated with the retirement of our former Chief Executive Officer, (iv) other business optimization and restructuring-related charges and (v) the favorable impact of tax legislation to arrive at our non-GAAP financial measures, non-GAAP net income and non-GAAP diluted EPS.
Fiscal 2015
We made adjustments to net income and diluted EPS to exclude (i) amortization expense of purchased intangible assets, (ii) acquisition related items, (iii) the loss on sale of our specialty packaging business and (iv) the impairment of an acquired license to arrive at our non-GAAP financial measures, non-GAAP net income and non-GAAP diluted EPS.
The reconciliations of net income and diluted EPS to non-GAAP net income and non-GAAP diluted EPS were calculated as follows:
July 31, | |||||||||||||||||||||||
2017 | 2016 | 2015 | |||||||||||||||||||||
Net income/Diluted EPS, as reported | $ | 71,378 | $ | 1.71 | $ | 59,953 | $ | 1.44 | $ | 47,953 | $ | 1.15 | |||||||||||
Intangible amortization, net of tax(1) | 12,800 | 0.30 | 9,283 | 0.22 | 8,778 | 0.21 | |||||||||||||||||
Acquisition related items, net of tax(2) | 1,533 | 0.04 | 2,290 | 0.06 | 747 | 0.02 | |||||||||||||||||
CEO retirement costs, net of tax(1) | 1,213 | 0.03 | 2,212 | 0.05 | — | — | |||||||||||||||||
Restructuring related charges, net of tax(1) | 2,057 | 0.05 | — | — | — | — | |||||||||||||||||
Excess tax benefit(3) | (2,241 | ) | (0.05 | ) | — | — | — | — | |||||||||||||||
Loss on sale of business, net of tax | — | — | — | — | 1,746 | 0.04 | |||||||||||||||||
Impairment of acquired license, net of tax(1) | — | — | — | — | 815 | 0.02 | |||||||||||||||||
Tax legislative changes(3) | — | — | (800 | ) | (0.02 | ) | — | — | |||||||||||||||
Non-GAAP net income/Non-GAAP diluted EPS | $ | 86,740 | $ | 2.08 | $ | 72,938 | $ | 1.75 | $ | 60,039 | $ | 1.44 | |||||||||||
________________________________________________
(1) | Amounts were recorded in general and administrative expenses. |
(2) | In fiscal 2017, pre-tax acquisition related items of $353 were recorded in cost of sales and $2,094 were recorded in general and administrative expenses. In fiscal 2016, pre-tax acquisition related items of $959 were recorded in cost of sales and $2,254 were recorded in general and administrative expenses. In fiscal 2015, pre-tax acquisition related items of $1,981 were recorded in cost of sales and a $402 favorable pre-tax benefit was recorded in general and administrative expenses. |
(3) | Amounts are recorded in income taxes. |
(dollar amounts in thousands except share and per share data or as otherwise specified) 31
Cantel Medical Corp. 2017 Annual Report on Form 10-K
We believe EBITDAS is an important valuation measurement for management and investors given the increasing effect that non-cash charges, such as stock-based compensation, amortization related to acquisitions and depreciation of capital equipment have on the Company’s net income. In particular, acquisitions have historically resulted in significant increases in amortization of purchased intangible assets that reduce the Company’s net income. Additionally, we regard EBITDAS as a useful measure of operating performance and cash flow before the effect of interest expense and is a complement to operating income, net income and other GAAP financial performance measures.
We define adjusted EBITDAS as EBITDAS excluding the same non-GAAP adjustments to net income discussed previously in this document. We use adjusted EBITDAS when evaluating the operating performance of the Company because we believe the exclusion of such adjustments, of which a significant portion are non-cash items, is necessary to provide the most accurate measure of on-going core operating results and to evaluate comparative results period over period.
The reconciliations of net income to EBITDAS and adjusted EBITDAS were calculated as follows:
July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Net income, as reported | $ | 71,378 | $ | 59,953 | $ | 47,953 | |||||
Interest expense, net | 4,303 | 3,320 | 2,364 | ||||||||
Income taxes | 34,855 | 33,978 | 28,238 | ||||||||
Depreciation | 15,045 | 11,989 | 10,692 | ||||||||
Amortization | 18,407 | 13,095 | 13,265 | ||||||||
Loss on disposal of fixed assets | 966 | 553 | 360 | ||||||||
Stock-based compensation expense | 8,844 | 8,361 | 5,867 | ||||||||
EBITDAS | 153,798 | 131,249 | 108,739 | ||||||||
Acquisition related items | 2,447 | 3,213 | 1,579 | ||||||||
CEO retirement costs | 1,937 | 3,487 | — | ||||||||
Restructuring related charges | 3,284 | — | — | ||||||||
Loss on sale of business | — | — | 2,206 | ||||||||
Impairment of acquired license | — | — | 1,287 | ||||||||
Adjusted EBITDAS | $ | 161,466 | $ | 137,949 | $ | 113,811 | |||||
We define net debt as long-term debt less cash and cash equivalents. Each of the components of net debt appears on our consolidated balance sheets. We believe that the presentation of net debt provides useful information to investors because we review net debt as part of our management of our overall liquidity, financial flexibility, capital structure and leverage.
July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Long-term debt | $ | 126,000 | $ | 116,000 | $ | 78,500 | |||||
Less cash and cash equivalents | (36,584 | ) | (28,367 | ) | (31,720 | ) | |||||
Net debt | $ | 89,416 | $ | 87,633 | $ | 46,780 | |||||
We define organic sales as net sales less (i) the impact of foreign currency translation and (ii) net sales related to acquired businesses during the first twelve months of ownership and (iii) divestures during the periods being compared. We believe that reporting organic sales provides useful information to investors by helping identify underlying growth trends in our business and facilitating easier comparisons of our revenue performance with prior periods. We exclude the effect of foreign currency translation from organic sales because foreign currency translation is not under management’s control, is subject to volatility and can obscure underlying business trends. We exclude the effect of acquisitions because the nature, size, and number of acquisitions can vary dramatically from period to period and can obscure underlying business trends and make comparisons of financial performance difficult. The reconciliation of net sales to organic sales can be found elsewhere in this MD&A in “Fiscal 2017 compared with Fiscal 2016.”
(dollar amounts in thousands except share and per share data or as otherwise specified) 32
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Liquidity and Capital Resources
We assess our liquidity in terms of our ability to generate cash to fund operating, investing and financing activities. Significant factors affecting the management of liquidity are cash flows generated from operating activities, capital expenditures, acquisitions of businesses and cash dividends. Cash provided by operating activities continues to be a primary source of funds. As necessary, we supplement operating cash flow with borrowings from our revolving credit facility to fund our business activities.
Cash Flows
Net Cash Provided by Operating Activities. Net cash provided by operating activities increased by $27,925, or 34.8%, to $108,193 for fiscal 2017 from $80,268 for fiscal 2016, primarily due to the increase in net income (after adjusting for non-cash items) and decreases in inventory levels (net of acquisitions), partially offset by decreases in accounts payable (due to timing of payments). Net cash provided by operating activities increased by $21,198, or 35.9%, to $80,268 for fiscal 2016 from $59,070 for fiscal 2015, primarily due to the increase in net income (after adjusting for non-cash items) and increases in accounts payable and other current liabilities (due to the timing of payments), partially offset by increases in inventories (due to planned strategic increases in stock levels of certain products primarily in our Endoscopy segment) and accounts receivables (due to strong sales of endoscopy products and services).
Net Cash Used in Investing Activities. Net cash used in investing activities decreased by $15,920, or 14.1%, to $97,062 for fiscal 2017 from $112,982 for fiscal 2016, primarily due to a decrease in cash consideration paid for acquisitions, partially offset by an increase in capital expenditures. Net cash used in investing activities increased by $60,688, or 116.1%, to $112,982 for fiscal 2016 from $52,294 for fiscal 2015, primarily due to an increase in cash consideration paid for acquisitions. During fiscal 2017, 2016 and 2015 net cash used in investing activities included capital expenditures of $27,065, $18,889 and $12,760, respectively, which included expenditures for building improvements and purchases of manufacturing and computer equipment.
Net Cash Provided by (used in) Financing Activities. Net cash provided by (used in) financing activities increased by $32,693, or 109.2%, to $2,751 of cash used for fiscal 2017 from $29,942 provided by for fiscal 2016. Net cash provided by financing activities increased by $36,047, or 590.5%, to $29,942 of cash provided for fiscal 2016 from $6,105 of cash used for fiscal 2015. The changes in net cash provided by (used in) financing activities were primarily due to borrowings under our revolving credit facility to fund acquisitions, offset by repayments under our credit facility.
Dividends
For a discussion of our dividend policy, see the information set forth under the heading "Dividends" in Part II, Item 5 of this report.
Debt
On March 4, 2014, we entered into a $250,000 Third Amended and Restated Credit Agreement (the “2014 Credit Agreement”). The 2014 Credit Agreement includes a five-year $250,000 senior secured revolving facility with sublimits of up to $100,000 for borrowings in foreign currencies, $30,000 for letters of credit and $10,000 for swing line loans (the “2014 Revolving Credit Facility”). Subject to the satisfaction of certain conditions precedent including the consent of the lenders, the Company may from time to time increase the 2014 Revolving Credit Facility by an aggregate amount not to exceed $100,000. The 2014 Credit Agreement expires on March 4, 2019. Additionally, subject to certain restrictions and conditions (i) any of our domestic or foreign subsidiaries may become borrowers and (ii) borrowings may occur in multi-currencies.
On July 31, 2017, we had $126,000 of outstanding borrowings under the 2014 Credit Agreement. Subsequent to July 31, 2017, we borrowed $61,300 to fund the purchase price and transaction costs of the BHT acquisition.
Borrowings under the 2014 Credit Agreement bear interest at rates ranging from 0.25% to 1.25% above the lender’s base rate, or at rates ranging from 1.25% to 2.25% above the London Interbank Offered Rate (“LIBOR”), depending upon the Company’s “Consolidated Leverage Ratio,” which is defined as the consolidated ratio of total funded debt to earnings before interest, taxes, depreciation and amortization, and as further adjusted under the terms of the 2014 Credit Agreement (“Consolidated EBITDA”). The 2014 Credit Agreement also provides for fees on the unused portion of our facility at rates ranging from 0.20% to 0.40%, depending upon our Consolidated Leverage Ratio.
For further information regarding the 2014 Credit Agreement, including a description of affirmative and negative covenants, see Note 10 to our consolidated financial statements in Part II, Item 8 of this report.
(dollar amounts in thousands except share and per share data or as otherwise specified) 33
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Financing Needs
On July 31, 2017, our long-term debt of $126,000, net of our cash and cash equivalents of $36,584, was $89,416. Stockholders' equity as of that date was $523,932.
Our operating segments generate significant cash from operations. At July 31, 2017, we had a cash balance of $36,584, of which $15,032 was held by foreign subsidiaries. Our foreign cash is needed by our foreign subsidiaries for working capital purposes as well as for current international growth initiatives. Accordingly, our foreign unremitted earnings are considered permanently reinvested and unavailable for repatriation.
We believe that our current cash position, anticipated cash flows from operations and the funds available under our 2014 Credit Agreement will be sufficient to satisfy our worldwide cash operating requirements for the foreseeable future based upon our existing operations, particularly given that we historically have not needed to borrow for working capital purposes. At September 28, 2017, approximately $76,000 was available under our 2014 Credit Agreement.
Inflation
Although overall inflation did not have a significant effect on our business, an increase in commodity prices can adversely affect our gross margins. Specifically, our businesses can be adversely impacted by rising fuel and oil prices and are heavily reliant on certain raw materials, such as chemicals, paper, resin, stainless steel and plastic components. From time to time, we experience price increases for raw materials. If we are unable to implement price increases to our customers, our gross margins could be adversely affected.
Compensation Agreements
We have previously entered into various severance contracts with executives of the Company, including our corporate executive officers and certain of our subsidiary Chief Executive Officers, which define certain compensation arrangements relating to various employment termination scenarios, and multi-year employment agreements with certain executive officers of businesses we have acquired. Additionally, in March 2016 we entered into a succession plan agreement due to the planned retirement of our Chief Executive Officer who was succeeded on July 31, 2016, but remained employed as a senior advisor until October 15, 2016. This succession plan agreement requires future payments to our former Chief Executive Officer beginning in fiscal 2017 for transition-related services. The majority of those future payments were recorded in general and administrative expenses from March 17, 2016 through his October 15, 2016 retirement date.
Other Long-Term Obligations
Other long-term obligations include monies owed to the central bank of Italy related to a liability assumed as part of the International Medical Service S.r.l. acquisition in fiscal 2015 and deferred compensation arrangements for certain former Medivators directors and officers and is recorded in other long-term liabilities.
Commitments and Contractual Obligations
As of July 31, 2017, aggregate annual required payments over the next five years and thereafter under our contractual obligations that have long-term components are as follows:
Year Ended July 31, | |||||||||||||||||||||||||||
2018 | 2019 | 2020 | 2021 | 2022 | Thereafter | Total | |||||||||||||||||||||
Maturity of the credit facility | $ | — | $ | 126,000 | $ | — | $ | — | $ | — | $ | — | $ | 126,000 | |||||||||||||
Expected interest payments under the credit facility | 3,150 | 1,838 | — | — | — | — | 4,988 | ||||||||||||||||||||
Minimum commitments under noncancelable operating leases | 6,522 | 5,278 | 3,779 | 2,719 | 1,231 | 2,454 | 21,983 | ||||||||||||||||||||
Compensation agreements | 7,836 | 1,210 | 510 | 384 | 292 | 292 | 10,524 | ||||||||||||||||||||
Other long-term obligations | 202 | 220 | 12 | 3 | — | — | 437 | ||||||||||||||||||||
Total contractual obligations | $ | 17,710 | $ | 134,546 | $ | 4,301 | $ | 3,106 | $ | 1,523 | $ | 2,746 | $ | 163,932 | |||||||||||||
(dollar amounts in thousands except share and per share data or as otherwise specified) 34
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Critical Accounting Policies
Our discussion and analysis of our financial condition and results of operations are based upon our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States. The preparation of these financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses, and related disclosures of contingent assets and liabilities. On an ongoing basis, we continually evaluate our estimates. We base our estimates on historical experience and on various other assumptions that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates.
Our significant accounting policies are described more fully in Note 2 to our consolidated financial statements in Part II, Item 8 of this report. We believe the following critical accounting policies affect our more significant judgments and estimates used in the preparation of our consolidated financial statements.
Revenue Recognition
Revenue on product sales is recognized as products are shipped to customers and title passes. The passing of title is determined based upon the FOB terms specified for each shipment. With respect to endoscopy and dialysis products, shipment terms are generally FOB origin for common carrier and when our distribution fleet is utilized (except for three customers in our Water Purification and Filtration segment and several customers in our Endoscopy segment whereby all products are shipped FOB destination). With respect to water purification and filtration and healthcare disposable products, shipment terms may be either FOB origin or destination. Customer acceptance for the majority of our product sales occurs at the time of delivery. With respect to a portion of water purification and filtration and endoscopy product sales, equipment is sold as part of a system for which the equipment is functionally interdependent or the customer’s purchase order specifies “ship-complete” as a condition of delivery. Revenue recognition on such sales is deferred until all equipment has been delivered, or post-delivery obligations such as installation have been substantially fulfilled such that the products are deemed functional by the end-user. All shipping and handling fees invoiced to customers, such as freight, are recorded as revenue (and related costs are included within cost of sales) at the time the sale is recognized.
A portion of our endoscopy, water purification and filtration and dialysis sales are recognized as multiple element arrangements, whereby revenue is allocated to the equipment, installation and consumable components based upon vendor specific objective evidence, which includes comparable historical transactions of similar equipment, installation and consumables sold as stand-alone components. If vendor-specific objective evidence of selling price is not available, we allocate revenue to the elements of the bundled arrangement using the estimated selling price method in order to qualify the components as separate units of accounting. Revenue on the equipment and consumables components are recognized as the equipment or consumable is shipped to customers and title passes. Revenue on the installation component is recognized when the installation is complete.
A portion of our healthcare disposables sales relating to the mail-in spore test kit is recorded as deferred revenue when initially sold. We recognize the revenue on these test kits using an estimate based on historical experience of the amount of time that elapses from the point of sale to when the kit is returned to us and we communicate to the customer the results of the required laboratory test. The related cost of the kits is recorded in inventory and recognized in cost of sales as the revenue is earned.
Revenue on service sales is recognized when repairs are completed at the customer’s location or when repairs are completed at our facilities and the products are shipped to customers. With respect to certain service contracts in our Endoscopy and Water Purification and Filtration operating segments, service revenue is recognized on a straight-line basis over the contractual term of the arrangement.
None of our sales contain right-of-return provisions. Customer claims for credit or return due to damage, defect, shortage or other reason must be pre-approved by us before credit is issued or such product is accepted for return. No cash discounts for early payment are offered except with respect to a small portion of our product sales in each segment. We do not offer price protection, although advance pricing contracts or required notice periods prior to implementation of price increases exist for certain customers with respect to many of our products. With respect to certain of our dialysis, healthcare disposables, water purification and filtration and endoscopy customers, rebates are provided. Such rebates, which consist primarily of volume rebates, are provided for as a reduction of sales at the time of revenue recognition, and amounted to $6,291, $5,944, and $5,597 in fiscal 2017, 2016, and 2015, respectively. Such allowances are determined based on estimated projections of sales volume for the entire rebate periods. If it becomes known that sales volume to customers will deviate from original projections, the rebate provisions originally established would be adjusted accordingly.
(dollar amounts in thousands except share and per share data or as otherwise specified) 35
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Accounts Receivable and Allowance for Doubtful Accounts
Accounts receivable consist of amounts due to us from normal business activities. Allowances for doubtful accounts are reserves for the estimated loss from the inability of customers to make required payments. We use historical experience as well as current market information in determining the estimate. While actual losses have historically been within management’s expectations and provisions established, if the financial condition of our customers were to deteriorate, resulting in an impairment of their ability to make payments, additional allowances may be required. Alternatively, if certain customers paid their delinquent receivables, reductions in allowances may be required.
Inventories
Inventories consist of raw materials, work-in-process and finished products which are sold in the ordinary course of our business and are stated at the lower of cost (first-in, first-out) or market. In assessing the value of inventories, we must make estimates and judgments regarding reserves required for product obsolescence, aging of inventories and other issues potentially affecting the saleable condition of products. In performing such evaluations, we use historical experience as well as current market information. With few exceptions, the saleable value of our inventories has historically been within management’s expectation and provisions established, however, rapid changes in the market due to competition, technology and various other factors could impact the saleable value of our inventories, resulting in the need for additional reserves.
Goodwill and Intangible Assets
Certain of our identifiable intangible assets, including customer relationships, technology, brand names, non-compete agreements and patents, are amortized using the straight-line method over their estimated useful lives which range from 3 to 20 years. Additionally, we have recorded goodwill and trademarks and trade names, all of which have indefinite useful lives and are therefore not amortized. All of our intangible assets and goodwill are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable, and goodwill and intangible assets with indefinite lives are reviewed for impairment at least annually. Our management is responsible for determining if impairment exists and considers a number of factors, including third-party valuations, when making these determinations.
While the results of these annual reviews have historically not indicated impairment, impairment reviews are highly dependent on management’s projections of our future operating results and cash flows (which management believes to be reasonable), discount rates based on the Company’s weighted average cost of capital and appropriate benchmark peer companies. Assumptions used in determining future operating results and cash flows include current and expected market conditions and future sales and earnings forecasts. Subsequent changes in these assumptions and estimates could result in future impairment. Although we consistently use the same methods in developing the assumptions and estimates underlying the fair value calculations, such estimates are uncertain by nature and can vary from actual results.
Long-Lived Assets
We evaluate the carrying value of long-lived assets including property, equipment and other assets whenever events or changes in circumstances indicate that the carrying value may not be recoverable. An assessment is made to determine if the sum of the expected future non-discounted cash flows from the use of the assets and eventual disposition is less than the carrying value. If the sum of the expected non-discounted cash flows is less than the carrying value, an impairment loss is recognized based on fair value. With the exception of the impairment on an acquired license as further described in Note 6 to our consolidated financial statements in Part II, Item 8 of this report, our historical assessments of our long-lived assets have not differed significantly from the actual amounts realized. However, the determination of fair value requires us to make certain assumptions and estimates and is highly subjective. On July 31, 2017, management concluded that no other events or changes in circumstances have occurred that would indicate that the carrying amount of our long-lived assets may not be recoverable.
Warranties
We provide for estimated costs that may be incurred to remedy deficiencies of quality or performance of our products at the time of revenue recognition. Most of our products have a one year warranty, although certain endoscopy and water purification and filtration products that require installation may carry a warranty period of up to twenty-four months. Additionally, many of our consumables, accessories, parts and service have a 90 day warranty. We record provisions for product warranties as a component of cost of sales based upon an estimate of the amounts necessary to settle existing and future claims on products sold. The historical relationship of warranty costs to products sold is the primary basis for the estimate. A significant increase in third party service repair rates, the cost and availability of parts or the frequency of claims could have a material adverse impact on our results for the period or periods in which such claims or additional costs materialize.
(dollar amounts in thousands except share and per share data or as otherwise specified) 36
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Management reviews its warranty exposure periodically and believes that the warranty reserves are adequate; however, actual claims incurred could differ from original estimates, requiring adjustments to the reserves.
Stock-Based Compensation
Stock compensation expense is recognized for any option or stock award grant based upon the fair value of the award. We estimate the fair value of our stock-based compensation using fair value pricing models which require the use of significant assumptions. The determination of fair value using valuation models is affected by our stock price as well as assumptions regarding a numbers of subjective variables. These variables may include, but are not limited to, the expected stock price volatility over the term of the expected life of the award, the expected dividend yield, the expected life of the award, the probability of meeting performance objectives and the stock price of our peers in the S&P Healthcare Equipment Index.
The stock-based compensation expense recorded in our financial statements may not be representative of the effect of stock-based compensation expense in future periods due to the level of awards issued in prior years (which level may not be similar in the future), modifications to existing awards, accelerated vesting related to certain employment terminations, the level of actual forfeitures, the ability to meet performance objectives and assumptions used in determining fair value.
Business Combinations
Acquisitions require significant estimates and judgments related to the fair value of assets acquired and liabilities assumed. We determine fair value based on the estimated price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. Such initial fair value amounts as well as other acquired assets and liabilities, including deferred tax assets and liabilities, are sometimes refined requiring subsequent adjustments.
Certain liabilities and reserves are subjective in nature. We reflect such liabilities and reserves based upon the most recent information available. In conjunction with our acquisitions, such subjective liabilities and reserves principally include contingent consideration, certain deferred income tax liabilities, income tax and sales and use tax exposures, including tax liabilities related to our foreign subsidiaries, as well as reserves for accounts receivable, inventories, warranties and contingent obligations. We account for contingent consideration relating to business combinations as a liability and an increase to goodwill at the date of the acquisition and continually re-measure the liability at each balance sheet date by recording changes in the fair value through our consolidated statements of income. We determine the fair value of contingent consideration based on future operating projections under various potential scenarios and weight the probability of these outcomes. Similarly, other acquisition related liabilities can be required to be recorded at fair value at the date of the acquisition and continually re-measured at each balance sheet date. The ultimate settlement of liabilities relating to business combinations may be for amounts which are materially different from the amounts initially recorded and may cause volatility in our results of operations.
Off-balance Sheet Arrangements
As of July 31, 2017, we did not have any off-balance sheet arrangements as defined in Item 303(a)(4)(ii) of Regulation S-K.
Recent Accounting Pronouncements
Refer to Note 2 to the consolidated financial statements in Part II, Item 8 of this report.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk.
We are exposed to market risks arising principally from changes in interest rates and foreign currency.
Interest Rate Market Risk
With respect to interest rate risk, since our credit facility consists of outstanding debt at prevailing market rates of interest, principally under LIBOR contracts ranging from one to twelve months, our market risk with respect to such debt is the increase in interest expense which would result from higher interest rates associated with LIBOR. Our outstanding debt of $126,000 at July 31, 2017 has expected annual interest payments of approximately $3,150 using an effective interest rate of 2.50% as described above. Therefore, a 100 basis-point increase in average LIBOR interest rates would result in incremental interest expense of approximately $126. We monitor our interest rate risk, but presently do not utilize any interest rate derivatives that would mitigate our interest rate exposure. However, all of our outstanding borrowings were under LIBOR contracts at July 31, 2017 that have expiration dates ranging from one to three months at fixed interest rates for the contract periods; therefore, we
(dollar amounts in thousands except share and per share data or as otherwise specified) 37
Cantel Medical Corp. 2017 Annual Report on Form 10-K
are substantially protected throughout the majority of fiscal 2018 from any significant exposure associated with increasing LIBOR rates, assuming we do not increase our outstanding debt. Additionally, we maintained a cash balance of $36,584 at July 31, 2017 which is maintained in cash or invested in low risk and low return cash equivalents such as U.S. money market funds with leading banking institutions. An increase in interest rates would generate additional interest income for us from these low risk cash equivalents, which would partially offset the adverse impact of the additional interest expense. Our other long-term liabilities would not be materially affected by an increase in interest rates.
Foreign Currency Market Risk
Changes in the value of the Euro, British Pound, Singapore dollar, Canadian dollar, Australian dollar and the Chinese Renminbi against the U.S. dollar affect our results of operations because certain cash bank accounts, accounts receivable and liabilities of Cantel and its subsidiaries are denominated and ultimately settled in U.S. dollars or these foreign currencies, but must be converted into each entity’s functional currency. Furthermore, the financial statements of most of our international subsidiaries are translated using the accounting policies described in Note 2 to the consolidated financial statements in Part II, Item 8 of this report, and therefore are impacted by changes in the international entities’ functional currency relative to the U.S. dollar.
We use a sensitivity analysis to assess the market risk associated with our foreign currency transactions. Market risk is defined here as the potential change in fair value resulting from an adverse movement in foreign currency exchange rates. Overall for fiscal 2017 and 2016, a uniform 15% adverse movement in foreign currency rates would have resulted in realized losses (after tax) of approximately $3,595 and $1,680, respectively. Conversely, for fiscal 2017 and 2016, a uniform 15% favorable movement in foreign currency rates would have resulted in realized gains (after tax) of approximately $3,595 and $1,680, respectively.
For fiscal 2017 and 2016, the realized losses (after tax) would have resulted primarily from increases in the values of the Euro and Canadian dollar relative to the U.S. dollar and decreases in the values of the British Pound and Singapore dollar relative to the U.S. dollar due to the composition of our assets and liabilities denominated in foreign currencies and the translation of our foreign subsidiaries’ financial statements. However, the use of foreign currency forward contracts would partially offset such realized losses.
In order to hedge against the impact of fluctuations in the value of the Euro, British Pound, Canadian dollar, Australian dollar and Singapore dollar relative to the U.S. dollar on the conversion of such net assets into the functional currencies, we enter into short-term contracts to purchase Euros, British Pounds, Canadian dollars, Australian dollars and Singapore dollars forward, which contracts are one-month in duration. These short-term contracts are designated as fair value hedge instruments. There were nine foreign currency forward contracts with an aggregate notional value of $24,762 at July 31, 2017, which covered certain assets and liabilities that were denominated in currencies other than each entity’s functional currency. These foreign currency forward contracts are continually replaced with new one-month contracts as long as we have significant net assets that are denominated and ultimately settled in currencies other than each entity’s functional currency. Gains and losses related to these hedging contracts to buy Euros, British Pounds, Canadian dollars, Australian dollars and Singapore dollars forward are immediately realized within general and administrative expenses due to the short-term nature of such contracts. For fiscal 2017, such forward contracts partially offset the impact on operations related to certain assets and liabilities that are denominated in currencies other than each entity’s functional currency. We do not currently hedge against the impact of fluctuations in the value of the Chinese Renminbi relative to the U.S. dollar because the overall foreign currency exposures relating to those currencies are currently not deemed significant.
Overall, fluctuations in the rates of currency exchange did not have a material impact upon our net income in fiscal 2017 compared with fiscal 2016.
For purposes of translating the balance sheet at July 31, 2017 compared with July 31, 2016, the total of the foreign currency movements resulted in a foreign currency translation gain of $1,895 for fiscal 2017, primarily due to the decrease in the value of the U.S. dollar relative to the Euro and Canadian dollar, partially offset by the weakening of the British Pound relative to the U.S. dollar.
(dollar amounts in thousands except share and per share data or as otherwise specified) 38
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Item 8. Financial Statements and Supplementary Data.
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders
Cantel Medical Corp.
We have audited the accompanying consolidated balance sheets of Cantel Medical Corp. as of July 31, 2017 and 2016, and the related consolidated statements of income, comprehensive income, changes in stockholders’ equity and cash flows for each of the three years in the period ended July 31, 2017. Our audits also included the financial statement schedule included in the Index at Item 15(a). These financial statements and schedule are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements and schedule based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position of Cantel Medical Corp. at July 31, 2017 and 2016, and the consolidated results of its operations and its cash flows for each of the three years in the period ended July 31, 2017, in conformity with U.S. generally accepted accounting principles. Also, in our opinion, the related financial statement schedule, when considered in relation to the basic financial statements taken as a whole, presents fairly in all material respects the information set forth therein.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), Cantel Medical Corp.’s internal control over financial reporting as of July 31, 2017, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) and our report dated September 28, 2017 expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
New York, New York
September 28, 2017
(dollar amounts in thousands except share and per share data or as otherwise specified) 39
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders
Cantel Medical Corp.
We have audited Cantel Medical Corp.’s internal control over financial reporting as of July 31, 2017, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). Cantel Medical Corp.’s management is responsible for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the company’s internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
As indicated in the accompanying Management’s Report on Internal Control over Financial Reporting, management’s assessment of and conclusion on the effectiveness of internal control over financial reporting did not include the internal controls of Accutron, Inc., which is included in the 2017 consolidated financial statements of Cantel Medical Corp. and constituted 7.6% and 10.3% of total and net assets, respectively, as of July 31, 2017, and 3.0% and 4.1% of net sales and net income for the year then ended. Our audit of internal control over financial reporting of Cantel Medical Corp. also did not include an evaluation of the internal control over financial reporting of Accutron, Inc.
In our opinion, Cantel Medical Corp. maintained, in all material respects, effective internal control over financial reporting as of July 31, 2017, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets of Cantel Medical Corp. as of July 31, 2017 and 2016 and the related consolidated statements of income, comprehensive income, changes in stockholders’ equity and cash flows for each of the three years in the period ended July 31, 2017 of Cantel Medical Corp. and our report dated September 28, 2017 expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
New York, New York
September 28, 2017
(dollar amounts in thousands except share and per share data or as otherwise specified) 40
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Consolidated Balance Sheets
July 31, | |||||||
2017 | 2016 | ||||||
Assets | |||||||
Current assets: | |||||||
Cash and cash equivalents | $ | 36,584 | $ | 28,367 | |||
Accounts receivable, net of allowance for doubtful accounts of $1,808 in 2017 and $1,850 in 2016 | 110,656 | 93,332 | |||||
Inventories, net | 98,724 | 91,486 | |||||
Prepaid expenses and other current assets | 11,407 | 9,557 | |||||
Total current assets | 257,371 | 222,742 | |||||
Property and equipment, net | 88,338 | 74,604 | |||||
Intangible assets, net | 124,512 | 111,719 | |||||
Goodwill | 311,445 | 280,318 | |||||
Other assets | 4,707 | 5,149 | |||||
Total assets | $ | 786,373 | $ | 694,532 | |||
Liabilities and stockholders’ equity | |||||||
Current liabilities: | |||||||
Accounts payable | $ | 27,469 | $ | 26,263 | |||
Compensation payable | 27,468 | 25,555 | |||||
Accrued expenses | 23,393 | 20,283 | |||||
Deferred revenue | 25,282 | 20,173 | |||||
Income taxes payable | 3,167 | 4,061 | |||||
Total current liabilities | 106,779 | 96,335 | |||||
Long-term debt | 126,000 | 116,000 | |||||
Deferred income taxes | 24,714 | 23,579 | |||||
Other long-term liabilities | 4,948 | 4,248 | |||||
Total liabilities | 262,441 | 240,162 | |||||
Commitments and Contingencies (Note 12) | |||||||
Stockholders’ equity: | |||||||
Preferred Stock, par value $1.00 per share; authorized 1,000,000 shares; none issued | — | — | |||||
Common Stock, par value $.10 per share; authorized 75,000,000 shares; issued 2017 - 46,194,370 shares, outstanding 2017 - 41,728,934 shares; issued 2016 - 46,084,047 shares, outstanding 2016 - 41,708,214 shares | 4,619 | 4,608 | |||||
Additional paid-in capital | 174,602 | 165,573 | |||||
Retained earnings | 407,590 | 342,053 | |||||
Accumulated other comprehensive loss | (9,900 | ) | (11,795 | ) | |||
Treasury Stock, at cost; 2017 - 4,465,440 shares; 2016 - 4,375,833 shares | (52,979 | ) | (46,069 | ) | |||
Total stockholders’ equity | 523,932 | 454,370 | |||||
Total liabilities and stockholders' equity | $ | 786,373 | $ | 694,532 | |||
See accompanying Notes to Consolidated Financial Statements.
(dollar amounts in thousands except share and per share data or as otherwise specified) 41
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Consolidated Statements of Income
Year Ended July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Net sales | |||||||||||
Product sales | $ | 684,678 | $ | 584,750 | $ | 493,656 | |||||
Product service | 85,479 | 80,005 | 71,348 | ||||||||
Total net sales | 770,157 | 664,755 | 565,004 | ||||||||
Cost of sales | |||||||||||
Product sales | 343,641 | 300,704 | 260,903 | ||||||||
Product service | 59,356 | 54,865 | 50,634 | ||||||||
Total cost of sales | 402,997 | 355,569 | 311,537 | ||||||||
Gross profit | 367,160 | 309,186 | 253,467 | ||||||||
Expenses: | |||||||||||
Selling | 116,113 | 99,062 | 80,787 | ||||||||
General and administrative | 122,270 | 97,463 | 77,897 | ||||||||
Research and development | 18,367 | 15,410 | 14,022 | ||||||||
Total operating expenses | 256,750 | 211,935 | 172,706 | ||||||||
Income from operations | 110,410 | 97,251 | 80,761 | ||||||||
Interest expense, net | 4,303 | 3,320 | 2,364 | ||||||||
Other income | (126 | ) | — | — | |||||||
Loss on sale of business | — | — | 2,206 | ||||||||
Income before income taxes | 106,233 | 93,931 | 76,191 | ||||||||
Income taxes | 34,855 | 33,978 | 28,238 | ||||||||
Net income | $ | 71,378 | $ | 59,953 | $ | 47,953 | |||||
Earnings per common share: | |||||||||||
Basic | $ | 1.71 | $ | 1.44 | $ | 1.16 | |||||
Diluted | $ | 1.71 | $ | 1.44 | $ | 1.15 | |||||
Dividends per common share | $ | 0.14 | $ | 0.12 | $ | 0.10 | |||||
See accompanying Notes to Consolidated Financial Statements.
(dollar amounts in thousands except share and per share data or as otherwise specified) 42
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Consolidated Statements of Comprehensive Income
Year Ended July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Net income | $ | 71,378 | $ | 59,953 | $ | 47,953 | |||||
Other comprehensive income (loss): | |||||||||||
Foreign currency translation | 1,895 | (13,019 | ) | (7,064 | ) | ||||||
Reclassification adjustment to loss on sale of business for foreign currency translation gain included in net income during the year | — | — | (1,264 | ) | |||||||
Total other comprehensive income (loss) | 1,895 | (13,019 | ) | (8,328 | ) | ||||||
Comprehensive income | $ | 73,273 | $ | 46,934 | $ | 39,625 | |||||
See accompanying Notes to Consolidated Financial Statements.
(dollar amounts in thousands except share and per share data or as otherwise specified) 43
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Consolidated Statements of Changes in Stockholders' Equity
Additional Paid-in Capital | Retained Earnings | Accumulated Other Comprehensive Loss | Treasury Stock, at cost | Total Stockholders' Equity | ||||||||||||||||||||||
Common Stock | ||||||||||||||||||||||||||
Shares | Amount | |||||||||||||||||||||||||
Balance, July 31, 2014 | 41,442,260 | $ | 4,564 | $ | 146,048 | $ | 243,306 | $ | 9,552 | $ | (38,224 | ) | $ | 365,246 | ||||||||||||
Exercises of options | 130,911 | 13 | 981 | — | — | (386 | ) | 608 | ||||||||||||||||||
Repurchases of shares | (100,286 | ) | — | — | — | — | (3,727 | ) | (3,727 | ) | ||||||||||||||||
Stock-based compensation | — | — | 5,867 | — | — | — | 5,867 | |||||||||||||||||||
Issuance of restricted stock | 144,278 | 15 | (15 | ) | — | — | — | — | ||||||||||||||||||
Cancellations of restricted stock | (12,804 | ) | (1 | ) | 1 | — | — | — | — | |||||||||||||||||
Excess tax benefit from exercises of stock options and vesting of restricted stock | — | — | 3,168 | — | — | — | 3,168 | |||||||||||||||||||
Dividends on common stock | — | — | — | (4,154 | ) | — | — | (4,154 | ) | |||||||||||||||||
Net income | — | — | — | 47,953 | — | — | 47,953 | |||||||||||||||||||
Other comprehensive loss | — | — | — | — | (8,328 | ) | — | (8,328 | ) | |||||||||||||||||
Balance, July 31, 2015 | 41,604,359 | $ | 4,591 | $ | 156,050 | $ | 287,105 | $ | 1,224 | $ | (42,337 | ) | $ | 406,633 | ||||||||||||
Repurchases of shares | (67,038 | ) | — | — | — | — | (3,732 | ) | (3,732 | ) | ||||||||||||||||
Stock-based compensation | — | — | 8,361 | — | — | — | 8,361 | |||||||||||||||||||
Issuance of restricted stock | 175,700 | 17 | (17 | ) | — | — | — | — | ||||||||||||||||||
Cancellations of restricted stock | (4,807 | ) | — | — | — | — | — | — | ||||||||||||||||||
Excess tax benefit from exercises of stock options and vesting of restricted stock | — | — | 1,179 | — | — | — | 1,179 | |||||||||||||||||||
Dividends on common stock | — | — | — | (5,005 | ) | — | — | (5,005 | ) | |||||||||||||||||
Net income | — | — | — | 59,953 | — | — | 59,953 | |||||||||||||||||||
Other comprehensive loss | — | — | — | — | (13,019 | ) | — | (13,019 | ) | |||||||||||||||||
Balance, July 31, 2016 | 41,708,214 | $ | 4,608 | $ | 165,573 | $ | 342,053 | $ | (11,795 | ) | $ | (46,069 | ) | $ | 454,370 | |||||||||||
Repurchases of shares | (89,607 | ) | — | — | — | — | (6,910 | ) | (6,910 | ) | ||||||||||||||||
Stock-based compensation | — | — | 8,844 | — | — | — | 8,844 | |||||||||||||||||||
Issuance of restricted stock | 116,506 | 12 | (12 | ) | — | — | — | — | ||||||||||||||||||
Cancellations of restricted stock | (6,179 | ) | (1 | ) | 1 | — | — | — | — | |||||||||||||||||
Excess tax benefit from exercises of stock options and vesting of restricted stock | — | — | 196 | — | — | — | 196 | |||||||||||||||||||
Dividends on common stock | — | — | — | (5,841 | ) | — | — | (5,841 | ) | |||||||||||||||||
Net income | — | — | — | 71,378 | — | — | 71,378 | |||||||||||||||||||
Other comprehensive income | — | — | — | — | 1,895 | — | 1,895 | |||||||||||||||||||
Balance, July 31, 2017 | 41,728,934 | $ | 4,619 | $ | 174,602 | $ | 407,590 | $ | (9,900 | ) | $ | (52,979 | ) | $ | 523,932 | |||||||||||
See accompanying Notes to Consolidated Financial Statements.
(dollar amounts in thousands except share and per share data or as otherwise specified) 44
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Consolidated Statements of Cash Flows
Year Ended July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Cash flows from operating activities | |||||||||||
Net income | $ | 71,378 | $ | 59,953 | $ | 47,953 | |||||
Adjustments to reconcile net income to net cash provided by operating activities: | |||||||||||
Depreciation | 15,045 | 11,989 | 10,692 | ||||||||
Amortization | 18,407 | 13,095 | 13,265 | ||||||||
Stock-based compensation expense | 8,844 | 8,361 | 5,867 | ||||||||
Amortization of debt issuance costs | 401 | 401 | 401 | ||||||||
Loss on disposal of fixed assets | 966 | 553 | 360 | ||||||||
Loss on sale of business | — | — | 2,206 | ||||||||
Impairment of assets | — | — | 1,287 | ||||||||
Fair value adjustments to acquisition related liabilities | (265 | ) | (687 | ) | (2,585 | ) | |||||
Deferred income taxes | 118 | (1,710 | ) | (1,449 | ) | ||||||
Excess tax benefits from stock-based compensation | — | (1,179 | ) | (3,168 | ) | ||||||
Changes in assets and liabilities, net of effects of business acquisitions/divestiture: | |||||||||||
Accounts receivable | (12,860 | ) | (12,729 | ) | (3,905 | ) | |||||
Inventories | 887 | (15,558 | ) | (10,075 | ) | ||||||
Prepaid expenses and other current assets | (1,005 | ) | (2,850 | ) | (2,996 | ) | |||||
Accounts payable and other current liabilities | 7,039 | 17,657 | (3,347 | ) | |||||||
Income taxes | (895 | ) | 2,972 | 4,564 | |||||||
Other assets and liabilities | 133 | — | — | ||||||||
Net cash provided by operating activities | 108,193 | 80,268 | 59,070 | ||||||||
Cash flows from investing activities | |||||||||||
Capital expenditures | (27,065 | ) | (18,889 | ) | (12,760 | ) | |||||
Proceeds from disposal of fixed assets | 47 | 96 | 25 | ||||||||
Proceeds from sale of business, net of cash retained and disposal costs | — | — | 3,767 | ||||||||
Acquisition of businesses, net of cash acquired | (70,044 | ) | (94,528 | ) | (43,567 | ) | |||||
Other, net | — | 339 | 241 | ||||||||
Net cash used in investing activities | (97,062 | ) | (112,982 | ) | (52,294 | ) | |||||
Cash flows from financing activities | |||||||||||
Borrowings under revolving credit facility | 74,000 | 96,500 | 47,000 | ||||||||
Repayments under revolving credit facility | (64,000 | ) | (59,000 | ) | (49,000 | ) | |||||
Proceeds from exercises of stock options | — | — | 608 | ||||||||
Dividends paid | (5,841 | ) | (5,005 | ) | (4,154 | ) | |||||
Excess tax benefits from stock-based compensation | — | 1,179 | 3,168 | ||||||||
Purchases of treasury stock | (6,910 | ) | (3,732 | ) | (3,727 | ) | |||||
Net cash (used in) provided by financing activities | (2,751 | ) | 29,942 | (6,105 | ) | ||||||
Effect of exchange rate changes on cash and cash equivalents | (163 | ) | (581 | ) | (732 | ) | |||||
Increase (decrease) in cash and cash equivalents | 8,217 | (3,353 | ) | (61 | ) | ||||||
Cash and cash equivalents at beginning of period | 28,367 | 31,720 | 31,781 | ||||||||
Cash and cash equivalents at end of period | $ | 36,584 | $ | 28,367 | $ | 31,720 | |||||
Supplemental disclosures of cash flow information: | |||||||||||
Cash interest payments | $ | 3,455 | $ | 3,001 | $ | 1,970 | |||||
Cash income tax payments | $ | 35,858 | $ | 33,559 | $ | 25,239 | |||||
See accompanying Notes to Consolidated Financial Statements.
(dollar amounts in thousands except share and per share data or as otherwise specified) 45
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Notes to Consolidated Financial Statements.
1. | Business Description |
Throughout this document, references to “Cantel,” “us,” “we,” “our,” and the “Company” are references to Cantel Medical Corp. and its subsidiaries, except where the context makes it clear the reference is to Cantel itself and not its subsidiaries. Unless otherwise indicated, references in this Form 10-K to 2017, 2016, 2015 or “fiscal” 2017, 2016, 2015 or other years refer to our fiscal year ended July 31 of that respective year, and references to 2017 or “fiscal” 2018 refer to our fiscal year ending July 31, 2018.
Cantel is a leading provider of infection prevention products and services in the healthcare market, specializing in the following reportable segments:
Endoscopy: designs, develops, manufactures, sells and installs a comprehensive offering of products and services comprising a complete circle of infection prevention solutions. Our products include endoscope reprocessing and endoscopy procedure products.
Water Purification and Filtration: designs, develops, manufactures, sells and installs water purification systems for medical, pharmaceutical and other bacteria controlled applications. We also provide filtration/separation and disinfectant technologies to the medical and life science markets through a worldwide distributor network.
Healthcare Disposables: designs, manufactures, sells, supplies and distributes a broad selection of infection prevention healthcare products, the majority of which are single-use products used by dental practitioners.
Dialysis: designs, develops, manufactures, sells and services reprocessing systems and sterilants for dialyzers (a device serving as an artificial kidney), as well as dialysate concentrates and supplies utilized for renal dialysis.
In addition, through April 7, 2015, we had another operating segment, known as Specialty Packaging. This segment included specialty packaging and thermal control products, as well as related compliance training, for the transport of infectious and biological specimens and thermally sensitive pharmaceutical, medical and other products. The Specialty Packaging operating segment, which comprised the Other reporting segment for financial reporting purposes, was divested on April 7, 2015. See Note 17, "Information as to Operating Segments and Foreign and Domestic Operations."
Most of our equipment, consumables and supplies are used to help prevent the occurrence or spread of infections.
2. | Summary of Significant Accounting Policies |
The following is a summary of our significant accounting policies used to prepare our consolidated financial statements.
Principles of Consolidation
The consolidated financial statements include the accounts of Cantel and its wholly-owned subsidiaries. All intercompany transactions and balances have been eliminated in consolidation. Certain prior year amounts have been reclassified to conform to the current year's presentation.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States requires us to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. Actual results could differ from those estimates. On an ongoing basis, we evaluate the adequacy of our reserves and the estimates used in calculations of reserves as well as other judgmental financial statement items, including, but not limited to: collectability of accounts receivable, volume rebates and trade-in allowances, inventory values and obsolescence reserves, warranty reserves, contingent consideration, contingent guaranteed obligations, depreciation and amortization periods, deferred income taxes, goodwill and intangible assets, impairment of long-lived assets, unrecognized tax benefits for uncertain tax positions, reserves for legal exposure, stock-based compensation and expense accruals. Such estimates and assumptions are subjective in nature. We reflect such amounts based upon the most recent information available.
Subsequent Events
We have evaluated subsequent events for disclosure through the date of issuance of the accompanying consolidated financial statements.
(dollar amounts in thousands except share and per share data or as otherwise specified) 46
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Revenue Recognition
Revenue on product sales is recognized as products are shipped to customers and title passes. The passing of title is determined based upon the FOB terms specified for each shipment. With respect to endoscopy and dialysis products, shipment terms are generally FOB origin for common carrier and when our distribution fleet is utilized (except for one large customer in dialysis and several endoscopy customers whereby all products are shipped FOB destination). With respect to water purification and filtration and healthcare disposable products, shipment terms may be either FOB origin or destination. Customer acceptance for the majority of our product sales occurs at the time of delivery. With respect to a portion of water purification and filtration and endoscopy product sales, equipment is sold as part of a system for which the equipment is functionally interdependent or the customer’s purchase order specifies “ship-complete” as a condition of delivery; revenue recognition on such sales is deferred until all equipment has been delivered, or post-delivery obligations such as installation have been substantially fulfilled such that the products are deemed functional by the end-user. All shipping and handling fees invoiced to customers, such as freight, are recorded as revenue (and related costs are included within cost of sales) at the time the sale is recognized.
A portion of our endoscopy, water purification and filtration and dialysis sales are recognized as multiple element arrangements, whereby revenue is allocated to the equipment, installation and consumable components based upon vendor specific objective evidence, which includes comparable historical transactions of similar equipment, installation and consumables sold as stand-alone components. If vendor-specific objective evidence of selling price is not available, we allocate revenue to the elements of the bundled arrangement using the estimated selling price method in order to qualify the components as separate units of accounting. Revenue on the equipment and consumables components are recognized as the equipment or consumable is shipped to customers and title passes. Revenue on the installation component is recognized when the installation is complete.
A portion of our healthcare disposables sales relating to the mail-in spore test kit is recorded as deferred revenue when initially sold. We recognize the revenue on these test kits using an estimate based on historical experience of the amount of time that elapses from the point of sale to when the kit is returned to us and we communicate to the customer the results of the required laboratory test. The related cost of the kits is recorded in inventory and recognized in cost of sales as the revenue is earned.
Revenue on service sales is recognized when repairs are completed at the customer’s location or when repairs are completed at our facilities and the products are shipped to customers. With respect to certain service contracts in our Endoscopy and Water Purification and Filtration operating segments, service revenue is recognized on a straight-line basis over the contractual term of the arrangement.
Our endoscopy products and services are sold directly to hospitals and other end-users in the United States and primarily to distributors internationally except for the United Kingdom, Italy, Netherlands, Singapore, China and Germany where we sell directly to hospitals and other end-users. Water purification and filtration products and services are sold directly to hospitals, dialysis clinics, pharmaceutical and biotechnology companies, laboratories, medical products and service companies and other end-users as well as through third-party distributors. The majority of our healthcare disposable products are sold to third party distributors, and with respect to some of our sterility assurance products, to hospitals, surgery centers, physician and dental offices, dental schools, medical research companies, laboratories and other end-users. The majority of our dialysis products are sold to dialysis clinics and hospitals. Sales to all of these customers follow our revenue recognition policies.
None of our sales contain right-of-return provisions. Customer claims for credit or return due to damage, defect, shortage or other reason must be pre-approved by us before credit is issued or such product is accepted for return. No cash discounts for early payment are offered except with respect to a small portion of our product sales in each segment. We do not offer price protection, although advance pricing contracts or required notice periods prior to implementation of price increases exist for certain customers with respect to many of our products. With respect to certain of our dialysis, healthcare disposables, water purification and filtration and endoscopy customers, rebates are provided; such rebates, which consist primarily of volume rebates, are provided for as a reduction of sales at the time of revenue recognition and amounted to $6,291, $5,944, and $5,597 in fiscal 2017, 2016, and 2015, respectively. Such allowances are determined based on estimated projections of sales volume for the entire rebate periods. If it becomes known that sales volume to customers will deviate from original projections, the rebate provisions originally established would be adjusted accordingly.
Translation of Foreign Currency Financial Statements
Assets and liabilities of our foreign subsidiaries are translated into U.S. dollars at year-end exchange rates; sales and expenses are translated using average exchange rates during the year. The cumulative effect of the translation of the accounts of the foreign subsidiaries is presented as a component of accumulated other comprehensive income or loss. Foreign exchange gains and losses related to the purchase of inventories denominated in foreign currencies are included in cost of sales and foreign exchange gains
(dollar amounts in thousands except share and per share data or as otherwise specified) 47
Cantel Medical Corp. 2017 Annual Report on Form 10-K
and losses related to the incurrence of operating costs denominated in foreign currencies and the conversion of foreign assets and liabilities into functional currencies are included in general and administrative expenses.
Cash and Cash Equivalents
We consider all highly liquid investments with maturities of three months or less when purchased to be cash equivalents.
Accounts Receivable and Allowance for Doubtful Accounts
Accounts receivable consist of amounts due to us from normal business activities. Allowances for doubtful accounts are reserves for the estimated loss from the inability of customers to make required payments. We use historical experience as well as current market information in determining the estimate. While actual losses have historically been within management’s expectations and provisions established, if the financial condition of our customers were to deteriorate, resulting in an impairment of their ability to make payments, additional allowances may be required. Alternatively, if certain customers paid their delinquent receivables, reductions in allowances may be required.
Inventories
Inventories consist of raw materials, work-in-process and finished products which are sold in the ordinary course of our business and are stated at the lower of cost (first-in, first-out) or market. In assessing the value of inventories, we must make estimates and judgments regarding reserves required for product obsolescence, aging of inventories and other issues potentially affecting the saleable condition of products. In performing such evaluations, we use historical experience as well as current market information. With few exceptions, the saleable value of our inventories has historically been within management’s expectation and provisions established, however, rapid changes in the market due to competition, technology and various other factors could impact the value of our inventories, resulting in the need for additional reserves.
Property and Equipment
Property and equipment are stated at cost. Additions and improvements are capitalized, while maintenance and repair costs are expensed. When assets are retired or otherwise disposed, the cost and related accumulated depreciation or amortization is removed from the respective accounts and any resulting gain or loss is included in income. Depreciation and amortization is provided on the straight-line method over the estimated useful lives of the assets which generally range from 2-15 years for furniture and equipment, 5-32 years for buildings and improvements and the shorter of the life of the asset or the life of the lease for leasehold improvements. Depreciation expense related to property and equipment in fiscal 2017, 2016 and 2015 was $15,045, $11,989 and $10,692, respectively.
Goodwill and Intangible Assets
Certain of our identifiable intangible assets, including customer relationships, technology, brand names, non-compete agreements and patents, are amortized using the straight-line method over their estimated useful lives which range from 3 to 20 years. Additionally, we have recorded goodwill and trademarks and trade names, all of which have indefinite useful lives and are therefore not amortized. All of our intangible assets and goodwill are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable, and goodwill and intangible assets with indefinite lives are reviewed for impairment at least annually. Our management is responsible for determining if impairment exists and considers a number of factors, including third-party valuations, when making these determinations.
We first assess qualitative factors to determine whether it is more likely than not that the fair value of the reporting unit is less than the carrying amount before proceeding to step one of the two-step quantitative goodwill impairment test, if necessary. Such qualitative factors that are assessed include evaluating a segment’s financial performance, industry and market conditions, macroeconomic conditions and specific issues that can directly affect the segment such as changes in business strategies, competition, supplier relationships, operating costs, regulatory matters, litigation and the composition of the segment’s assets due to acquisitions or other events. At July 31, 2017, because we determined through qualitative factors that the fair values of our Endoscopy, Water Purification and Filtration and Healthcare Disposables segments were unlikely to be less than the carrying value, we did not proceed to step one of the two-step quantitative goodwill impairment test for those three segments. We performed step one of the two-step quantitative goodwill impairment test for Dialysis due to the continuing shift by our customers from reusable to single-use dialyzers, which is having an adverse impact on our business and is expected to continue. In performing a detailed quantitative review for goodwill impairment, management uses a two-step process that begins with an estimation of the fair value of the related reporting units by using weighted fair value results of the discounted cash flow methodology, as well as the market
(dollar amounts in thousands except share and per share data or as otherwise specified) 48
Cantel Medical Corp. 2017 Annual Report on Form 10-K
multiple and comparable transaction methodologies, where applicable. The first step is a review for potential impairment, and the second step measures the amount of impairment, if any.
We perform our annual impairment review for indefinite lived intangibles by first assessing qualitative factors, such as those described above, to determine whether it is more likely than not that the fair value of such assets is less than the carrying values, and if necessary, we perform a quantitative analysis comparing the current fair value of our indefinite lived intangibles assets to their carrying values. At July 31, 2017, because we determined through qualitative factors that the fair values of all of our indefinite lived intangible assets were unlikely to be less than the carrying value, we did not perform a quantitative analysis for those assets. With respect to amortizable intangible assets when impairment indicators are present, management would determine whether expected future non-discounted cash flows would be sufficient to recover the carrying value of the assets; if not, the carrying value of the assets would be adjusted to their fair value.
Management concluded that none of our intangible assets or goodwill was impaired as of July 31, 2017.
Long-Lived Assets
We evaluate the carrying value of long-lived assets including property, equipment and other assets whenever events or changes in circumstances indicate that the carrying value may not be recoverable. An assessment is made to determine if the sum of the expected future non-discounted cash flows from the use of the assets and eventual disposition is less than the carrying value. If the sum of the expected non-discounted cash flows is less than the carrying value, an impairment loss is recognized based on fair value. With the exception of the impairment on an acquired license, our historical assessments of our long-lived assets have not differed significantly from the actual amounts realized. See Note 8, "Intangibles and Goodwill." However, the determination of fair value requires us to make certain assumptions and estimates and is highly subjective. On July 31, 2017, management concluded that no other events or changes in circumstances have occurred that would indicate that the carrying amount of our long-lived assets may not be recoverable.
Other Assets
Debt issuance costs associated with our credit facilities are amortized to interest expense over the life of the credit facilities. As of July 31, 2017 and 2016, such debt issuance costs, net of related amortization, were included in other assets and amounted to $580 and $946, respectively.
Warranties
We provide for estimated costs that may be incurred to remedy deficiencies of quality or performance of our products at the time of revenue recognition. Most of our products have a one year warranty, although certain endoscopy and water purification and filtration products that require installation may carry a warranty period of up to 24 months. Additionally, many of our consumables, accessories, parts and service have a 90 day warranty. We record provisions for product warranties as a component of cost of sales based upon an estimate of the amounts necessary to settle existing and future claims on products sold. The historical relationship of warranty costs to products sold is the primary basis for the estimate. A significant increase in third party service repair rates, the cost and availability of parts or the frequency of claims could have a material impact on our results for the period or periods in which such claims or additional costs materialize.
Management reviews its warranty exposure periodically and believes that the warranty reserves are adequate; however, actual claims incurred could differ from original estimates, requiring adjustments to the reserves.
Stock-Based Compensation
Stock compensation expense is recognized for any option or stock award grant based upon the fair value of the award. Our stock options and time-based stock awards are subject to graded vesting in which portions of the award vest ratably over the vesting period. We recognize compensation expense for awards subject to graded vesting using the straight-line basis over the vesting period. In October 2016, we granted for the first time to certain employees equity awards with performance conditions and equity awards with market conditions. We recognize compensation expense for the awards with performance conditions using the accelerated attribution method over the requisite service period for each separately vesting portion of the award when it is probable that the performance condition will be achieved. We record expense for the awards with market conditions ratably over the vesting period regardless of whether the market condition is satisfied. As a result of the adoption of ASU 2016-09 on August 1, 2016, we have elected to account for forfeitures as they occur, rather than estimate forfeitures over the course of the vesting period.
(dollar amounts in thousands except share and per share data or as otherwise specified) 49
Cantel Medical Corp. 2017 Annual Report on Form 10-K
We determine the fair value if each time-based stock award and performance-based stock award by using the closing market price of our common stock on the last trading date immediately prior to the date of grant. We determine the fair value of each award with market conditions using a Monte Carlo simulation model on the date of grant. We estimate the fair value of each option grant on the date of grant using the Black Scholes option valuation model. The determination of fair value using valuation models is affected by our stock price as well as assumptions regarding a number of subjective variables. These variables may include, but are not limited to, the expected price volatility over the term of the expected equity award life, the expected dividend yield, the expected equity award life, the probability of meeting performance objectives and the stock price of our peers in the S&P Healthcare Equipment Index.
Advertising Costs
Our policy is to expense advertising costs as they are incurred. Advertising costs charged to expense were $3,694, $3,349 and $3,333 in fiscal 2017, 2016 and 2015, respectively.
Income Taxes
Our provision for income taxes is based on our current period income, changes in deferred income tax assets and liabilities, statutory income tax rates, changes in uncertain tax benefits and the deductibility of expenses or availability of tax credits in various taxing jurisdictions. Tax laws are complex, subject to different interpretations by the taxpayer and the respective governmental taxing authorities and are subject to future modification, expiration or repeal by government legislative bodies. We use significant judgment on a quarterly basis in determining our annual effective income tax rate and evaluating our tax positions.
We regularly review our deferred tax assets for recoverability and establish a valuation allowance, if necessary, based on historical taxable income, projected future taxable income, and the expected timing of the reversals of existing temporary differences. Although realization is not assured, management believes it is more likely than not that the recorded deferred tax assets, as adjusted for valuation allowances, will be realized. Additionally, deferred tax liabilities are regularly reviewed to confirm that such amounts are appropriately stated. A review of our deferred tax items considers known future changes in various income tax rates, principally in the United States. If income tax rates were to change in the future, particularly in the United States and to a lesser extent Canada, the U.K. and Italy, our items of deferred tax could be materially affected. All of such evaluations require significant management judgments.
We record liabilities for an unrecognized tax benefit when a tax benefit for an uncertain tax position is taken or expected to be taken on a tax return, but is not recognized in our consolidated financial statements because it does not meet the more-likely-than-not recognition threshold that the uncertain tax position would be sustained upon examination by the applicable taxing authority. Any adjustments upon resolution of income tax uncertainties are recognized in our results of operations. Unrecognized tax benefits are analyzed periodically and adjustments are made as events occur to warrant adjustment to the related liability. Historically, we have not had significant unrecognized tax benefits.
Newly Adopted Accounting Standards
In March 2016, the Financial Accounting Standards Board ("FASB") issued Accounting Standards Update ("ASU") 2016-09, “Improvements to Employee Share-Based Payment Accounting” (“ASU 2016-09”), which simplifies the accounting for employee share-based payment transactions, including the accounting for income taxes, forfeitures and statutory tax withholding requirements. The new guidance also requires that all tax-related cash flows resulting from share-based payments to be reported as operating activities in the statement of cash flows. We early adopted ASU 2016-09 on August 1, 2016, on a prospective basis. As a result, we no longer record excess tax benefits as an adjustment to additional paid-in capital, we record such excess tax benefits as a reduction of income tax expense, which amounted to $2,241 for the year ended July 31, 2017. See Note 11, "Income Taxes" and Note 15, "Stock-based Compensation." In addition, we elected to record excess tax benefits as an operating cash flow prospectively and not adjust the prior year period. As such, the current period excess tax benefits were reflected as an operating cash flow rather than a financing cash flow on our consolidated statement of cash flows for the year ended July 31, 2017. Furthermore, we have elected to account for forfeitures as they occur, rather than estimate expected forfeitures over the course of a vesting period since forfeitures have been insignificant historically.
Recently Issued Accounting Standards
In May 2017, the FASB issued ASU 2017-09, "Scope of Modification Accounting" ("ASU 2017-09") to provide guidance about which changes to the terms or conditions of a share-based payment award require an entity to apply modification accounting in Topic 718. ASU 2017-12 is effective for fiscal years beginning after December 15, 2017 (our fiscal year 2019), including interim
(dollar amounts in thousands except share and per share data or as otherwise specified) 50
Cantel Medical Corp. 2017 Annual Report on Form 10-K
periods within that reporting period. We are currently in the process of evaluating the impact of ASU 2017-09 on our financial position and result of operations.
In January 2017, the FASB issued ASU 2017-04, “Intangibles - Goodwill and Other” (“ASU 2017-04”) to simplify the test for goodwill impairment. The revised guidance eliminates the existing Step 2 of the goodwill impairment test which required an entity to compute the implied fair value of its goodwill at the testing date in order to measure the amount of the impairment charge when the fair value of the reporting unit failed Step 1 of the goodwill impairment test. Under the revised guidance, an entity would recognize an impairment charge for the amount by which the carrying amount of the reporting unit exceeds its fair value; however, the loss recognized would not exceed the total amount of goodwill allocated to the reporting unit. The guidance will be applied on a prospective basis on or after the effective date. ASU 2017-04 is effective for fiscal years beginning after December 31, 2018 (our fiscal year 2020) and early adoption is permitted for interim or annual goodwill impairment tests performed on testing dates after January 1, 2017. The adoption of ASU 2017-04 is not expected to have a significant impact on our financial position and result of operations.
In January 2017, the FASB issued ASU 2017-01, “Business Combinations (Topic 805)” (“ASU 2017-01”) to clarify the definition of a business. The revised guidance creates a more robust framework to use in determining whether a set of assets and activities is a business. The guidance will be applied on a prospective basis on or after the effective date. ASU 2017-01 is effective for fiscal years beginning after December 15, 2017 (our fiscal year 2019), including interim periods within that reporting period. We are currently in the process of evaluating the impact of ASU 2017-01 on our financial position and result of operations.
In August 2016, the FASB issued ASU 2016-15, “Statement of Cash Flows” (“ASU 2016-15”). This new guidance will make eight targeted changes to how cash receipts and cash payments are presented and classified in the statement of cash flows. ASU 2016-15 is effective for fiscal years beginning after December 15, 2017 (our fiscal year 2019). ASU 2016-15 will require adoption on a retrospective basis unless it is impracticable to apply, in which case we would be required to apply the amendments prospectively as of the earliest date practicable. We are currently in the process of evaluating the impact of ASU 2016-15 on our financial position and result of operations.
In February 2016, FASB issued ASU 2016-02, “Leases (Topic 842)” (“ASU 2016-02”). The new guidance requires the recording of assets and liabilities arising from leases on the balance sheet accompanied by enhanced qualitative and quantitative disclosures in the notes to the financial statements. The new guidance is expected to provide transparency of information and comparability among organizations. ASU 2016-02 is effective for fiscal years beginning after December 31, 2018 (our fiscal year 2020), including interim periods within that reporting period. Early adoption is permitted as of the beginning of an interim or annual period. We are currently in the process of evaluating the impact of ASU 2016-02 on our financial position and results of operations.
In September 2015, the FASB issued ASU 2015-16, “Simplifying the Accounting for Measurement-Period Adjustments (Topic 805)” (“ASU 2015-16”). The new guidance requires an acquirer in a business combination to recognize a measurement-period adjustment during the period in which it determines the amount, and eliminates the requirement for an acquirer to account for measurement-period adjustments retrospectively. The acquirer must also disclose the amounts and reasons for adjustments to the provisional amounts. ASU 2015-16 is effective for fiscal years beginning after December 15, 2016 (our fiscal year 2018), including interim periods within that reporting period. Accordingly, we will adopt ASU 2015-06 in our first quarter of fiscal 2018. The adoption of ASU 2015-06 is not expected to have a material impact upon on our financial position and results of operations.
In July 2015, the FASB issued ASU 2015-11, “Inventory (Topic 330) Simplifying the Measurement of Inventory,” (“ASU 2015-11”). The new guidance requires companies using the first-in, first-out and average costs methods to measure inventory using the lower of cost and net realizable value, where net realizable value is the estimated selling prices in the ordinary course of business, less reasonably predictable costs of completion, disposal and transportation. ASU 2015-11 is effective for fiscal years beginning after December 15, 2016 (our fiscal year 2018), including interim periods within that reporting period. Accordingly, we will adopt ASU 2015-11 in our first quarter of fiscal 2018. The adoption of ASU 2015-11 is not expected to have a material impact upon on our financial position and results of operations.
In May 2014, the FASB issued ASU 2014-09, “Revenue from Contracts with Customers (Topic 606)” (“ASU 2014-09”), which will supersede the revenue recognition requirements in Accounting Standards Codification 605, “Revenue Recognition” ("ASC 605"). ASU 2014-09 is based on the principle that revenue is recognized to depict the transfer of goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services. It also requires additional disclosure about the nature, amount, timing and uncertainty of revenue and cash flows arising from customer contracts, significant judgments and changes in judgments, and assets recognized from costs incurred to obtain or fulfill a contract. In August 2015, the FASB issued ASU 2015-14, “Revenue from Contracts with Customers (Topic 606),” which defers the effective date of ASU 2014-09 by one year to fiscal years beginning after December 15, 2017 (our fiscal year 2019), including interim
(dollar amounts in thousands except share and per share data or as otherwise specified) 51
Cantel Medical Corp. 2017 Annual Report on Form 10-K
periods within that reporting period. In May 2016, the FASB issued ASU 2016-12, “Revenue from Contracts with Customers (Topic 606),” (“ASU 2016-12”), which provided narrow scope improvements and practical expedients relating to ASU 2014-09. In preparation for our adoption of ASU 2014-09 and ASU 2016-12 on August 1, 2018, we are obtaining representative samples of contracts and other forms of agreements with our customers in the United States and international locations and plan to evaluate the provisions contained therein in light of the five-step model specified by the new guidance. We are also evaluating the impact of the new standard on certain common practices currently employed by us and by other health care manufacturers and service providers, such as multiple-element arrangements, deferred revenues, warranties, rebates and other pricing allowances. We anticipate adopting the standard using the modified retrospective method. There may be differences in timing of revenue recognition under the new standard compared to recognition under ASC 605.
3. | Acquisitions |
Post-Fiscal 2017
BHT Group
On August 23, 2017, we purchased all of the issued and outstanding stock of BHT Group, a leader in the German market in automated endoscope reprocessing and related equipment and services for total consideration, excluding acquisition related costs, of $61,236. The BHT Group consists of a portfolio of high-quality automatic endoscope reprocessors, advanced endoscope storage and drying cabinets (products globally distributed by our Company prior to the acquisition under an agreement with BHT Group), washer-disinfectors for central sterile applications, associated technical service and parts as well as flexible endoscope repair services. BHT Group will be included in our Endoscopy segment.
Fiscal 2017
CR Kennedy
On April 1, 2017, we purchased certain endoscopy-related net assets of CR Kennedy related to its distribution and sale of our Medivators endoscopy products in Australia for total consideration, excluding acquisition related costs, of $11,999. The CR Kennedy business includes a full sales and service organization and our Medivators-branded automated endoscope reprocessors, chemistries, endoscopy procedure products and other consumables in Australia, and is included in our Endoscopy segment.
Vantage Endoscopy Inc.’s Medivators® Endoscopy Business
On September 26, 2016, we acquired certain net assets of Vantage related to its distribution and sale of our Medivators endoscopy products in Canada for total consideration, excluding acquisition-related costs, of $4,044. Vantage was our exclusive distributor of Medivators capital equipment (e.g., automated endoscope reprocessors) and related consumables and accessories in Canada, and is included in our Endoscopy segment.
Accutron, Inc.
On August 1, 2016, we acquired all of the issued and outstanding stock of Accutron, a Phoenix-based company, for total consideration, excluding acquisition-related costs, of $53,049. The Accutron business designs, manufactures and sells nitrous oxide conscious sedation equipment and single use nasal masks for use in dental procedures, and is included in our Healthcare Disposables segment.
Fiscal 2016
North American Science Associates, Inc.
On March 1, 2016, we acquired certain net assets of NAMSA for total consideration, excluding acquisition-related costs, of $13,424. The NAMSA business manufactures a broad suite of high-quality biological and chemical indicators which are used to accurately monitor the effectiveness of sterilization processes primarily for manufacturers of medical device, life science and other products, and is included in our Healthcare Disposables segment.
(dollar amounts in thousands except share and per share data or as otherwise specified) 52
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Medical Innovations Group Holdings Limited
On September 14, 2015, we acquired all of the issued and outstanding stock of MI, a company providing specialized endoscopy medical devices and products primarily in the United Kingdom for total consideration, excluding acquisition-related costs, of $79,597. The MI business includes proprietary short-term and long-term endoscope transport and storage systems, a comprehensive range of endoscopic consumable accessories, OEM mobile medical carts, as well as specialized products for patient warming and patient transfer, and is included in our Endoscopy segment.
2017 | 2016 | |||||||||||||||||||
Purchase Price Allocation | CR Kennedy | Vantage(1) | Accutron(1) | NAMSA(1) | MI | |||||||||||||||
Purchase Price: | ||||||||||||||||||||
Cash paid | $ | 11,999 | $ | 4,044 | $ | 53,049 | $ | 13,424 | $ | 79,597 | ||||||||||
Debt acquired | — | — | — | — | — | |||||||||||||||
Total | $ | 11,999 | $ | 4,044 | $ | 53,049 | $ | 13,424 | $ | 79,597 | ||||||||||
Allocation: | ||||||||||||||||||||
Property and equipment | — | 433 | 1,676 | 437 | 6,464 | |||||||||||||||
Amortizable intangible assets: | ||||||||||||||||||||
Customer relationships | 4,200 | 992 | 12,800 | 5,820 | 24,430 | |||||||||||||||
Technology | — | — | 10,000 | 1,320 | 10,930 | |||||||||||||||
Brand names | — | — | 2,000 | — | 2,030 | |||||||||||||||
Goodwill | 5,894 | 2,299 | 21,989 | 3,687 | 40,006 | |||||||||||||||
Deferred income taxes | — | — | 112 | — | (8,683 | ) | ||||||||||||||
Other working capital | 1,905 | 320 | 4,472 | 2,160 | 4,420 | |||||||||||||||
Total | $ | 11,999 | $ | 4,044 | $ | 53,049 | $ | 13,424 | $ | 79,597 | ||||||||||
_______________________________________________
(1) | The excess purchase price over net assets acquired was assigned to goodwill, all of which is deductible for income tax purposes. |
Unaudited Pro Forma Summary of Operations
The acquisitions above, both individually and in the aggregate, were not material to our consolidated results of operations or financial position and, therefore, pro forma financial information is not presented.
4. Inventories, Net
A summary of inventories, net, is as follows:
July 31, | |||||||
2017 | 2016 | ||||||
Raw materials and parts | $ | 45,831 | $ | 45,867 | |||
Work-in-process | 13,484 | 13,178 | |||||
Finished goods | 48,262 | 37,831 | |||||
Less: reserve for excess and obsolete inventory | (8,853 | ) | (5,390 | ) | |||
Total | $ | 98,724 | $ | 91,486 | |||
(dollar amounts in thousands except share and per share data or as otherwise specified) 53
Cantel Medical Corp. 2017 Annual Report on Form 10-K
5. | Property and Equipment, Net |
A summary of property and equipment, net, is as follows:
July 31, | |||||||
2017 | 2016 | ||||||
Land, buildings and improvements | $ | 46,921 | $ | 44,387 | |||
Furniture and equipment | 119,682 | 95,033 | |||||
Leasehold improvements | 7,858 | 6,048 | |||||
Less: accumulated depreciation | (86,123 | ) | (70,864 | ) | |||
Total | $ | 88,338 | $ | 74,604 | |||
6. | Derivatives |
We recognize all derivatives on the balance sheet at fair value. Derivatives that are not designated as hedges must be adjusted to fair value through earnings. If the derivative is designated as a hedge, depending on the nature of the hedge, changes in the fair value of the derivative will either be offset against the change in the fair value of the hedged assets, liabilities or firm commitments through earnings or recognized in other comprehensive income until the hedged item is recognized in earnings. The ineffective portion of the change in fair value of a derivative that is designated as a hedge will be recognized immediately in earnings. As of July 31, 2017, all of our derivatives were designated as hedges. We do not hold any derivative financial instruments for speculative or trading purposes.
Changes in the value of the Euro, British Pound, Singapore dollar, Canadian dollar, Australian dollar and the Chinese Renminbi against the U.S. dollar affect our results of operations because certain cash bank accounts, accounts receivable, and liabilities of Cantel and its subsidiaries are denominated and ultimately settled in U.S. dollars or these foreign currencies, but must be converted into each entity’s functional currency.
In order to hedge against the impact of fluctuations in the value of the Euro, British Pound, Canadian dollar, Australian dollar and Singapore dollar relative to the U.S. dollar on the conversion of such net assets into the functional currencies, we enter into short-term contracts to purchase Euros, British Pounds, Canadian dollars, Australian dollars and Singapore dollars forward, which contracts are one-month in duration. These short-term contracts are designated as fair value hedge instruments. There were nine foreign currency forward contracts with an aggregate notional value of $24,762 at July 31, 2017, which covered certain assets and liabilities that were denominated in currencies other than each entity’s functional currency. These foreign currency forward contracts are continually replaced with new one-month contracts as long as we have significant net assets that are denominated and ultimately settled in currencies other than each entity’s functional currency. For the fiscal year ended July 31, 2017, such forward contracts partially offset the impact on operations relating to certain assets and liabilities that were denominated in currencies other than each entity’s functional currency. This resulted in an immaterial amount of net currency conversion losses, net of tax, on the hedged items. Gains and losses related to hedging contracts to buy Euros, British Pounds, Canadian dollars, Australian dollars and Singapore dollars forward are immediately realized within general and administrative expenses due to the short-term nature of such contracts. We do not currently hedge against the impact of fluctuations in the value of the Chinese Renminbi relative to the U.S. dollar because the overall foreign currency exposures relating to those currencies are currently not deemed significant.
7. | Fair Value Measurements |
Fair Value Hierarchy
We apply the provisions of Accounting Standards Codification (“ASC”) 820, “Fair Value Measurements and Disclosures,” (“ASC 820”), for our financial assets and liabilities that are re-measured and reported at fair value each reporting period and our nonfinancial assets and liabilities that are re-measured and reported at fair value on a non-recurring basis. We define fair value as the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. ASC 820 establishes a three level fair value hierarchy to prioritize the inputs used in valuations, as defined below:
Level 1: Observable inputs that reflect unadjusted quoted prices for identical assets or liabilities in active markets.
(dollar amounts in thousands except share and per share data or as otherwise specified) 54
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Level 2: Inputs other than quoted prices included within Level 1 that are observable for the asset or liability, either directly or indirectly.
Level 3: Unobservable inputs for the asset or liability.
Assets and Liabilities Measured and Recorded at Fair Value on a Recurring Basis
Our financial assets that are re-measured at fair value on a recurring basis include money market funds that are classified as cash and cash equivalents in the consolidated balance sheets. These money market funds are classified within Level 1 of the fair value hierarchy and are valued using quoted market prices for identical assets.
In connection with our June 2014 acquisition of a U.K. endoscopy company (“Cantel Medical (U.K.)”), we acquired a contingent guaranteed obligation to reimburse an endoscope service company for endoscope repair costs it incurs when servicing its customers’ endoscopes that are damaged by one of Cantel Medical (U.K.)’s discontinued endoscope reprocessing machine models. The fair value of the contingent liability was $441 as of July 31, 2016. This liability continued to be adjusted periodically by the reimbursement of repair costs, as well as adjustments associated with changes in the fair value through our consolidated statements of income. During the third quarter of fiscal 2017, we ended the agreement with the endoscopy service company and decreased this liability to $0 at July 31, 2017.
In connection with the Jet Prep Ltd. ("Jet Prep") acquisition in fiscal 2014, we assumed a contingent obligation payable to the Israeli Government based on future sales. This fair value measurement was based on significant inputs not observed in the market and thus represent Level 3 measurements. As a result of the exit of the Jet Prep business, we did not update our fair value assumptions associated with this contingent obligation payable and the balance of such obligation remained at $1,138 as of July 31, 2017. See Note 8, "Intangibles and Goodwill."
The fair values of the Company’s financial instruments measured on a recurring basis were categorized as follows:
July 31, 2017 | |||||||||||||||
Level 1 | Level 2 | Level 3 | Total | ||||||||||||
Assets: | |||||||||||||||
Cash and cash equivalents: | |||||||||||||||
Money markets | $ | 102 | $ | — | $ | — | $ | 102 | |||||||
Total assets | 102 | — | — | 102 | |||||||||||
Liabilities: | |||||||||||||||
Accrued expenses: | |||||||||||||||
Assumed contingent obligation | — | — | 12 | 12 | |||||||||||
Contingent guaranteed obligation | — | — | — | — | |||||||||||
Total accrued expenses | — | — | 12 | 12 | |||||||||||
Long-term debt(1) | — | 126,000 | — | 126,000 | |||||||||||
Other long-term liabilities: | |||||||||||||||
Assumed contingent obligation | — | — | 1,126 | 1,126 | |||||||||||
Contingent guaranteed obligation | — | — | — | — | |||||||||||
Total other long-term liabilities: | — | — | 1,126 | 1,126 | |||||||||||
Total liabilities | $ | — | $ | 126,000 | $ | 1,138 | $ | 127,138 | |||||||
________________________________________________
(1) | Fair value estimated using Level 2 inputs, which were quoted prices for identical or similar instruments in markets that are not active. |
(dollar amounts in thousands except share and per share data or as otherwise specified) 55
Cantel Medical Corp. 2017 Annual Report on Form 10-K
July 31, 2016 | |||||||||||||||
Level 1 | Level 2 | Level 3 | Total | ||||||||||||
Assets: | |||||||||||||||
Cash and cash equivalents: | |||||||||||||||
Money markets | $ | 740 | $ | — | $ | — | $ | 740 | |||||||
Total assets | $ | 740 | $ | — | $ | — | $ | 740 | |||||||
Liabilities: | |||||||||||||||
Accrued expenses: | |||||||||||||||
Assumed contingent obligation | — | — | 12 | 12 | |||||||||||
Contingent guaranteed obligation | — | — | 366 | 366 | |||||||||||
Total accrued expenses | — | — | 378 | 378 | |||||||||||
Long-term debt(1) | — | 116,000 | — | 116,000 | |||||||||||
Other long-term liabilities: | |||||||||||||||
Contingent consideration | — | — | — | — | |||||||||||
Assumed contingent obligation | — | — | 1,126 | 1,126 | |||||||||||
Contingent guaranteed obligation | — | — | 75 | 75 | |||||||||||
Total other long-term liabilities: | — | — | 1,201 | 1,201 | |||||||||||
Total liabilities | $ | — | $ | 116,000 | $ | 1,579 | $ | 117,579 | |||||||
________________________________________________
(1) | Fair value estimated using Level 2 inputs, which were quoted prices for identical or similar instruments in markets that are not active. |
A reconciliation of our liabilities that are measured and recorded at fair value on a recurring basis using significant unobservable inputs (Level 3) for fiscal 2017, 2016 and 2015 is as follows:
Jet Prep Contingent Consideration | Jet Prep Assumed Contingent Obligation | Cantel Medical (U.K.) Contingent Guaranteed Obligation | Total | |||||||||||||
Balance, July 31, 2014 | $ | 2,722 | $ | 1,752 | $ | 1,395 | $ | 5,869 | ||||||||
Total net unrealized gains included in general and administrative expense in earnings | (1,971 | ) | (614 | ) | — | (2,585 | ) | |||||||||
Net purchases, issuances, sales and settlements | — | — | (507 | ) | (507 | ) | ||||||||||
Balance, July 31, 2015 | 751 | 1,138 | 888 | 2,777 | ||||||||||||
Total net unrealized (gains) losses included in general and administrative expense in earnings | (751 | ) | — | 64 | (687 | ) | ||||||||||
Net purchases, issuances, sales and settlements | — | — | (511 | ) | (511 | ) | ||||||||||
Balance, July 31, 2016 | — | 1,138 | 441 | 1,579 | ||||||||||||
Total net unrealized gains included in general and administrative expense in earnings | — | — | (265 | ) | (265 | ) | ||||||||||
Net purchases, issuances, sales and settlements | — | — | (176 | ) | (176 | ) | ||||||||||
Balance, July 31, 2017 | $ | — | $ | 1,138 | $ | — | $ | 1,138 | ||||||||
Disclosure of Fair Value of Financial Instruments
As of July 31, 2017 and 2016, the carrying amounts for cash and cash equivalents (excluding money markets), accounts receivable and accounts payable approximated fair value due to the short maturity of these instruments.
8. | Intangibles and Goodwill |
Our intangible assets with definite lives consist primarily of customer relationships, technology, brand names, non-compete agreements and patents. These intangible assets are being amortized on the straight-line method over the estimated useful lives of
(dollar amounts in thousands except share and per share data or as otherwise specified) 56
Cantel Medical Corp. 2017 Annual Report on Form 10-K
the assets ranging from 3-20 years and have a weighted average amortization period of 12 years. Amortization expense related to intangible assets was $18,407, $13,095 and $13,265 for fiscal 2017, 2016 and 2015, respectively. Our intangible assets that have indefinite useful lives, and therefore are not amortized, consist of trademarks and trade names.
The Company’s intangible assets consist of the following:
July 31, 2017 | July 31, 2016 | ||||||||||||||||||||||
Gross | Accumulated Amortization | Net | Gross | Accumulated Amortization | Net | ||||||||||||||||||
Intangible assets with finite lives: | |||||||||||||||||||||||
Customer relationships | $ | 119,576 | $ | (34,773 | ) | $ | 84,803 | $ | 100,649 | $ | (24,689 | ) | $ | 75,960 | |||||||||
Technology | 42,794 | (18,990 | ) | 23,804 | 32,767 | (11,813 | ) | 20,954 | |||||||||||||||
Brand names | 8,188 | (3,225 | ) | 4,963 | 6,194 | (2,394 | ) | 3,800 | |||||||||||||||
Non-compete agreements | 3,092 | (1,428 | ) | 1,664 | 3,092 | (1,193 | ) | 1,899 | |||||||||||||||
Patents and other registrations | 2,783 | (1,053 | ) | 1,730 | 2,508 | (913 | ) | 1,595 | |||||||||||||||
176,433 | (59,469 | ) | 116,964 | 145,210 | (41,002 | ) | 104,208 | ||||||||||||||||
Trademarks and tradenames | 7,548 | — | 7,548 | 7,511 | — | 7,511 | |||||||||||||||||
Total intangible assets | $ | 183,981 | $ | (59,469 | ) | $ | 124,512 | $ | 152,721 | $ | (41,002 | ) | $ | 111,719 | |||||||||
During fiscal 2017, we decided to exit the Jet Prep business that was acquired in fiscal 2014. The Jet Prep acquisition was a fully integrated business within our Endoscopy segment. The useful life of the technology related intangible asset was revised to its respective cease use date, which resulted in accelerated amortization of approximately $2,401 that was recorded in the consolidated statements of income. In addition, we performed a relative fair value analysis for the goodwill recorded as part of the Jet Prep acquisition and determined that all of the goodwill would remain within the Endoscopy segment. We performed our annual goodwill impairment test of all of our reportable segments as of July 31, 2017, including the Endoscopy segment, which did not result in any impairment of our goodwill.
We expect to recognize $15,617, $15,293, $13,539, $13,204 and $12,821 of amortization expense related to intangible assets in fiscal years 2018, 2019, 2020, 2021 and 2022, respectively.
Goodwill changed during fiscal 2017 and 2016 as follows:
Endoscopy | Water Purification and Filtration | Healthcare Disposables | Dialysis | Total Goodwill | |||||||||||||||
Balance, July 31, 2015 | $ | 87,007 | $ | 58,872 | $ | 87,939 | $ | 8,133 | $ | 241,951 | |||||||||
Acquisitions | 40,047 | — | 4,351 | — | 44,398 | ||||||||||||||
Foreign currency translation | (6,039 | ) | 8 | — | — | (6,031 | ) | ||||||||||||
Balance, July 31, 2016 | 121,015 | 58,880 | 92,290 | 8,133 | 280,318 | ||||||||||||||
Acquisitions | 8,193 | — | 21,989 | — | 30,182 | ||||||||||||||
Foreign currency translation | 737 | 208 | — | — | 945 | ||||||||||||||
Balance, July 31, 2017 | $ | 129,945 | $ | 59,088 | $ | 114,279 | $ | 8,133 | $ | 311,445 | |||||||||
On July 31, 2017, we performed impairment analysis of the Company’s goodwill and indefinite lived trademarks and trade names and concluded that such assets were not impaired, as more fully described in Note 2, "Summary of Significant Accounting Policies."
In fiscal 2014, we acquired a license from a third party granting us the exclusive right to manufacture, commercialize, distribute and sell an endoscopy product in exchange for a series of payments, which totaled $1,000 at January 31, 2015 and was recorded in other assets in our consolidated balance sheets. We evaluated this long-lived asset for potential impairment and determined that the future use of this acquired license was unlikely based on a recent product analysis. Accordingly, we deemed the acquired license, together with related fixed assets, to be fully impaired and recorded a loss of $1,287 during fiscal 2015 based on expected cash flows of the related endoscopy product, which was recorded in general and administrative expenses and as reductions in other assets and property and equipment in the consolidated financial statements.
(dollar amounts in thousands except share and per share data or as otherwise specified) 57
Cantel Medical Corp. 2017 Annual Report on Form 10-K
9. | Warranties |
A summary of activity in the warranty reserves follows:
Year Ended July 31, | |||||||
2017 | 2016 | ||||||
Beginning balance | $ | 2,575 | $ | 1,740 | |||
Acquisitions | 179 | 28 | |||||
Provisions | 4,880 | 4,554 | |||||
Settlements | (5,306 | ) | (3,622 | ) | |||
Foreign currency translation | — | (125 | ) | ||||
Ending balance | $ | 2,328 | $ | 2,575 | |||
The warranty provisions and settlements in fiscal 2017 and 2016 relate principally to the Company’s endoscope reprocessing and water purification products. Warranty reserves are included in accrued expenses in the consolidated balance sheets.
10. | Financing Arrangements |
On March 4, 2014, we entered into a Third Amended and Restated Credit Agreement (the “2014 Credit Agreement”). The 2014 Credit Agreement includes a five-year $250,000 senior secured revolving facility with sublimits of up to $100,000 for borrowings in foreign currencies, $30,000 for letters of credit and $10,000 for swing line loans (the “2014 Revolving Credit Facility”). Subject to the satisfaction of certain conditions precedent including the consent of the lenders, the Company may from time to time increase the 2014 Revolving Credit Facility by an aggregate amount not to exceed $100,000. The 2014 Credit Agreement expires on March 4, 2019. Additionally, subject to certain restrictions and conditions (i) any of our domestic or foreign subsidiaries may become borrowers and (ii) borrowings may occur in multi-currencies.
Borrowings under the 2014 Credit Agreement bear interest at rates ranging from 0.25% to 1.25% above the lender’s base rate, or at rates ranging from 1.25% to 2.25% above the London Interbank Offered Rate (“LIBOR”), depending upon the Company’s “Consolidated Leverage Ratio,” which is defined as the consolidated ratio of total funded debt to earnings before interest, taxes, depreciation and amortization, and as further adjusted under the terms of the 2014 Credit Agreement (“Consolidated EBITDA”). At July 31, 2017, the lender’s base rate was 4.00% and the LIBOR rates ranged from 1.22% to 1.31%. The margins applicable to our outstanding borrowings were 0.50% above the lender’s base rate or 1.50% above LIBOR. All of our outstanding borrowings were under LIBOR contracts at July 31, 2017. The 2014 Credit Agreement also provides for fees on the unused portion of our facility at rates ranging from 0.20% to 0.40%, depending upon our Consolidated Leverage Ratio, which was 0.20% at July 31, 2017.
The 2014 Credit Agreement contains affirmative and negative covenants reasonably customary for similar credit facilities and is secured by (i) substantially all assets of Cantel and its U.S.-based subsidiaries, (ii) a pledge by Cantel of all of the outstanding shares of its U.S.-based subsidiaries and 65% of the outstanding shares of certain of Cantel’s foreign-based subsidiaries and (iii) a guaranty by Cantel’s domestic subsidiaries. We are in compliance with all financial and other covenants under the 2014 Credit Agreement.
As of July 31, 2017, we had $126,000 of outstanding borrowings under the 2014 Credit Agreement. Subsequent to July 31, 2017, we borrowed $61,300 to fund the purchase price and transaction costs of the BHT Group acquisition.
Debt issuance costs associated with our credit facilities are capitalized and amortized to interest expense over the term of the credit facilities. As of July 31, 2017 and 2016, such debt issuance costs, net of related amortization, were included in other assets, and amounted to $580 and $946, respectively.
11. | Income Taxes |
The consolidated effective tax rate was 32.8%, 36.2% and 37.1% for fiscal 2017, 2016 and 2015, respectively, and reflects income tax expense for our U.S. and international operations at their respective statutory rates.
(dollar amounts in thousands except share and per share data or as otherwise specified) 58
Cantel Medical Corp. 2017 Annual Report on Form 10-K
The provision for income taxes consists of the following:
Year Ended July 31, | |||||||||||||||||||||||
2017 | 2016 | 2015 | |||||||||||||||||||||
Current | Deferred | Current | Deferred | Current | Deferred | ||||||||||||||||||
United States: | |||||||||||||||||||||||
Federal | $ | 28,900 | $ | 2,020 | $ | 29,392 | $ | (216 | ) | $ | 24,602 | $ | (425 | ) | |||||||||
State | 4,352 | 261 | 4,433 | (153 | ) | 3,920 | (218 | ) | |||||||||||||||
International | 1,545 | (2,223 | ) | 1,863 | (1,341 | ) | 1,165 | (806 | ) | ||||||||||||||
Total | $ | 34,797 | $ | 58 | $ | 35,688 | $ | (1,710 | ) | $ | 29,687 | $ | (1,449 | ) | |||||||||
The geographic components of income before income taxes are as follows:
Year Ended July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
United States | $ | 108,329 | $ | 92,744 | $ | 73,645 | |||||
International | (2,096 | ) | 1,187 | 2,546 | |||||||
Total | $ | 106,233 | $ | 93,931 | $ | 76,191 | |||||
The consolidated effective income tax rate differed from the U.S. statutory tax rate of 35.0% in fiscal 2017, 2016 and 2015 due to the following:
Year Ended July 31, | ||||||||
2017 | 2016 | 2015 | ||||||
Expected statutory tax | 35.0 | % | 35.0 | % | 35.0 | % | ||
Differential attributable to: | ||||||||
Foreign operations | — | % | 0.6 | % | 1.2 | % | ||
State and local taxes | 3.9 | % | 3.2 | % | 3.4 | % | ||
Domestic production deduction | (2.7 | )% | (2.3 | )% | (2.4 | )% | ||
Acquisition related items, net | 0.1 | % | — | % | (1.6 | )% | ||
Loss on sale of business | — | % | — | % | 1.1 | % | ||
R&E tax credit | (1.4 | )% | (1.1 | )% | (0.5 | )% | ||
Change in foreign tax rates | — | % | (0.4 | )% | — | % | ||
Excess tax benefits | (2.2 | )% | — | % | — | % | ||
Other | 0.1 | % | 1.2 | % | 0.9 | % | ||
Consolidated effective tax rate | 32.8 | % | 36.2 | % | 37.1 | % | ||
As a result of the adoption of ASU 2016-09 on August 1, 2016, we no longer record excess tax benefits as an adjustment to additional paid-in-capital, but record such excess tax benefits on a prospective basis as a reduction of income tax expense, which amounted to $2,241 for fiscal 2017.
(dollar amounts in thousands except share and per share data or as otherwise specified) 59
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Deferred income tax assets and liabilities are comprised of the following:
July 31, | |||||||
2017 | 2016 | ||||||
Deferred tax assets: | |||||||
Accrued expenses | $ | 6,308 | $ | 5,140 | |||
Inventories | 4,655 | 2,990 | |||||
Accounts receivable | 729 | 793 | |||||
Other long-term liabilities | 180 | 252 | |||||
Stock-based compensation | 3,402 | 3,665 | |||||
Capital investment | 545 | 546 | |||||
Foreign NOLs | 6,490 | 5,154 | |||||
Subtotal | 22,309 | 18,540 | |||||
Valuation allowance | (2,984 | ) | (2,334 | ) | |||
19,325 | 16,206 | ||||||
Deferred tax liabilities: | |||||||
Property and equipment | (9,957 | ) | (8,089 | ) | |||
Intangible assets | (20,107 | ) | (19,818 | ) | |||
Goodwill | (13,975 | ) | (11,878 | ) | |||
(44,039 | ) | (39,785 | ) | ||||
Net deferred tax liabilities - noncurrent | $ | (24,714 | ) | $ | (23,579 | ) | |
For foreign tax reporting purposes, our Net Operating Losses (“NOLs”) at July 31, 2017 are $6,490 and originated primarily from foreign acquisitions. Most of these NOLs do not expire and are fully available for utilization against future profits in certain non-U.S. tax jurisdictions. However, we have recorded a valuation allowance of $2,984 for these foreign NOLs, which are primarily associated with certain early-stage foreign operations as well as the exit of the Jet Prep business more fully described in Note 8, "Intangibles and Goodwill." We believe it is more likely than not that we will be unable to utilize these NOLs.
During fiscal 2017 and 2016, no dividends were repatriated from our foreign subsidiaries. All of the undistributed earnings of our foreign subsidiaries are considered to be indefinitely reinvested at July 31, 2017. Accordingly, deferred taxes are not provided on undistributed earnings of foreign subsidiaries that are indefinitely reinvested. At July 31, 2017, the cumulative amount of such undistributed earnings indefinitely reinvested outside the United States was approximately $44,509. Determining the tax liability that would arise if these earnings were remitted is not practical.
We record liabilities for an unrecognized tax benefit when a tax benefit for an uncertain tax position is taken or expected to be taken on a tax return, but is not recognized in our consolidated financial statements because it does not meet the more-likely-than-not recognition threshold that the uncertain tax position would be sustained upon examination by the applicable taxing authority. The tax benefits recognized in the financial statements from such a position are measured based on the largest benefit that has a greater than fifty percent likelihood of being realized upon settlement with the tax authorities. Any adjustments upon resolution of income tax uncertainties are recognized in our results of operations. Our policy is to record potential interest and penalties related to income tax positions in interest expense and general and administrative expense, respectively, in our consolidated financial statements. However, such amounts have been relatively insignificant due to the nominal amount of our unrecognized tax benefits relating to uncertain tax positions.
The Company concluded an audit by the Internal Revenue Service (“IRS”) for fiscal years 2015, 2013 and 2012. With respect to state or foreign income tax examinations, the Company is generally no longer subject to examinations for fiscal years ended prior to July 31, 2009.
(dollar amounts in thousands except share and per share data or as otherwise specified) 60
Cantel Medical Corp. 2017 Annual Report on Form 10-K
12. Commitments and Contingencies
Operating Leases
We have several non-cancelable operating leases, primarily for our corporate headquarters, certain of our leased manufacturing facilities, warehouses, office space and equipment. Total rental expense related to our operating leases was $7,715, $6,675 and $6,025 for fiscal 2017, 2016 and 2015, respectively.
As of July 31, 2017, future minimum lease payments under non-cancelable operating leases (with initial or remaining lease terms in excess of one year) for the periods set forth below were as follows:
Fiscal year ending: | Total | ||
2018 | $ | 6,522 | |
2019 | 5,278 | ||
2020 | 3,779 | ||
2021 | 2,719 | ||
2022 | 1,231 | ||
Thereafter | 2,454 | ||
Total | $ | 21,983 | |
Contingent Consideration and Assumed Contingent Liability
We have $1,138 recorded as of July 31, 2017 related to the Jet Prep acquisition, which is for the estimated fair value of an assumed contingent obligation payable to the Israeli Government, as further described in Note 8, "Intangibles and Goodwill," which will be payable based on future sales. We are currently working with the Israeli Government to forgive any future amounts payable due to our decision to exit the Jet Prep business and we expect a decision from the Israeli government in the first half of fiscal 2018. Additionally, in connection with the PuriCore plc acquisition in fiscal 2014, we assumed a contingent guaranteed obligation to reimburse an endoscope service company for endoscope repair costs it incurs when servicing its customers’ endoscopes that are damaged by one of PuriCore’s discontinued endoscope reprocessing machine models. During fiscal 2017, we ended the agreement with the endoscope service company and decreased the remaining liability of $283 through our consolidated statements of income.
Legal Proceedings
In the normal course of business, we are subject to pending and threatened legal actions. It is our policy to accrue for amounts related to these legal matters if it is probable that a liability has been incurred and an amount of anticipated exposure can be reasonably estimated. We do not believe that any of these pending claims or legal actions will have a material effect on our business, financial condition, results of operations or cash flows.
(dollar amounts in thousands except share and per share data or as otherwise specified) 61
Cantel Medical Corp. 2017 Annual Report on Form 10-K
13. | Accumulated Other Comprehensive Loss |
The components and changes in accumulated other comprehensive loss for fiscal 2017, 2016 and 2015 were as follows:
Foreign Currency Translation Adjustments | |||
Balance, July 31, 2014 | $ | 9,552 | |
Other comprehensive loss before reclassification adjustments | (7,064 | ) | |
Reclassification adjustment to loss on sale of business for foreign currency translation gain included in net income during the year | (1,264 | ) | |
Balance, July 31, 2015 | 1,224 | ||
Other comprehensive loss | (13,019 | ) | |
Balance, July 31, 2016 | (11,795 | ) | |
Other comprehensive income | 1,895 | ||
Balance, July 31, 2017 | $ | (9,900 | ) |
14. | Earnings Per Common Share |
Basic EPS is computed based upon the weighted average number of common shares outstanding for the year. Diluted EPS is computed based upon the weighted average number of common shares outstanding for the year plus the dilutive effect of common stock equivalents using the treasury stock method and the average market price of our common stock for the year. We include participating securities (unvested share-based payment awards that contain non-forfeitable rights to dividends or dividend equivalents) in the computation of EPS pursuant to the two-class method. Our participating securities consist solely of unvested restricted stock awards, which have contractual participation rights equivalent to those of stockholders of unrestricted common stock. The two-class method of computing earnings per share is an allocation method that calculates earnings per share for common stock and participating securities.
The following table sets forth the computation of basic and diluted EPS available to stockholders of common stock (excluding participating securities):
Year Ended July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Numerator for basic and diluted earnings per share: | |||||||||||
Net income | $ | 71,378 | $ | 59,953 | $ | 47,953 | |||||
Less income allocated to participating securities | (431 | ) | (488 | ) | (433 | ) | |||||
Net income available to common shareholders | $ | 70,947 | $ | 59,465 | $ | 47,520 | |||||
Denominator for basic and diluted earnings per share, as adjusted for participating securities: | |||||||||||
Denominator for basic earnings per share - weighted average number of shares outstanding attributable to common stock | 41,468,487 | 41,344,013 | 41,139,467 | ||||||||
Dilutive effect of stock options using the treasury stock method and the average market price for the year | 74,278 | 46,181 | 63,133 | ||||||||
Denominator for diluted earnings per share - weighted average number of shares and common stock equivalents attributable to common stock | 41,542,765 | 41,390,194 | 41,202,600 | ||||||||
Earnings per share attributable to common stock: | |||||||||||
Basic earnings per share | $ | 1.71 | $ | 1.44 | $ | 1.16 | |||||
Diluted earnings per share | $ | 1.71 | $ | 1.44 | $ | 1.15 | |||||
Stock options excluded from weighted average dilutive common shares outstanding because their inclusion would have been antidilutive | — | — | — | ||||||||
(dollar amounts in thousands except share and per share data or as otherwise specified) 62
Cantel Medical Corp. 2017 Annual Report on Form 10-K
A reconciliation of weighted average number of shares and common stock equivalents attributable to common stock, as determined above, to the Company’s total weighted average number of shares and common stock equivalents, including participating securities, is set forth in the following table:
Year Ended July 31, | ||||||||
2017 | 2016 | 2015 | ||||||
Denominator for diluted earnings per share - weighted average number of shares and common stock equivalents attributable to common stock | 41,542,765 | 41,390,194 | 41,202,600 | |||||
Participating securities | 254,727 | 340,363 | 378,706 | |||||
Total weighted average number of shares and common stock equivalents attributable to both common stock and participating securities | 41,797,492 | 41,730,557 | 41,581,306 | |||||
15. | Stock-Based Compensation |
2016 Equity Incentive Plan
On January 7, 2016, the Company terminated the Cantel Medical Corp. 2006 Equity Incentive Plan (the “2006 Plan”) and adopted the Cantel Medical Corp. 2016 Equity Incentive Plan (the “2016 Plan”). As a result, no further options or awards will be granted under the Cantel Medical Corp. 2006 Equity Incentive Plan.
The 2016 Plan provides for the granting of stock options, stock appreciation rights (SARs), restricted stock awards, restricted stock units (RSUs) and performance-based awards to our employees, independent contractors and consultants. It also provides the flexibility to grant equity-based awards to our non-employee directors. The 2016 Plan does not permit the granting of discounted options or discounted stock appreciation rights.
The maximum number of shares as to which equity awards may be granted under the 2016 Plan is 1,200,000 shares. The 2016 Plan will terminate on the date of our annual meeting of stockholders following the close of our fiscal year ending in 2025, unless terminated earlier by the Board of Directors. Stock awards under this plan:
• | will be granted at the closing market price at the time of the grant, |
• | will include terms which may not exceed ten years, subject to certain exceptions set forth in the Plan, and |
• | may be granted in the form of Restricted Stock and Restricted Stock Units, Performance Awards, or Dividends. |
Stock awards outstanding under this plan are subject to risk of forfeiture solely due to an employment length-of-service restriction, with such restriction lapsing as to one-third of the shares of each of the first three anniversaries of the grant date subject to being employed by the Company through such vesting date. At July 31, 2017, 113,926 unvested restricted stock shares were outstanding under the 2016 plan. No options were outstanding under the 2016 plan. At July 31, 2017, 1,086,911 shares are collectively available pursuant to restricted stock and other stock awards and stock options and stock appreciation rights.
2006 Equity Incentive Plan
A total of 5,591,000 shares of common stock, of which 2,700,000 shares were authorized for issuance pursuant to stock options and stock appreciation rights and 2,891,000 shares were authorized for issuance pursuant to restricted stock and other stock awards under the 2006 Plan, which was terminated on January 7, 2016 in conjunction with the adoption of the 2016 Plan.
Restricted stock shares outstanding under this plan are subject to risk of forfeiture solely due to an employment length-of-service restriction, with such restriction lapsing as to one-third of the shares on each of the first three anniversaries of the grant date subject to being employed by the Company through such vesting date. At July 31, 2017, options to purchase 122,500 shares of common stock were outstanding, and 108,372 unvested restricted stock shares were outstanding under the 2006 Plan. No additional awards will be granted under this plan.
(dollar amounts in thousands except share and per share data or as otherwise specified) 63
Cantel Medical Corp. 2017 Annual Report on Form 10-K
The following table shows the income statement components of stock-based compensation expense recognized in the consolidated statements of income:
Year Ended July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Cost of sales | $ | 371 | $ | 438 | $ | 270 | |||||
Operating expenses: | |||||||||||
Selling | 1,582 | 929 | 608 | ||||||||
General and administrative | 6,774 | 6,881 | 4,897 | ||||||||
Research and development | 117 | 113 | 92 | ||||||||
Total operating expenses | 8,473 | 7,923 | 5,597 | ||||||||
Stock-based compensation before income taxes | $ | 8,844 | $ | 8,361 | $ | 5,867 | |||||
Our stock options and time-based stock awards are subject to graded vesting in which portions of the awards vest at different times during the vesting period. We recognize compensation expense for awards subject to graded vesting using the straight-line basis over the vesting period.
In October 2016, we granted for the first time to certain employees both equity awards with performance conditions and equity awards with market conditions. The actual number of equity awards earned and eligible to vest will be determined based on the level of achievement against budgeted revenue and a defined gross profit percentage, with respect to the awards with performance conditions, and the Company’s 3-year relative total stockholder return performance as measured against the S&P Healthcare Equipment Index, with respect to the awards with market conditions. The maximum share attainment of these awards are 200% of the initial granted shares. We recognize compensation expense for the awards with performance conditions using the accelerated attribution method over the requisite service period for each separately vesting portion of the award when it is probable that the performance condition will be achieved. We record expense for the awards that are subject to market conditions ratably over the vesting period regardless of whether the market condition is satisfied.
At July 31, 2017, total unrecognized stock-based compensation expense, before income taxes, related to total nonvested stock options and restricted stock awards was $9,741 with a remaining weighted average period of 17 months over which such expense is expected to be recognized. The majority of our nonvested awards relate to restricted stock awards. As a result of the adoption of ASU 2016-09 on August 1, 2016, we have elected to account for forfeitures as they occur, rather than estimate expected forfeitures over the vesting period.
We determine the fair value of each time-based stock award and performance-based stock award by using the closing market price of our common stock on the date of grant. We determine the fair value of each stock award with market conditions using the Monte Carlo simulation on the date of grant using the following assumptions:
Year Ended | |||
July 31, 2017 | |||
Volatility of common stock | 27.75 | % | |
Average volatility of peer companies | 32.98 | % | |
Average correlation coefficient of peer companies | 35.35 | % | |
Risk-free interest rate | 0.96 | % | |
(dollar amounts in thousands except share and per share data or as otherwise specified) 64
Cantel Medical Corp. 2017 Annual Report on Form 10-K
A summary of nonvested stock award activity for the year ended July 31, 2017 follows:
Number of Time-based Shares | Number of Performance-based Shares | Number of Market-based Shares | Number of Total Shares | Weighted Average Fair Value | ||||||||||||
July 31, 2014 | 525,842 | — | — | 525,842 | $ | 22.25 | ||||||||||
Granted | 144,278 | — | — | 144,278 | $ | 39.77 | ||||||||||
Vested(1) | (313,797 | ) | — | — | (313,797 | ) | $ | 18.62 | ||||||||
Forfeited | (12,804 | ) | — | — | (12,804 | ) | $ | 26.20 | ||||||||
July 31, 2015 | 343,519 | — | — | 343,519 | $ | 32.77 | ||||||||||
Granted | 175,700 | — | — | 175,700 | $ | 55.40 | ||||||||||
Vested(1) | (183,045 | ) | — | — | (183,045 | ) | $ | 30.06 | ||||||||
Forfeited | (4,807 | ) | — | — | (4,807 | ) | $ | 45.06 | ||||||||
July 31, 2016 | 331,367 | — | — | 331,367 | $ | 46.09 | ||||||||||
Granted | 86,305 | 16,960 | 9,800 | 113,065 | $ | 81.77 | ||||||||||
Vested(1) | (214,932 | ) | (725 | ) | (555 | ) | (216,212 | ) | $ | 43.62 | ||||||
Forfeited | (5,922 | ) | — | — | (5,922 | ) | $ | 59.40 | ||||||||
July 31, 2017 | 196,818 | 16,235 | 9,245 | 222,298 | $ | 66.28 | ||||||||||
_______________________________________________
(1) | The aggregate fair value of all nonvested stock awards which vested was approximately $9,431, $5,503 and $5,844 in fiscal 2017, 2016 and 2015, respectively. |
There were no options granted during the fiscal year ended July 31, 2017. The fair value of each option grant in the prior years was estimated on the date of grant using the Black-Scholes option valuation model with the following assumptions:
Year Ended | Year Ended | |||||
July 31, 2016 | July 31, 2015 | |||||
Dividend yield | 0.22 | % | 0.25 | % | ||
Expected volatility(1) | 55.90 | % | 33.90 | % | ||
Risk-free interest rate(2) | 1.41 | % | 1.55 | % | ||
Expected lives (in years)(3) | 5.00 | 5.00 | ||||
________________________________________________
(1) | Volatility was based on historical closing prices of our common stock. |
(2) | The U.S. Treasury rate based on the expected life at the date of grant. |
(3) | Based on historical exercise behavior. |
A summary of stock option activity for the year ended July 31, 2017 follows:
Number of shares | Weighted Average Exercise Price | Weighted Average Contractual Life Remaining (Years) | Aggregate Intrinsic Value | |||||||||
Outstanding at July 31, 2016 | 122,500 | $ | 29.36 | |||||||||
Granted | — | — | ||||||||||
Exercised | — | — | ||||||||||
Outstanding at July 31, 2017 | 122,500 | $ | 29.36 | 1.27 | $ | 5,493 | ||||||
Exercisable at July 31, 2017 | 104,167 | $ | 26.28 | 1.02 | $ | 4,992 | ||||||
In fiscal 2017, 2016 and 2015, 23,333, 35,834 and 27,500, respectively, options vested, with an aggregate fair value of approximately $349, $344 and $248, respectively. There were no option grants and no options were exercised during fiscal 2017. The weighted average fair value of options granted was $26.49 and $11.54 in fiscal 2016 and 2015, respectively. At July 31, 2017, 2016 and 2015, there were 122,500, 122,500 and 107,500, respectively, outstanding options with an aggregate fair value of $5,493, $4,605 and $3,133, respectively. As of July 31, 2017 and 2016, all of the outstanding options had vested or were expected to vest in future periods.
(dollar amounts in thousands except share and per share data or as otherwise specified) 65
Cantel Medical Corp. 2017 Annual Report on Form 10-K
The Company does not currently have a publicly announced stock repurchase program. All of the shares purchased during fiscal 2017 and 2016 represent shares surrendered to the Company relating to cashless exercises of stock options and to pay employee withholding taxes due upon the vesting of restricted stock or the exercise of stock options. In fiscal 2017 and 2016, such purchases amounted to 89,607 and 67,038 shares at a total average price per share of $77.12 and $55.68, respectively.
Upon exercise of stock options or grant of stock awards, we typically issue new shares of our common stock as opposed to using treasury shares. Additionally, all options were considered to be deductible for tax purposes in the valuation model. Such non-qualified options were tax-effected using the Company’s estimated U.S. effective tax rate at the time of grant. All of our stock options and restricted stock awards are expected to be deductible for tax purposes, except for certain stock awards granted to employees residing outside of the United States, and were tax-effected using the Company’s estimated U.S. effective tax rate at the time of grant.
Excess tax benefits arise when the ultimate tax effect of the deduction for tax purposes is greater than the income tax benefit on stock-based compensation described above. Prior to the adoption of ASU 2016-09 on August 1, 2016, the differences noted above between actual tax deductions and the previously recorded deferred income tax assets were recorded as additional paid-in capital. For the year ended July 31, 2016, income tax deductions of $3,059 were generated, of which $1,880 were recorded as a reduction in income tax expense over the equity awards’ vesting period and the remaining excess tax benefits of $1,179 were recorded as an increase to additional paid-in capital. As a result of the adoption of ASU 2016-09, we no longer record excess tax benefits as an adjustment to additional paid-in capital, but record such excess tax benefits prospectively as a reduction of income tax expense. For the year ended July 31, 2017, income tax deductions of $5,592 were generated, of which $3,351 were previously recorded as a reduction in income tax expense over the equity awards’ vesting period and the remaining excess tax benefit of $2,241 was recorded as a reduction in income tax expense.
16. | Retirement Plans |
We have 401(k) Savings and Retirement Plans for the benefit of eligible U.S. employees. Additionally, our Canadian and certain European subsidiaries maintain profit sharing plans for the benefit of eligible employees. Contributions by the Company are both discretionary and non-discretionary and are limited in any year to the amount allowable by government tax authorities.
Aggregate employer contributions recognized under these plans were $3,863, $3,406 and $2,541 for fiscal 2017, 2016 and 2015, respectively.
17. | Information as to Reportable Segments and Foreign and Domestic Operations |
In accordance with FASB ASC Topic 280, “Segment Reporting,” (“ASC 280”), we have determined our reportable business segments based upon an assessment of product types, organizational structure, customers and internally prepared financial statements. The primary factors used by us in analyzing segment performance are net sales and operating income.
None of our customers accounted for 10% or more of our consolidated net sales during fiscal 2017, 2016 and 2015.
The Company’s reportable segments are as follows:
Endoscopy: designs, develops, manufactures, sells and installs a comprehensive offering of products and services comprising a complete circle of infection prevention solutions. Our products include endoscope reprocessing and endoscopy procedure products.
Water Purification and Filtration: designs, develops, manufactures, sells, and installs water purification systems for medical, pharmaceutical and other bacteria controlled applications. We also provide filtration/separation and disinfectant technologies to the medical and life science markets through a worldwide distributor network.
Two customers, who collectively accounted for approximately 50.2% of our Water Purification and Filtration segment net sales in fiscal 2017.
Healthcare Disposables: designs, manufactures, sells, supplies and distributes a broad selection of infection prevention healthcare products, the majority of which are single-use products used by dental practitioners.
Three customers, who collectively accounted for approximately 43.4% of our Healthcare Disposables segment net sales in fiscal 2017.
(dollar amounts in thousands except share and per share data or as otherwise specified) 66
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Dialysis: designs, develops, manufactures, sells and services reprocessing systems and sterilants for dialyzers (a device serving as an artificial kidney), as well as dialysate concentrates and supplies utilized for renal dialysis.
Two customers collectively accounted for approximately 44.2% of our Dialysis segment net sales in fiscal 2017.
Other: On April 7, 2015, we completed the sale of our Specialty Packaging business and recorded a $2,206 loss associated with this divestiture, which is included in our consolidated statements of income in fiscal 2015. The operating results of the Specialty Packaging business through April 7, 2015 are reported in Other in the tables below. The results were not significant in relation to our overall consolidated operating results and did not have a major effect on our operations and financial results, and accordingly, has not been classified as a discontinued operation for any of the periods presented.
Information as to reportable segments is summarized below:
Year Ended July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Net sales: | |||||||||||
Endoscopy | $ | 398,773 | $ | 341,752 | $ | 248,654 | |||||
Water Purification and Filtration | 196,446 | 177,669 | 173,834 | ||||||||
Healthcare Disposables | 144,457 | 112,584 | 106,920 | ||||||||
Dialysis | 30,481 | 32,750 | 31,240 | ||||||||
Other | — | — | 4,356 | ||||||||
Total | $ | 770,157 | $ | 664,755 | $ | 565,004 | |||||
Year Ended July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Operating income: | |||||||||||
Endoscopy | $ | 73,440 | $ | 61,021 | $ | 40,863 | |||||
Water Purification and Filtration | 33,159 | 30,620 | 30,606 | ||||||||
Healthcare Disposables | 28,000 | 24,486 | 19,904 | ||||||||
Dialysis | 8,154 | 7,907 | 6,749 | ||||||||
Other | — | — | 1,118 | ||||||||
142,753 | 124,034 | 99,240 | |||||||||
General corporate expenses | 32,343 | 26,783 | 18,479 | ||||||||
Income from operations | 110,410 | 97,251 | 80,761 | ||||||||
Interest expense, net | 4,303 | 3,320 | 2,364 | ||||||||
Other income | (126 | ) | — | — | |||||||
Loss on sale of business | — | — | 2,206 | ||||||||
Income before income taxes | $ | 106,233 | $ | 93,931 | $ | 76,191 | |||||
Year Ended July, 31 | |||||||||||
2017 | 2016 | 2015 | |||||||||
Identifiable assets: | |||||||||||
Endoscopy | $ | 368,820 | $ | 347,107 | $ | 238,799 | |||||
Water Purification and Filtration | 147,477 | 137,731 | 138,069 | ||||||||
Healthcare Disposables | 208,328 | 157,918 | 145,391 | ||||||||
Dialysis | 17,211 | 20,147 | 26,452 | ||||||||
General corporate, including cash and cash equivalents | 44,537 | 31,629 | 35,320 | ||||||||
Total | $ | 786,373 | $ | 694,532 | $ | 584,031 | |||||
(dollar amounts in thousands except share and per share data or as otherwise specified) 67
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Year Ended July, 31 | |||||||||||
2017 | 2016 | 2015 | |||||||||
Capital expenditures: | |||||||||||
Endoscopy | $ | 13,816 | $ | 11,299 | $ | 7,042 | |||||
Water Purification and Filtration | 3,689 | 3,376 | 2,984 | ||||||||
Healthcare Disposables | 2,492 | 2,606 | 1,587 | ||||||||
Dialysis | 1,296 | 667 | 894 | ||||||||
Other | — | — | 19 | ||||||||
General corporate | 5,772 | 941 | 234 | ||||||||
Total | $ | 27,065 | 18,889 | 12,760 | |||||||
Year Ended July, 31 | |||||||||||
2017 | 2016 | 2015 | |||||||||
Depreciation and amortization: | |||||||||||
Endoscopy | $ | 18,245 | $ | 14,333 | $ | 10,729 | |||||
Water Purification and Filtration | 5,706 | 5,441 | 5,257 | ||||||||
Healthcare Disposables | 8,556 | 4,361 | 6,354 | ||||||||
Dialysis | 427 | 681 | 1,382 | ||||||||
Other | — | — | 78 | ||||||||
General corporate | 518 | 268 | 157 | ||||||||
Total | $ | 33,452 | $ | 25,084 | $ | 23,957 | |||||
Information as to geographic areas (including net sales which represent the geographic area from which the Company derives its net sales from external customers) is summarized below:
Year Ended July, 31 | |||||||||||
2017 | 2016 | 2015 | |||||||||
Net sales: | |||||||||||
United States | $ | 599,657 | $ | 515,055 | $ | 447,848 | |||||
Europe/Africa/Middle East | 95,753 | 88,355 | 62,193 | ||||||||
Asia/Pacific | 40,964 | 33,374 | 28,529 | ||||||||
Canada | 26,648 | 20,975 | 19,306 | ||||||||
Latin America/South America | 7,135 | 6,996 | 7,128 | ||||||||
Total | $ | 770,157 | $ | 664,755 | $ | 565,004 | |||||
July 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Total long-lived assets: | |||||||||||
United States | $ | 74,401 | $ | 62,820 | $ | 57,080 | |||||
Europe/Africa/Middle East | 16,209 | 14,863 | 9,122 | ||||||||
Asia/Pacific | 1,381 | 1,607 | 1,081 | ||||||||
Canada | 1,054 | 463 | 600 | ||||||||
Total | 93,045 | 79,753 | 67,883 | ||||||||
Goodwill and intangible assets, net | 435,957 | 392,037 | 327,787 | ||||||||
Total | $ | 529,002 | $ | 471,790 | $ | 395,670 | |||||
(dollar amounts in thousands except share and per share data or as otherwise specified) 68
Cantel Medical Corp. 2017 Annual Report on Form 10-K
18. | Quarterly Results of Operations (unaudited) |
The following is a summary of the quarterly results of operations for the years ended July 31, 2017 and 2016:
2017 | First Quarter | Second Quarter | Third Quarter | Fourth Quarter | |||||||||||
Net sales | $ | 187,725 | $ | 184,817 | $ | 192,113 | $ | 205,502 | |||||||
Cost of sales | 98,218 | 96,340 | 100,665 | 107,774 | |||||||||||
Gross profit | 89,507 | 88,477 | 91,448 | 97,728 | |||||||||||
Gross profit percentage | 47.7 | % | 47.9 | % | 47.6 | % | 47.6 | % | |||||||
Net income | $ | 18,800 | $ | 18,070 | $ | 17,511 | $ | 16,997 | |||||||
Earnings per common share: | |||||||||||||||
Basic | $ | 0.45 | $ | 0.43 | $ | 0.42 | $ | 0.41 | |||||||
Diluted | $ | 0.45 | $ | 0.43 | $ | 0.42 | $ | 0.41 | |||||||
2016 | First Quarter | Second Quarter | Third Quarter | Fourth Quarter | |||||||||||
Net sales | $ | 153,779 | $ | 158,271 | $ | 173,703 | $ | 179,002 | |||||||
Cost of sales | 82,581 | 85,934 | 93,382 | 93,672 | |||||||||||
Gross profit | 71,198 | 72,337 | 80,321 | 85,330 | |||||||||||
Gross profit percentage | 46.3 | % | 45.7 | % | 46.2 | % | 47.7 | % | |||||||
Net income | $ | 14,254 | $ | 15,389 | $ | 14,019 | $ | 16,291 | |||||||
Earnings per common share: | |||||||||||||||
Basic | $ | 0.34 | $ | 0.37 | $ | 0.34 | $ | 0.39 | |||||||
Diluted | $ | 0.34 | $ | 0.37 | $ | 0.34 | $ | 0.39 | |||||||
(dollar amounts in thousands except share and per share data or as otherwise specified) 69
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Schedule II - Valuation and Qualifying Accounts
Balance at Beginning of Period | Additions | Deductions | Translation Adjustments | Balance at End of Period | |||||||||||||||
Allowance for doubtful accounts: | |||||||||||||||||||
Year ended July 31, 2017 | $ | 1,850 | $ | 998 | $ | (1,056 | ) | $ | 16 | $ | 1,808 | ||||||||
Year ended July 31, 2016 | $ | 2,092 | $ | 15 | $ | (223 | ) | $ | (34 | ) | $ | 1,850 | |||||||
Year ended July 31, 2015 | $ | 1,874 | $ | 464 | $ | (227 | ) | $ | (19 | ) | $ | 2,092 | |||||||
Reserve for excess and obsolete inventory: | |||||||||||||||||||
Year ended July 31, 2017 | $ | 5,390 | $ | 5,016 | $ | (1,580 | ) | $ | 27 | $ | 8,853 | ||||||||
Year ended July 31, 2016 | $ | 3,895 | $ | 3,182 | $ | (1,569 | ) | $ | (118 | ) | $ | 5,390 | |||||||
Year ended July 31, 2015 | $ | 4,419 | $ | 1,494 | $ | (1,796 | ) | $ | (222 | ) | $ | 3,895 | |||||||
Deferred tax asset valuation allowance: | |||||||||||||||||||
Year ended July 31, 2017 | $ | 2,334 | $ | 615 | $ | — | $ | 35 | $ | 2,984 | |||||||||
Year ended July 31, 2016 | $ | 2,037 | $ | 929 | $ | (712 | ) | $ | 80 | $ | 2,334 | ||||||||
Year ended July 31, 2015 | $ | 3,538 | $ | 1,010 | $ | (2,420 | ) | (1) | $ | (91 | ) | $ | 2,037 | ||||||
________________________________________________
(1) | The amounts primarily include deductions of valuation allowances associated with New Jersey net operating losses. |
(dollar amounts in thousands except share and per share data or as otherwise specified) 70
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure.
Not applicable.
Item 9A. Controls and Procedures.
Under the supervision and with the participation of our Chief Executive Officer and our Chief Financial Officer, we conducted an evaluation of the effectiveness of the design and operation of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) as of July 31, 2017. Based on this evaluation, our Chief Executive Officer and Chief Financial Officer each concluded that the design and operation of these disclosure controls and procedures were, as of the end of the period covered by this report, effective and designed to ensure that material information relating to the Company, including our consolidated subsidiaries, required to be disclosed in our SEC reports is (i) recorded, processed, summarized and reported within the time periods specified by the SEC and (ii) accumulated and communicated to the Company’s management, including the Chief Executive Officer and the Chief Financial Officer, as appropriate to allow timely decisions regarding disclosure.
Management’s Report on Internal Control over Financial Reporting
The management of Cantel Medical Corp. is responsible for establishing and maintaining adequate internal control over financial reporting for the Company. The Company’s internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with U.S. generally accepted accounting principles. The Company’s internal control over financial reporting includes those policies and procedures that:
(i) | pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the Company, |
(ii) | provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the Company are being made only in accordance with authorizations of management and directors of the Company, and |
(iii) | provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the Company’s assets that could have a material effect on the financial statements. |
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of the effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in condition, or that the degree of compliance with the policies and procedures included in such controls may deteriorate.
We, under the supervision and with the participation of our Chief Executive Officer and our Chief Financial Officer, carried out an evaluation of the effectiveness of our internal controls over financial reporting based on the framework and criteria established in “Internal Control — Integrated Framework (2013 framework),” issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”). Based on that evaluation, the Chief Executive Officer and the Chief Financial Officer each concluded that our internal control over financial reporting was effective as of July 31, 2017.
Our independent auditors, Ernst & Young LLP, have issued a report on our internal control over financial reporting, which is included in Part II, Item 8 of this report.
Changes in Internal Control
We have evaluated our internal controls over financial reporting and determined that no changes occurred during the period covered by this report that have materially affected, or are reasonably likely to materially affect, our internal controls over financial reporting, except as described below.
On August 1, 2016 we acquired Accutron, Inc., as more fully described in Note 3 to the consolidated financial statements. The Accutron business is included in our 2017 consolidated financial statements and constituted 7.6% and 10.3% of total assets and net assets, respectively, as of July 31, 2017, and 3.0% and 4.1% of net sales and net income for the year then ended. During the initial transition period following the acquisition, we enhanced our internal control process to ensure that all financial information related to this acquisition was properly reflected in our consolidated financial statements. However, since the Accutron business was acquired on August 1, 2016, a complete integration of the internal controls relating to the acquired businesses was not practical for purposes of inclusion in our evaluation of the effectiveness of our internal controls over financial reporting. We expect that all aspects of the Accutron business will be fully integrated into our existing internal control structure in fiscal 2018.
Item 9B. Other Information.
None.
71
Cantel Medical Corp. 2017 Annual Report on Form 10-K
PART III
Item 10. Directors, Executive Officers and Corporate Governance.
Information required to be disclosed by this Item with respect to our executive officers is incorporated in this Annual Report on Form 10-K by reference from the section entitled “Executive Officers of Cantel” contained in our definitive proxy statement for our 2017 annual meeting of stockholders, which we intend to file within 120 days of the end of our fiscal year.
Information required to be disclosed by this Item with respect to our board of directors is incorporated in this Annual Report on Form 10-K by reference from the section entitled “Election of Directors” contained in our definitive proxy statement for our 2017 annual meeting of stockholders, which we intend to file within 120 days of the end of our fiscal year.
Information required to be disclosed by this Item with respect to the Section 16(a) compliance of our directors and executive officers is incorporated in this Annual Report on Form 10-K by reference from the section entitled “Section 16(a) Beneficial Ownership Reporting Compliance” contained in our definitive proxy statement for our 2017 annual meeting of stockholders, which we intend to file within 120 days of the end of our fiscal year.
Information required to be disclosed by this Item with respect to the audit committee of our board of directors, our audit committee financial expert, and other board of directors and corporate governance matters is incorporated in this Annual Report on Form 10-K by reference from the sections entitled “Board Matters; Committees” and “Corporate Governance Matters” contained in our definitive proxy statement related to our 2017 annual meeting of stockholders, which we intend to file within 120 days of the end of our fiscal year.
We have adopted a Code of Ethics for the Chief Executive Officer, the Chief Financial Officer and other officers and management personnel that is posted on our website, www.cantelmedical.com. We intend to satisfy the disclosure requirement regarding any amendment to, or a waiver of, a provision of the Code of Ethics for the Chief Executive Officer, Chief Financial Officer and other officers and management personnel by posting such information on our website.
Item 11. Executive Compensation.
Information required to be disclosed by this Item is incorporated in this Annual Report on Form 10-K by reference from the sections entitled “Board Matters; Committees,” “Compensation Committee Report” and “Executive Compensation” contained in our definitive proxy statement for our 2017 annual meeting of stockholders, which we intend to file within 120 days of the end of our fiscal year.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.
The following sets forth certain information as of July 31, 2017 with respect to our equity compensation plans under which our securities may be issued:
Number of securities to be issued upon exercise of outstanding options | Weighted-average exercise price of outstanding options | Number of securities remaining available for future issuance under compensation plans (excluding securities reflected in (a)) | |||||||||
Plan Category | (a) | (b) | (c) | ||||||||
Equity compensation plans approved by security holders | 122,500 | $ | 29.36 | 1,086,911 | (1) | ||||||
Equity compensation plans not approved by security holders | — | — | — | ||||||||
Total | 122,500 | $ | 29.36 | 1,086,911 | (1) | ||||||
________________________________________________
(1) | Collectively consists of stock option and SARs awards and restricted stock and performance awards available for grant under the Plan. |
Other Information required to be disclosed by this Item is incorporated in this Annual Report on Form 10-K by reference from the section entitled “Security Ownership of Principal Stockholders and Management” contained in our definitive proxy statement for our 2017 annual meeting of stockholders, which we intend to file within 120 days of the end of our fiscal year.
72
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Item 13. Certain Relationships and Related Transactions and Director Independence.
The information required to be disclosed by this Item is incorporated in this Annual Report on Form 10-K by reference from the sections entitled “Corporate Governance,” “Election of Directors,” and “Board Matters; Committees” contained in our definitive proxy statement for our 2017 annual meeting of stockholders, which we intend to file within 120 days of the end of our fiscal year.
Item 14. Principal Accounting Fees and Services.
The information required to be disclosed by this Item is incorporated in this Annual Report on Form 10-K by reference from the section entitled “Ratification of Appointment of Independent Registered Public Accounting Firm” contained in our definitive proxy statement for our 2017 annual meeting of stockholders, which we intend to file within 120 days of the end of our fiscal year.
73
Cantel Medical Corp. 2017 Annual Report on Form 10-K
PART IV
Item 15. Exhibits, Financial Statement Schedules.
(a) | The following documents are filed as part of this Annual Report on Form 10-K for the fiscal year ended July 31, 2017. |
1. | Consolidated Financial Statements: |
(i) | Report of Independent Registered Public Accounting Firm. |
(ii) | Consolidated Balance Sheets as of July 31, 2017 and 2016. |
(iii) | Consolidated Statements of Income for the years ended July 31, 2017, 2016 and 2015. |
(iv) | Consolidated Statements of Comprehensive Income for the years ended July 31, 2017, 2016 and 2015. |
(v) | Consolidated Statements of Changes in Stockholders’ Equity for the years ended July 31, 2017, 2016 and 2015. |
(vi) | Consolidated Statements of Cash Flows for the years ended July 31, 2017, 2016 and 2015. |
(vii) | Notes to Consolidated Financial Statements. |
2. | Consolidated Financial Statement Schedules: |
(i) | Schedule II - Valuation and Qualifying Accounts for the years ended July 31, 2017, 2016 and 2015. |
All other financial statement schedules are omitted since they are not required, not applicable, or the information has been included in the Consolidated Financial Statements or Notes thereto.
3. | Exhibits: |
3(a) - Registrant’s Restated Certificate of Incorporation, dated July 20, 1978. (Incorporated herein by reference to Exhibit 3(a) to Registrant’s 1981 Annual Report on Form 10-K.)
3(b) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on February 16, 1982. (Incorporated herein by reference to Exhibit 3(b) to Registrant’s 1982 Annual Report on Form 10-K.)
3(c) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on May 4, 1984. (Incorporated herein by reference to Exhibit 3(c) to Registrant’s Quarterly Report on Form 10-Q for the quarter ended April 30, 1984.)
3(d) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on August 19, 1986. (Incorporated herein by reference to Exhibit 3(d) to Registrant’s 1986 Annual Report on Form 10-K.)
3(e) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on December 12, 1986. (Incorporated herein by reference to Exhibit 3(e) to Registrant’s 1987 Annual Report on Form 10-K [the “1987 10-K”].)
3(f) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on April 3, 1987. (Incorporated herein by reference to Exhibit 3(f) to Registrant’s 1987 10-K.)
3(g) - Certificate of Change of Registrant, filed on July 12, 1988. (Incorporated herein by reference to Exhibit 3(g) to Registrant’s 1988 Annual Report on Form 10-K, File No. 001-31337.)
3(h) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on April 17, 1989. (Incorporated herein by reference to Exhibit 3(h) to Registrant’s 1989 Annual Report on Form 10-K, File No. 001-31337.)
3(i) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on May 10, 1999. (Incorporated herein by reference to Exhibit 3(i) to Registrant’s 2000 Annual Report on Form 10-K, File No. 001-31337 [the “2000 10-K”].)
74
Cantel Medical Corp. 2017 Annual Report on Form 10-K
3(j) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on April 5, 2000. (Incorporated herein by reference to Exhibit 3(j) to Registrant’s 2000 10-K.)
3(k) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on September 6, 2001. (Incorporated herein by reference to Exhibit 3(k) to Registrant’s 2001 Annual Report on Form 10-K, File No. 001-31337.)
3(l) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on June 7, 2002. (Incorporated herein by reference to Exhibit 3(l) to Registrant’s 2002 Annual Report on Form 10-K, File No. 001-31337.)
3(m) - Certificate of Amendment of Certificate of Incorporation of Registrant, filed on December 22, 2005. (Incorporated herein by reference to Exhibit 3(m) to Registrant’s 2006 Annual Report on Form 10-K, File No. 001-31337.)
3(n) — Certificate of Amendment of Certificate of Incorporation of Registrant filed on January 14, 2013. (Incorporated herein by reference to Exhibit 3(n) to Registrant’s 2013 Annual Report on Form 10-K, File No. 001-31337.)
3(o)- Registrant’s By-Laws, as amended through November 1, 2013. (Incorporated herein by reference to Exhibit 3.1 to Registrant’s Current Report on Form 8-K filed on November 7, 2013, File No. 001-31337.)
10(a) - 2006 Equity Incentive Plan, as amended. (Incorporated herein by reference to Exhibit 10(a) to Registrant’s Quarterly Report on Form 10-Q for the quarter ended October 31, 2013, File No. 001-31337.)*
10(b) - Form of Stock Option Agreement for option grants to directors and executive officers under Registrant’s 2006 Equity Incentive Plan. (Incorporated herein by reference to Exhibit 10.4 to Registrant’s Current Report on Form 8-K filed on October 27, 2011, File No. 001-31337 [the “October 2011 8-K”].)*
10(c) - Form of Restricted Stock Agreement under Registrant’s 2006 Equity Incentive Plan for grants to executive officers. (Incorporated herein by reference to Exhibit 10.5 to Registrant’s October 2011 8-K.)*
10(d) - Form of Restricted Stock Agreement under Registrant’s 2006 Equity Incentive Plan for grants to directors. (Incorporated herein by reference to Exhibit 10.6 to Registrant’s October 2011 8-K.)*
10(e) - Third Amended and Restated Credit Agreement dated as of March 4, 2014 among Registrant, Bank of America N.A., PNC Bank, National Association, and Wells Fargo Bank, National Association. (Incorporated herein by reference to Exhibit 10.1 to Registrant’s Current Report on Form 8-K filed on March 10, 2014, File No. 001-31337.)
10(f) – Succession Plan Agreement dated as of March 17, 2016 between Registrant and Andrew A. Krakauer. (Incorporated herein by reference to Exhibit 10.1 to Registrant’s Current Report on Form 8-K filed on March 17, 2016, File No. 001-31337.)*
10(g) - Amended and Restated Executive Severance Agreement dated as of August 1, 2016 between Registrant and Jorgen B. Hansen. (Incorporated herein by reference to Exhibit 10.1 to Registrant’s Current Report on Form 8-K filed on August 1, 2016, File No. 001-31337.)*
10(h) - Amended and Restated Executive Severance Agreement dated as of November 17, 2014 between Registrant and Eric W. Nodiff (Incorporated herein by reference to Exhibit 10.3 to Registrant’s Current Report on Form 8-K filed on November 19, 2014, File No. 001-31337.)*
10(i) - Executive Severance Agreement dated as of March 23, 2015 between Registrant and Peter Clifford (Incorporated herein by reference to Exhibit 10.1 to Registrant’s Current Report on Form 8-K filed on March 25, 2015, File No. 001-31337 [the “March 2015 8-K”].)*
10(j) - Confidentiality and Non-Competition Agreement dated as of January 1, 2010 between Registrant and Andrew A. Krakauer (Incorporated herein by reference to Exhibit 10.6 to Registrant’s Current Report on Form 8-K filed on February 12, 2010, File No. 001-31337 [the “February 2010 8-K”].)*
10(k) - Confidentiality and Non-Competition Agreement dated as of November 15, 2012 between Registrant and Jorgen B. Hansen (Incorporated herein by reference to Exhibit 10(m) to Registrant’s Annual Report on Form 10-K for the fiscal year ended July 31, 2012, File No. 001-31337.)*
75
Cantel Medical Corp. 2017 Annual Report on Form 10-K
10(l) - Confidentiality and Non-Competition Agreement dated as of January 1, 2010 between Registrant and Eric W. Nodiff (Incorporated herein by reference to Exhibit 10.9 to Registrant’s February 2010 8-K.)*
10(m) - Confidentiality and Non-Competition Agreement dated as of March 23, 2015 between Registrant and Peter Clifford (Incorporated herein by reference to Exhibit 10.2 to Registrant’s March 2015 8-K.)*
10(n) – Cantel Medical Corp. 2016 Equity Incentive Plan (Incorporated herein by reference to Annex A to Registrant’s Proxy Statement for the 2015 Annual Meeting of Stockholders, filed with the SEC on November 30, 2015, File No. 001-31337.)*
10(o) - Form of Restricted Stock Agreement (Time-Based Grants) under Cantel Medical Corp. 2016 Equity Incentive Plan for grants to executive officers (Incorporated herein by reference to Exhibit 10(r) to Registrant's Annual Report on From 10-K for the fiscal year ended July 31, 2016, File No. 001-31337 [the "2016 10-K"].) *
10(p) - Form of Restricted Stock Agreement (Time-Based Grants) under Cantel Medical Corp. 2016 Equity Incentive Plan for grants to directors (Incorporated herein by reference to Exhibit 10(s) to Registrant's 2016 10-K.)*
10(q) - Form of Restricted Stock Agreement (Performance-Based Grants – Revenue Based) under Cantel Medical Corp. 2016 Equity Incentive Plan for grants to executive officers (Incorporated herein by reference to Exhibit 10(t) to Registrant's 2016 10-K.) *
10(r) - Form of Restricted Stock Agreement (Performance-Based Grants – TSR Based) under Cantel Medical Corp. 2016 Equity Incentive Plan for grants to executive officers (Incorporated herein by reference to Exhibit 10(u) to Registrant's 2016 10-K.)*
10(s) - Form of Restricted Stock Agreement (Time-Based) under Cantel Medical Corp. 2016 Equity Incentive Plan for annual grants to directors (Incorporated herein by reference to Exhibit 10(v) to Registrant's 2016 10-K.)*
Exhibit 21 - Subsidiaries of Registrant.
Exhibit 23 - Consent of Ernst & Young LLP.
Exhibit 31.1 - Certification of Principal Executive Officer.
Exhibit 31.2 - Certification of Principal Financial Officer.
Exhibit 32 - Certification of Chief Executive Officer and Chief Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002.
101 The following materials from Cantel Medical Corp.’s Form 10-K for the fiscal year ended July 31, 2017, formatted in Extensible Business Reporting Language (XBRL): (i) Consolidated Balance Sheets at July 31, 2017 and 2016, (ii) Consolidated Statements of Income for each of the three years in the period ended July 31, 2017, (iii) Consolidated Statements of Comprehensive Income for each of the three years in the period ended July 31, 2017, (iv) Consolidated Statements of Changes in Stockholders’ Equity for each of the three years in the period ended July 31, 2017, (v) Consolidated Statements of Cash Flows for each of the three years in the period ended July 31, 2017 and (vi) Notes to Consolidated Financial Statements.
*Management contract or compensatory plan or arrangement of the Company required to be filed as an exhibit.
Item 16. Form 10-K Summary
None.
76
Cantel Medical Corp. 2017 Annual Report on Form 10-K
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
CANTEL MEDICAL CORP. | |||
Date: September 28, 2017 | By: | /s/ Jorgen B. Hansen | |
Jorgen B. Hansen, President and Chief Executive | |||
Officer (Principal Executive Officer) | |||
By: | /s/ Peter G. Clifford | ||
Peter G. Clifford, Executive Vice President, | |||
Chief Financial Officer | |||
(Principal Financial Officer) | |||
By: | /s/ Brian R. Capone | ||
Brian R. Capone, Vice President, | |||
Chief Accounting Officer | |||
(Principal Accounting Officer) | |||
77
Cantel Medical Corp. 2017 Annual Report on Form 10-K
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated:
/s/ Charles M. Diker | Date: | September 28, 2017 | ||
Charles M. Diker, a Director and Chairman of the Board | ||||
/s/ George L. Fotiades | Date: | September 28, 2017 | ||
George L. Fotiades, a Director | ||||
and Vice Chairman of the Board | ||||
/s/ Alan. R. Batkin | Date: | September 28, 2017 | ||
Alan R. Batkin, a Director | ||||
/s/ Ann E. Berman | Date: | September 28, 2017 | ||
Ann E. Berman, a Director | ||||
/s/ Mark N. Diker | Date: | September 28, 2017 | ||
Mark N. Diker, a Director | ||||
/s/ Anthony B. Evnin | Date: | September 28, 2017 | ||
Anthony B. Evnin, a Director | ||||
/s/ Laura L. Forese | Date: | September 28, 2017 | ||
Laura L. Forese, a Director | ||||
/s/ Jorgen B. Hansen | Date: | September 28, 2017 | ||
Jorgen B. Hansen, a Director, President and CEO | ||||
/s/ Ronnie Myers | Date | September 28, 2017 | ||
Ronnie Myers, a Director | ||||
78
EXHIBIT 21
CANTEL MEDICAL CORP.
Subsidiaries of Registrant
Carsen Group, Inc. | (Incorporated under the laws of Ontario, Canada) |
Medivators Inc. | (Incorporated under the laws of Minnesota) |
Medivators B.V. | (Incorporated under the laws of the Netherlands) |
Cantel Medical Asia/Pacific Pte. Ltd. | (Incorporated under the laws of Singapore) |
Biolab Equipment Ltd. | (Amalgamated under the laws of Canada) |
Mar Cor Purification, Inc. | (Incorporated under the laws of Pennsylvania) |
Crosstex International, Inc. | (Incorporated under the laws of New York) |
SPS Medical Supply Corp. | (Incorporated under the laws of New York) |
Cantel Medical International LLC | (Organized under the laws of Delaware) |
CMCI C.V. | (Incorporated under the laws of the Netherlands) |
Cantel Medical International B.V. | (Incorporated under the laws of the Netherlands) |
Cantel Medical (UK) Limited | (Incorporated under the laws of England and Wales) |
Cantel Medical (Italy) S.r.l. | (Incorporated under the laws of Italy) |
Cantel Medical Devices (China) Co., Ltd. | (Incorporated under the laws of China) |
Cantel (UK) Limited | (Incorporated under the laws of England and Wales) |
Medical Innovations Group Limited | (Incorporated under the laws of England and Wales) |
Accutron, Inc. | (Incorporated under the laws of Arizona) |
Cantel Medical (Hong Kong) Limited | (Incorporated under the laws of Hong Kong) |
Cantel Medical (Malaysia) Sdn. Bhd. | (Incorporated under the laws of Malaysia) |
Cantel Medical Middle East FZ-LLC | (Incorporated under the laws of Dubai (UAE)) |
Cantel (Germany) GmbH | (Incorporated under the laws of Germany) |
Cantel (France) SAS | (Incorporated under the laws of France) |
Cantel (Australia) PTY LLTD | (Incorporated under the laws of Australia) |
BHT Hygienetechnik Holding GmbH | (Incorporated under the laws of Germany) |
BHT Hygienetechnik GmbH | (Incorporated under the laws of Germany) |
ESCAD Medical GmbH | (Incorporated under the laws of Germany) |
EXHIBIT 23
Consent of Independent Registered Public Accounting Firm
We consent to the incorporation by reference in the following Registration Statements:
(1) | Registration Statements (Form S-8 Nos. 333-140388, 333-157033, 333-163806 and 333-180171) pertaining to the Cantel Medical Corp. 2006 Equity Incentive Plan, as amended, and |
(2) Registration Statement (Form S-8 No. 333-210073) pertaining to the Cantel Medical Corp. 2016 Equity Incentive
Plan;
of our reports dated September 28, 2017, with respect to the consolidated financial statements and schedule of Cantel Medical Corp., and the effectiveness of internal control over financial reporting of Cantel Medical Corp., included in this Annual Report (Form 10‑K) of Cantel Medical Corp. for the year ended July 31, 2017.
/s/ Ernst & Young LLP
New York, New York
September 28, 2017
EXHIBIT 31.1
CERTIFICATIONS
I, Jorgen B. Hansen, certify that:
1. | I have reviewed this Annual Report on Form 10-K of Cantel Medical Corp.; |
2. | Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report; |
3. | Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report; |
4. | The registrant’s other certifying officers and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15 (e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and we have: |
a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared; |
b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; |
c) | Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and |
d) | Disclosed in this report any changes in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and |
5. | The registrant’s other certifying officers and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of registrant’s board of directors (or persons performing the equivalent functions): |
a) | All significant deficiencies and material weaknesses in the design or operation of internal controls over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and |
b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal controls over financial reporting. |
Date: September 28, 2017
By: | /s/ Jorgen B. Hansen |
Jorgen B. Hansen, President and Chief Executive Officer (Principal Executive Officer) | |
EXHIBIT 31.2
CERTIFICATIONS
I, Peter G. Clifford, certify that:
1. | I have reviewed this Annual Report on Form 10-K of Cantel Medical Corp.; |
2. | Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report; |
3. | Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report; |
4. | The registrant’s other certifying officers and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15 (e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and we have: |
a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared; |
b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; |
c) | Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and |
d) | Disclosed in this report any changes in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and |
5. | The registrant’s other certifying officers and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of registrant’s board of directors (or persons performing the equivalent functions): |
a) | All significant deficiencies and material weaknesses in the design or operation of internal controls over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and |
b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal controls over financial reporting. |
Date: September 28, 2017
By: | /s/ Peter G. Clifford |
Peter G. Clifford, Executive Vice President and Chief Financial Officer | |
Exhibit 32
CERTIFICATION
PURSUANT TO SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
(SUBSECTIONS (a) AND (b) OF SECTION 1350, CHAPTER 63 OF
TITLE 18, UNITED STATES CODE)
Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (subsections (a) and (b) of section 1350, chapter 63 of Title 18, United States Code), the undersigned officers of Cantel Medical Corp. (the “Company”), do hereby certify with respect to the Annual Report of the Company on Form 10-K for the year ended July 31, 2017 as filed with the Securities and Exchange Commission (the “Form 10-K”) that, to the best of their knowledge:
1. | The Form 10-K fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934; and |
2. | The information contained in the Form 10-K fairly presents, in all material respects, the financial condition and results of operations of the Company. |
Date: September 28, 2017
/s/ Jorgen B. Hansen |
Jorgen B. Hansen |
President and Chief Executive Officer |
(Principal Executive Officer) |
/s/ Peter G. Clifford |
Peter G. Clifford |
Executive Vice President, Chief Financial Officer |
(Principal Financial Officer) |
