Form S-1 Jaguar Health, Inc.
Use these links to rapidly review the document
TABLE OF CONTENTS
As filed with the Securities and Exchange Commission on September 12, 2018.
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
JAGUAR HEALTH, INC.
(Exact name of registrant as specified in its charter)
| Delaware (State or other jurisdiction of incorporation or organization) |
2834 (Primary Standard Industrial Classification Code Number) |
46-2956775 (I.R.S. Employer Identification Number) |
201 Mission Street, Suite 2375
San Francisco, California 94105
(415) 371-8300
(Address, including zip code, and telephone number, including area code, of registrant's principal executive office)
Lisa A. Conte
Chief Executive Officer and President
Jaguar Health, Inc.
201 Mission Street, Suite 2375
San Francisco, California 94105
(415) 371-8300
(Name, address, including zip code, and telephone number, including area code, of agent for service)
| Copies to: | ||
Donald C. Reinke, Esq. Reed Smith LLP 101 Second Street, Suite 1800 San Francisco, California 94105 (415) 543-8700 |
Robert F. Charron, Esq. Ellenoff Grossman & Schole LLP 1345 Avenue of the Americas New York, NY 10105 (212) 370-1300 |
|
Approximate date of commencement of proposed sale to the public:
As soon as practicable after this registration statement is declared effective.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933 check the following box: o
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of "large accelerated filer," "accelerated filer," "smaller reporting company," and "emerging growth company" in Rule 12b-2 of the Exchange Act.
| Large accelerated filer o | Accelerated filer o | Non-accelerated filer o |
Smaller reporting company ý Emerging growth company ý |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. ý
CALCULATION OF REGISTRATION FEE
|
||||
| Title of Each Class of Securities to be Registered(1) |
Proposed Maximum Aggregate Offering Price(2)(3) |
Amount of Registration Fee(3) |
||
|---|---|---|---|---|
Common Stock, par value $0.0001 per share |
$11,500,000 | $ | ||
Pre-funded warrants to purchase shares of common stock and common stock issuable upon exercise thereof |
— | — | ||
Underwriter warrants to purchase shares of common stock issuable upon exercise thereof(4) |
$1,150,000 | |||
Total |
$12,650,000 | $1,574.93 | ||
|
||||
- (1)
- Pursuant
to Rule 416 under the Securities Act of 1933, as amended, the securities being registered hereunder include such indeterminate number of additional
securities as may be issuable to prevent dilution resulting from stock splits, stock dividends or similar transactions.
- (2)
- The
proposed maximum aggregate offering price of the common stock proposed to be sold in the offering will be reduced on a dollar-for-dollar basis based on the
aggregate offering price of the pre-funded warrants offered and sold in the offering, and therefore the proposed aggregate maximum offering price of the common stock and pre-funded warrants (including
the common stock issuable upon exercise of the pre-funded warrants), if any, is $11,500,000.
- (3)
- Estimated
solely for the purpose of calculating the amount of the registration fee pursuant to Rule 457(o) of the Securities Act of 1933, as amended. Includes
the offering price of any additional securities that the underwriter has the option to purchase.
- (4)
- Represents warrants issuable to H.C. Wainwright & Co., LLC (the "Underwriter's Warrants") to purchase a number of shares of common stock equal to 8% of the number of shares of Common Stock and pre-funded warrants being offered at an exercise price equal to 125% of the public offering price. Resales of the Underwriter's Warrants on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, as amended, are registered hereby. Resales of shares of common stock issuable upon exercise of the Underwriter's Warrants are also being similarly registered on a delayed or continuous basis hereby. See "Underwriting."
The Registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until the registration statement shall become effective on such date as the Commission, acting pursuant to said Section 8(a), may determine.
The information in this preliminary prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities and it is not soliciting offers to buy these securities in any jurisdiction where the offer or sale is not permitted.
SUBJECT TO COMPLETION DATED SEPTEMBER 12, 2018
PRELIMINARY PROSPECTUS
Shares of Common Stock
Pre-Funded Warrants to Purchase Shares of Common Stock

We are offering shares of our common stock. We are also offering to certain purchasers whose purchase of shares of common stock in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock immediately following the consummation of this offering, the opportunity to purchase, if any such purchaser so chooses, pre-funded warrants, in lieu of shares of common stock that would otherwise result in such purchaser's beneficial ownership exceeding 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock. The purchase price of each pre-funded warrant will be equal to the price per share at which shares of common stock are sold to the public in this offering, minus $0.01, and the exercise price of each pre-funded warrant will be $0.01 per share. This offering also relates to the shares of common stock issuable upon exercise of any pre-funded warrants sold in this offering. The pre-funded warrants will be exercisable immediately and may be exercised at any time until all of the pre-funded warrants are exercised in full. For each pre-funded warrant we sell, the number of shares of common stock we are offering will be decreased on a one-for-one basis. Our common stock is listed on The NASDAQ Capital Market under the symbol "JAGX." On September 10, 2018, the last reported sale price of our common stock was $0.66 per share.
We are an "emerging growth company" as that term is used in the Jumpstart Our Business Startups Act of 2012 and, as such, have elected to comply with certain reduced public company reporting requirements for this prospectus and future filings.
Our business and an investment in our securities involves a high degree of risk. See "Risk Factors" beginning on page 7 of this prospectus for a discussion of information that you should consider before investing in our securities.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
|
||||||
| |
Per Share |
Per Pre-funded Warrant |
Total |
|||
|---|---|---|---|---|---|---|
Public offering price |
$ | $ | $ | |||
Underwriting discounts and commissions(1) |
$ | $ | $ | |||
Proceeds, before expenses, to us |
$ | $ | $ | |||
|
||||||
- (1)
- See "Underwriting" beginning on page 120 of this prospectus for a description of compensation payable to the underwriter.
The offering is being underwritten on a firm commitment basis. We have granted the underwriter an option for a period of 30 days from the date of this prospectus to purchase up to an additional shares of our common stock (15% of the number of shares of common stock and pre-funded warrants sold in this offering) at the public offering price, less the underwriting discounts and commissions, to cover over-allotments, if any. If the underwriter exercises this option in full, the total underwriting discounts and commissions payable by us will be $ , and the total proceeds to us, before expenses, will be $ .
The underwriter expects to deliver the shares and pre-funded warrants against payment therefor on or about , 2018.
Sole Book-Running Manager
H.C. Wainwright & Co.
, 2018
We have not, and the underwriter has not, authorized anyone to provide any information or to make any representations other than those contained in this prospectus or in any free writing prospectus prepared by or on behalf of us or to which we have referred you. We take no responsibility for, and can provide no assurance as to the reliability of, any other information that others may give you. This prospectus is an offer to sell only the shares offered hereby, but only under the circumstances and in the jurisdictions where it is lawful to do so. The information contained in this prospectus or in any applicable free writing prospectus is current only as of its date, regardless of its time of delivery or any sale of shares of our common stock. Our business, financial condition, results of operations and prospects may have changed since that date. We are not, and the underwriter is not, making an offer of these securities in any jurisdiction where such offer is not permitted.
For investors outside the United States: we have not, and the underwriter has not, done anything that would permit this offering or possession or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than in the United States. Persons outside the United States who come into possession of this prospectus must inform themselves about, and observe any restrictions relating to, the offering of securities and the distribution of this prospectus outside the United States.
Jaguar Health, our logo, Napo Pharmaceuticals, Mytesi, Canalevia, Equilevia and Neonorm are our trademarks that are used in this prospectus. This prospectus also includes trademarks, tradenames and service marks that are the property of other organizations. Solely for convenience, trademarks and tradenames referred to in this prospectus appear without the ©, ® or ™ symbols, but those references are not intended to indicate that we will not assert, to the fullest extent under applicable law, our rights or that the applicable owner will not assert its rights, to these trademarks and tradenames.
This summary highlights information contained elsewhere in this prospectus. This summary does not contain all of the information you should consider before investing in our common stock. You should read this entire prospectus carefully, especially the section in this prospectus titled "Risk Factors" appearing elsewhere in this prospectus the information incorporated by reference herein, including our financial statements and notes in our annual report on Form 10-K for 2017 and quarterly reports on Form 10-Q, before making an investment decision.
As used in this prospectus, references to "Jaguar," "we," "us" or "our" refer to Jaguar Health, Inc.
Overview
We are a commercial stage pharmaceutical company focused on developing novel, sustainably derived gastrointestinal products on a global basis. Our wholly-owned subsidiary, Napo Pharmaceuticals, Inc. ("Napo"), focuses on developing and commercializing proprietary human gastrointestinal pharmaceuticals for the global marketplace from plants used traditionally in rainforest areas. Our Mytesi (crofelemer) product is a first-in-class anti-secretory agent, approved by the U.S. Food and Drug Administration ("FDA") for the symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy. The full launch of Mytesi began in April 2018—with the support of a full complement of patient and healthcare practitioner programs—when Napo's direct sales force of 18 sales representatives, a national sales director and one regional sales director became fully deployed.
Jaguar was founded in San Francisco, California as a Delaware corporation on June 6, 2013. The Company was a majority-owned subsidiary of Napo until the close of the Company's initial public offering on May 18, 2015. On July 31, 2017 with the merger of Jaguar and Napo, Napo began operating as a wholly-owned subsidiary of Jaguar focused on human health and the ongoing commercialization of, and development of follow-on indications for, Mytesi.
We believe that Jaguar is poised to realize a number of value adding events—including continued commercial growth of Mytesi for the current HIV-diarrhea indication, an expanded pipeline of human follow-on indications, and a second-generation anti-secretory agent, upon which to build global partnerships. As previously announced, Jaguar, through Napo, now controls commercial rights for Mytesi for all indications, territories and patient populations globally, and crofelemer manufacturing is being conducted at a new, multimillion-dollar commercial manufacturing facility that has been FDA-inspected and approved. Additionally, several of the drug product candidates in Jaguar's Mytesi pipeline are backed by Phase 2 and/or proof of concept evidence from completed human clinical trials. The current approved indication in HIV-related diarrhea is a chronic approval, with a safety package that supports chronic use of Mytesi for follow-on indications as well.
Mytesi is a novel, first-in-class anti-secretory agent which has a basic normalizing effect locally on the gut, and this mechanism of action has the potential to benefit multiple gastrointestinal disorders. Mytesi is in development for multiple possible follow-on indications, including cancer therapy-related diarrhea; orphan-drug indications for infants and children with congenital diarrheal disorders and short bowel syndrome (SBS); supportive care for inflammatory bowel disease (IBD); irritable bowel syndrome (IBS); and as a second-generation anti-secretory agent for use in cholera patients. Mytesi has received orphan-drug designation for SBS.
Corporate Information
We were incorporated in the State of Delaware on June 6, 2013. Our principal executive offices are located at 201 Mission Street, Suite 2375, San Francisco, CA 94015 and our telephone number is (415) 371-8300. Our website address is www.jaguar.health. The information contained on, or that can
1
be accessed through, our website is not part of this prospectus. Our voting common stock is listed on the NASDAQ Capital Market and trades under the symbol "JAGX." On July 31, 2017, we completed the acquisition of Napo (the "Merger") pursuant to the Agreement and Plan of Merger, dated March 31, 2017, by and among the Company, Napo, Napo Acquisition Corporation, and Napo's representative (the "Merger Agreement").
Emerging Growth Company Information
We are an "emerging growth company," as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and we may take advantage of certain exemptions and relief from various reporting requirements that are applicable to other public companies that are not "emerging growth companies." In particular, while we are an "emerging growth company" (i) we will not be required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act, (ii) we will be subject to reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements and (iii) we will not be required to hold nonbinding advisory votes on executive compensation or stockholder approval of any golden parachute payments not previously approved. In addition, the JOBS Act provides that an emerging growth company can delay its adoption of any new or revised accounting standards, but we have irrevocably elected not to avail ourselves of this exemption and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies. In addition, investors may find our common stock less attractive if we rely on the exemptions and relief granted by the JOBS Act. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may decline and/or become more volatile.
We may remain an "emerging growth company" until as late as December 31, 2020 (the fiscal year-end following the fifth anniversary of the closing of our initial public offering, which occurred on May 18, 2015), although we may cease to be an "emerging growth company" earlier under certain circumstances, including (i) if the market value of our common stock that is held by non-affiliates exceeds $700.0 million as of any June 30, in which case we would cease to be an "emerging growth company" as of December 31 of such year, (ii) if our gross revenue exceeds $1.07 billion in any fiscal year or (iii) if we issue more than $1.0 billion of non-convertible debt over a three-year period.
Risks Associated with Our Business and this Offering
Our business and our ability to implement our business strategy are subject to numerous risks, as more fully described in the section of this prospectus entitled "Risk Factors" and under similarly titled headings of the documents incorporated herein by reference. You should read these risks before you invest in our securities
2
Common stock offered by us |
shares | |
Pre-funded warrants offered by us in this offering |
We are also offering to certain purchasers whose purchase of shares of common stock in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock immediately following the consummation of this offering, the opportunity to purchase, if such purchasers so choose, pre-funded warrants, in lieu of shares of common stock that would otherwise result in any such purchaser's beneficial ownership exceeding 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock. Each pre-funded warrant will be exercisable for one share of our common stock. The purchase price of each pre-funded warrant will equal the price per share at which the shares of common stock are being sold to the public in this offering, minus $0.01, and the exercise price of each pre-funded warrant will be $0.01 per share. The pre-funded warrants will be exercisable immediately and may be exercised at any time until all of the pre-funded warrants are exercised in full. This offering also relates to the shares of common stock issuable upon exercise of any pre-funded warrants sold in this offering. For each pre-funded warrant we sell, the number of shares of common stock we are offering will be decreased on a one-for-one basis. |
|
Option to purchase additional shares |
The underwriter has an option within 30 days of the date of this prospectus to purchase up to additional shares of our common stock in this offering. |
|
Common stock to be outstanding after this offering |
shares of common stock, assuming no sale of any pre-funded warrants (or shares of common stock if the underwriter exercises in full its option to purchase additional shares of common stock, assuming no sale of pre-funded warrants). |
|
Use of proceeds |
We estimate the net proceeds from this offering will be approximately $ million (or $ million if the underwriter exercises its option to purchase additional shares in full), based on an assumed public offering price of $ per share, the last reported sale price of our common stock on The Nasdaq Capital Market on , 2018, after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
|
|
We intend to use the net proceeds from this offering, together with our existing cash and cash equivalents, for general corporate purposes and working capital. See "Use of Proceeds." |
|
Dividend policy |
We have not paid or declared any dividends on our common stock. For more information, see the section titled "Dividend policy." |
3
Risk factors |
You should read the section titled "Risk Factors" beginning on page 7 of this prospectus and other information included in this prospectus for a discussion of factors to consider carefully before deciding to invest in shares of our common stock. |
|
National Securities Exchange Listing |
Our common stock is listed on the NASDAQ Capital Market under the symbol "JAGX." We do not intend to list the pre-funded warrants on any securities exchange or nationally recognized trading system. |
The number of shares of our common stock to be outstanding after this offering is based on 8,736,579 shares of voting common stock and 40,301,237 shares of nonvoting common shares outstanding as of June 30, 2018 and assumes the sale and issuance by us of shares of common stock in this offering and excludes, as of June 30, 2018:
- •
- 3,314,955 shares of common stock issuable upon conversion of outstanding preferred stock as of June 30, 2018 with a weighted-average
conversion price of $2.775 per share;
- •
- 2,704,692 shares of common stock issuable upon exercise of outstanding options as of June 30, 2018 with a weighted-average exercise
price of $6.40 per share;
- •
- 402,348 shares of common stock reserved for future issuance under our 2014 Stock Incentive Plan as of June 30, 2018;
- •
- 209,531 shares of common stock issuable upon exercise of outstanding inducement options as of June 30, 2018 with a weighted-average
exercise price of $1.75 per share;
- •
- 392,904 shares of common stock issuable upon exercise of outstanding restricted stock unit awards, or RSUs, as of June 30, 2018;
- •
- 270,761 shares of common stock issuable upon exercise of outstanding warrants as of June 30, 2018 with a weighted-average exercise price of $16.35 per share;
The number of shares of common stock outstanding after the offering also does not include the following:
- •
- 866,524 shares of common stock issued after June 30, 2018;
- •
- Up to 720,721 shares of common stock issuable upon conversion of outstanding convertible promissory notes in the aggregate principal amount of
$10,000,000 issued after June 30, 2018, convertible at a price of $13.875 per share ;
- •
- 38,675 shares of common stock issuable pursuant to a convertible promissory note issued to Chicago Venture Partners, L.P. ("CVP"), in
the aggregate principal amount of $580,127, convertible at a price of $15 per share of common stock;
- •
- 670,586 shares of common stock, issuable upon exercise of a warrant issued on August 28, 2018 to Pacific Capital Management, LLC
with an exercise price that is the lower of (i) $0.85 per share and (ii) the average of the closing sales price of the common stock for the 30 consecutive trading days
commencing on September 4, 2018;
- •
- 185,417 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to L2 Capital, LLC with an
exercise price of $0.90 per share;
- •
- 33,918 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to Charles C. Conte with an exercise price of $1.23 per share;
4
- •
- Up to 706,765 shares of common stock issuable upon conversion of a convertible promissory note delivered to L2 Capital, LLC on
September 11, 2018 in the aggregate principal amount of $445,000 with an exercise price of $0.85 per share; and
- •
- Up to 176,691 shares of common stock issuable upon conversion of a convertible promissory note delivered to Charles C. Conte on September 11, 2018 in the aggregate principal amount of $111,250 with an exercise price of $0.85 per share.
5
Summary Consolidated Financial Data
You should read the following summary consolidated financial data together with our consolidated financial statements and the notes to those consolidated financial statements appearing in our Annual Report on Form 10-K for the fiscal year ended December 31, 2017 and our Quarterly Report on Form 10-Q for the period ended June 30, 2018, which are incorporated by reference in this prospectus. We have derived the consolidated statement of operations data for the years ended December 31, 2017 and 2016 from our audited consolidated financial statements. The consolidated statement of operations data for the six months ended June 30, 2017 and 2018 and the consolidated balance sheet data as of June 30, 2018 have been derived from our unaudited consolidated financial statements incorporated by reference to our Quarterly Report of Form 10-Q for the quarter ended June 30, 2018 and have been prepared on the same basis as the audited consolidated financial statements. Our historical results are not necessarily indicative of the results that may be expected in the future, and our results for any interim period are not necessarily indicative of results that may be expected for any full year.
| |
Year Ended December 31, | Six Months Ended June 30, | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2016 | 2017 | 2017 | 2018 | |||||||||
Product revenue |
$ | 141,523 | $ | 1,485,114 | $ | 135,989 | $ | 1,510,813 | |||||
Collaboration revenue |
— | 2,876,072 | 1,582,942 | 177,389 | |||||||||
| | | | | | | | | | | | | | |
Total revenue |
141,523 | 4,361,186 | 1,718,931 | 1,688,202 | |||||||||
Operating Expenses |
|||||||||||||
Cost of product revenue |
51,966 | 880,405 | 40,907 | 1,072,185 | |||||||||
Research and development expense |
7,206,864 | 4,269,455 | 2,182,243 | 2,362,752 | |||||||||
Sales and marketing expense |
485,440 | 3,083,739 | 280,143 | 4,402,452 | |||||||||
General and administrative expense |
5,983,238 | 11,247,647 | 5,441,493 | 6,058,148 | |||||||||
Impairment of goodwill |
— | 16,827,000 | — | — | |||||||||
Impairment of indefinite-lived intangible assets |
— | 2,300,000 | — | — | |||||||||
| | | | | | | | | | | | | | |
Total operating expenses |
13,727,508 | 38,608,246 | 7,944,786 | 13,895,537 | |||||||||
Loss from operations |
(13,585,985 | ) | (34,247,060 | ) | (6,225,855 | ) | (12,207,335 | ) | |||||
Interest expense |
(985,549 | ) | (1,209,632 | ) | (336,201 | ) | (1,313,824 | ) | |||||
Other income/(expense) |
(11,046 | ) | 88,549 | 1,448 | 312,704 | ||||||||
Change in fair value of warrants |
(43,200 | ) | 695,341 | 247,321 | (145,365 | ) | |||||||
Loss on extinguishment of debt |
(108,000 | ) | (477,054 | ) | (207,713 | ) | — | ||||||
| | | | | | | | | | | | | | |
Net loss before income tax |
(14,733,780 | ) | (35,149,856 | ) | (6,521,000 | ) | (13,353,820 | ) | |||||
Income tax benefit |
— | 13,181,242 | — | — | |||||||||
| | | | | | | | | | | | | | |
Net loss and comprehensive loss |
$ | (14,733,780 | ) | $ | (21,968,614 | ) | $ | (6,521,000 | ) | $ | (13,353,820 | ) | |
Deemed dividend attributable to preferred stock |
(995,000 | ) | |||||||||||
Net loss attributable to common shareholders |
$ | (14,733,780 | ) | $ | (21,968,614 | ) | $ | (6,521,000 | ) | $ | (14,348,820 | ) | |
Net loss per share, basic and diluted |
$ | (5.09 | ) | $ | (30.09 | ) | $ | (6.78 | ) | $ | (1.43 | ) | |
Weighted-average common shares outstanding, basic and diluted |
2,895,729 | 730,079 | 961,821 | 10,010,862 | |||||||||
6
Investing in our securities involves a high degree of risk. You should carefully consider the risks described below, as well as the other information in this prospectus and incorporated by reference herein, including our financial statements and the related notes, before deciding whether to invest in our common stock. The occurrence of any of the events or developments described below could harm our business, financial condition, results of operations and prospects. In such an event, the market price of our common stock could decline, and you may lose all or part of your investment. Additional risks and uncertainties not presently known to us or that we currently deem immaterial also may harm our business, financial condition, results of operations and prospects.
Risks Related to Our Financial Position and Need for Additional Capital
We have a limited operating history, expect to incur further losses as we grow and may be unable to achieve or sustain profitability. Our independent registered public accounting firm has expressed substantial doubt about our ability to continue as a going concern.
From the date of our formation in June 2013, until the consummation of the Napo merger on July 31, 2017, our operations have been primarily limited to the research and development of our animal prescription drug product candidate, Canalevia, to treat various forms of diarrhea in dogs, our non-prescription product, Neonorm Calf, to help dairies and calf farms proactively retain fluid in calves, the ongoing commercialization of Neonorm Foal, our antidiarrheal for newborn horses, and Equilevia, our product for total gut health in high-performance equine athletes. Since the consummation of the Merger on July 31, 2017, our operations have been primarily focused on research, development and the ongoing commercialization of our lead prescription drug product candidate, Mytesi, which is approved by the U.S. FDA for the symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy. As a result, we have limited meaningful historical operations upon which to evaluate our business and prospects or to successfully overcome the risks and uncertainties frequently encountered by companies in emerging fields such as the gastrointestinal health industry in general. We also have not generated any material revenue to date, and expect to continue to incur significant research and development and other expenses. Our net loss and comprehensive loss for the year ended December 31, 2017 was $22.0 million. As of June 30, 2018, we had an accumulated deficit of $75.8 million. We expect to continue to incur losses for the foreseeable future, which will increase significantly from historical levels as we expand our product development activities, seek necessary approvals for our human and veterinary drug product candidates, conduct species-specific formulation studies for our non-prescription products and begin commercialization activities. Even if we succeed in developing and broadly commercializing one or more of our products or product candidates beyond Mytesi, we expect to continue to incur losses for the foreseeable future, and we may never become profitable. If we fail to achieve or maintain profitability, then we may be unable to continue our operations at planned levels and be forced to reduce or cease operations.
As more fully discussed in Note 1 to our consolidated financial statements for the year ended December 31, 2017 and the quarter ended June 30, 2018, we believe there is substantial doubt about our ability to continue as a going concern as we do not currently have sufficient cash resources to fund our operations through August 13, 2019, or one year from the filing date of our Form 10-Q. Our financial statements do not include any adjustments that may result from the outcome of this uncertainty. If we are unable to continue as a viable entity, our stockholders may lose their entire investment.
We currently generate limited revenue from the sale of products and may never become profitable.
We are a natural-products pharmaceuticals company focused on the development and commercialization of novel, sustainably derived gastrointestinal products for human prescription use
7
primarily and also for animals on a global basis. Napo, our wholly-owned subsidiary, began the commercial pre-launch activities of our first FDA approved product, Mytesi, in February 2017. Accordingly, we have only generated limited revenue from product sales. There is no guarantee that our ongoing commercialization efforts for Neonorm Calf for preweaned dairy calves in the United States and Neonorm Foal for newborn horses in the United States will be successful or that we will be able to generate a consistent revenue stream from the sale of any of these products in the future. Further, in order to commercialize our other prescription drug product candidates, we must receive regulatory approval from the FDA in the United States and other regulatory agencies in various jurisdictions. Other than Mytesi, we have not yet received any regulatory approvals for our prescription drug product candidates. In addition, certain of our non-prescription products, such as Neonorm Calf, may be subject to regulatory approval outside the United States prior to commercialization in other countries. Accordingly, until and unless we receive any necessary regulatory approvals, we cannot market or sell our products in many regions. Moreover, even if we receive the necessary approvals, we may not be successful in generating revenue from sales of our products as we do not have any meaningful experience marketing or distributing our products. Accordingly, we may never generate any material revenue from our operations.
Raising additional capital may cause dilution to our stockholders, including purchasers of our common stock in this offering, restrict our operations or require us to relinquish rights to our technologies or product candidates.
Unless and until we can generate a substantial amount of revenue from our products or product candidates, we expect to finance our future cash needs through public or private equity offerings, debt financings or collaborations, licensing arrangements and government funding arrangements. In addition, we may seek additional capital due to favorable market conditions or strategic considerations, even if we believe that we have sufficient funds for our current or future operating plans.
To the extent that we raise additional capital through the sale of common stock, convertible securities or other equity securities, your ownership interest may be materially diluted, and the terms of these securities could include liquidation or other preferences and anti-dilution protections that could adversely affect your rights as a stockholder. In addition, debt financing, if available, would result in increased fixed payment obligations and may involve agreements that include restrictive covenants that limit our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends, which could adversely affect our ability to conduct our business. In addition, securing additional financing would require a substantial amount of time and attention from our management and may divert a disproportionate amount of their attention away from day-to-day activities, which may adversely affect our management's ability to oversee the development of our product candidates.
If we raise additional funds through collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams or product candidates or grant licenses on terms that may not be favorable to us.
Our ability to continue as a going concern requires that we obtain sufficient funding to finance our operations.
Our ability to continue as a going concern requires that we obtain sufficient funding to finance our operations. If we are unable to obtain sufficient funding, our business, prospects, financial condition and results of operations will be materially and adversely affected and we may be unable to continue as a going concern. If we are unable to continue as a going concern, we may have to liquidate our assets and may receive less than the value at which those assets are carried on our audited financial statements, and it is likely that investors will lose all or a part of their investment. If we seek additional
8
financing to fund our business activities in the future and there remains substantial doubt about our ability to continue as a going concern, investors or other financing sources may be unwilling to provide additional funding to us on commercially reasonable terms or at all.
We expect to incur significant additional costs as we continue commercialization efforts for current prescription drug candidates, or other product candidates, and undertake the clinical trials necessary to obtain any necessary regulatory approvals, which will increase our losses.
We commenced sales of Neonorm for preweaned dairy calves in the United States under the brand name Neonorm Calf at the end of 2014, and Napo commenced sales of Mytesi for adults with HIV/AIDS on antiretroviral therapy in February 2017. We will need to continue to invest in developing our internal and third-party sales and distribution network and outreach efforts to key opinion leaders in the gastrointestinal health industry, including physicians, as applicable. We will also need to conduct clinical trials for Mytesi in order to broaden Mytesi to additional indications.
We are actively identifying additional products for development and commercialization, and will continue to expend substantial resources for the foreseeable future to develop Mytesi, a second generation anti-secretory agent, and Canalevia for the MUMS indication of chemotherapy-induced diarrhea in dogs. These expenditures will include costs associated with:
- •
- identifying additional potential prescription drug product candidates and non-prescription products;
- •
- formulation studies;
- •
- conducting pilot, pivotal and toxicology studies;
- •
- completing other research and development activities;
- •
- payments to technology licensors;
- •
- maintaining our intellectual property;
- •
- obtaining necessary regulatory approvals;
- •
- establishing commercial supply capabilities; and
- •
- sales, marketing and distribution of our commercialized products.
We also may incur unanticipated costs in connection with developing and commercializing our products. Because the outcome of our development activities and commercialization efforts is inherently uncertain, the actual amounts necessary to successfully complete the development and commercialization of our current or future products and product candidates may be greater than we anticipate.
Because we anticipate incurring significant costs for the foreseeable future, if we are not successful in broadly commercializing any of our current or future products or product candidates or raising additional funding, including potential business development and/or strategic partnerships, to pursue our research and development efforts, we may never realize the benefit of our development efforts and our business may be harmed.
9
We will need to raise substantial additional capital in the future in the event that we conduct clinical trials for new indications and we may be unable to raise such funds when needed and on acceptable terms, which would force us to delay, limit, reduce or terminate one or more of our product development programs.
We are forecasting continued losses and negative cash flows as we continue to fund our operating and marketing activities and research and development programs, and we will not have sufficient cash on hand to fund our operating plan through March 31, 2019 and to complete the development of all the current products in our pipeline, or any additional products we may identify. We will need to seek additional funds sooner than planned through public or private equity or debt financings or other sources such as strategic collaborations. Any such financings or collaborations may result in dilution to our stockholders, the imposition of debt covenants and repayment obligations or other restrictions that may harm our business or the value of our common stock. We may also seek from time to time to raise additional capital based upon favorable market conditions or strategic considerations such as potential acquisitions or potential license arrangements.
Our future capital requirements depend on many factors, including, but not limited to:
- •
- the scope, progress, results and costs of researching and developing our current and future prescription drug product candidates and
non-prescription products;
- •
- the timing of, and the costs involved in, obtaining any regulatory approvals for our current and any future products;
- •
- the number and characteristics of the products we pursue;
- •
- the cost of manufacturing our current and future products and any products we successfully commercialize;
- •
- the cost of commercialization activities for Mytesi, Neonorm, Equilevia and Canalevia, if approved, including sales, marketing and distribution
costs;
- •
- the expenses needed to attract and retain skilled personnel;
- •
- the costs associated with being a public company;
- •
- our ability to establish and maintain strategic collaborations, distribution or other arrangements and the financial terms of such agreements;
and
- •
- the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing possible patent claims, including litigation costs and the outcome of any such litigation.
Additional funds may not be available when we need them on terms that are acceptable to us, or at all. If adequate funds are not available to us on a timely basis, we may be required to delay, limit, reduce or terminate one or more of our product development programs or future commercialization efforts.
We are substantially dependent on the success of our current lead prescription drug product candidate, Mytesi, and cannot be certain that necessary approvals will be received for planned Mytesi follow-on indications or that these product candidates will be successfully commercialized, either by us or any of our partners.
Other than Mytesi, we currently do not have regulatory approval for any of our prescription drug product candidates, including Canalevia. Our current efforts are primarily focused on the ongoing commercialization of Mytesi, and development efforts related to Mytesi and Canalevia. With regard to Mytesi, we are focused on the commercial launch of the product in the United States as well as on development efforts related to a follow-on indication for Mytesi in CTD, an important supportive care indication for patients undergoing cancer treatment. Mytesi is also in development for rare disease indications for infants and children with congenital diarrheal disorders and short bowel syndrome; for
10
IBS (Mytesi has demonstrated benefit to IBS-D patients in published Phase 2 studies); for supportive care for IBD; and as a second-generation anti-secretory agent for use in cholera patients. Mytesi has received orphan-drug designation for SBS. Accordingly, our near term prospects, including our ability to generate material product revenue, obtain any new financing if needed to fund our business and operations or enter into potential strategic transactions, will depend heavily on the success of Mytesi and potential follow-on indications.
Substantial time and capital resources have been previously devoted by third parties in the development of crofelemer, the active pharmaceutical ingredient, or API, in Mytesi and Canalevia, and the development of the botanical extract used in Equilevia and Neonorm. Both crofelemer and the botanical extract used in Equilevia and Neonorm were originally developed at Shaman Pharmaceuticals, Inc. ("Shaman"), by certain members of our management team, including Lisa A. Conte, our chief executive officer and president, and Steven R. King, Ph.D., our executive vice president of sustainable supply, ethnobotanical research and intellectual property and secretary. Shaman spent significant development resources before voluntarily filing for bankruptcy in 2001 pursuant to Chapter 11 of the U.S. Bankruptcy Code. The rights to crofelemer and the botanical extract used in Equilevia and Neonorm, as well as other intellectual property rights, were subsequently acquired by Napo from Shaman in 2001 pursuant to a court approved sale of assets. Ms. Conte founded Napo in 2001 and was the current interim chief executive officer of Napo and a member of Napo's board of directors prior to the Merger. While at Napo, certain members of our management team, including Ms. Conte and Dr. King, continued the development of crofelemer. In 2005, Napo entered into license agreements with Glenmark and Luye Pharma Group Limited for rights to various human indications of crofelemer in certain territories as defined in the respective license agreements with these licensees. Subsequently, after expending significant sums developing crofelemer, including trial design and on-going patient enrollment in the final pivotal Phase 3 trial for crofelemer for non-infectious diarrhea in adults with HIV/AIDS on antiretroviral therapy, in late 2008, Napo entered into a collaboration agreement with Salix Pharmaceuticals, Inc., or Salix, for development and commercialization rights to certain indications worldwide and certain rights in North America, Europe, and Japan, to crofelemer for human use. In January 2014, Jaguar entered into the Napo License Agreement pursuant to which Jaguar acquired an exclusive worldwide license to Napo's intellectual property rights and technology, including crofelemer and the botanical extract used in Equilevia and Neonorm, for all veterinary treatment uses and indications for all species of animals. In February 2014, most of the executive officers of Napo, and substantially all Napo's employees, became Jaguar's employees. In March 2016, Napo settled with Salix for the return of all commercial rights to crofelemer. Following the merger of Jaguar and Napo in July 2017, Napo became Jaguar's wholly-owned subsidiary. If we are not successful in the development and commercialization of Mytesi, Neonorm, Equilevia and Canalevia, our business and our prospects will be harmed.
The successful development and commercialization of Mytesi, Equilevia and Neonorm, and, if approved, Canalevia will depend on a number of factors, including the following:
- •
- our ability to demonstrate to the satisfaction of the FDA and any other regulatory bodies, the safety and efficacy of Canalevia for
chemotherapy-induced diarrhea in dogs;
- •
- our ability and that of our contract manufacturers to manufacture supplies of Mytesi, Neonorm, Equilevia and Canalevia
- •
- our ability to successfully continue commercial efforts associated with Mytesi, whether alone or in collaboration with others;
- •
- our ability to successfully launch Canalevia, assuming approval is obtained, and Equilevia, whether alone or in collaboration with others;
11
- •
- the availability, perceived advantages, relative cost, relative safety and relative efficacy of our prescription drug product candidates and
non-prescription products compared to alternative and competing treatments;
- •
- the acceptance of our prescription drug product candidates and non-prescription products as safe and effective by physicians, veterinarians,
patients, animal owners and the human and animal health community, as applicable;
- •
- our ability to achieve and maintain compliance with all regulatory requirements applicable to our business; and
- •
- our ability to obtain and enforce our intellectual property rights and obtain marketing exclusivity for our prescription drug product candidates and non-prescription products, and avoid or prevail in any third-party patent interference, patent infringement claims or administrative patent proceedings initiated by third parties or the U.S. Patent and Trademark Office ("USPTO").
Many of these factors are beyond our control. Accordingly, we may not be successful in developing or commercializing Mytesi, Neonorm, Equilevia, Canalevia or any of our other potential products. If we are unsuccessful or are significantly delayed in developing and commercializing Mytesi, Neonorm, Equilevia, Canalevia or any of our other potential products, our business and prospects will be harmed and you may lose all or a portion of the value of your investment in our common stock.
If we are not successful in identifying, licensing, developing and commercializing additional product candidates and products, our ability to expand our business and achieve our strategic objectives could be impaired.
Although a substantial amount of our efforts is focused on the commercial performance of Mytesi, a key element of our strategy is to identify, develop and commercialize a portfolio of products to serve the gastrointestinal health market. Most of our potential products are based on our knowledge of medicinal plants. Our current focus is primarily on product candidates whose active pharmaceutical ingredient or botanical extract has been successfully commercialized or demonstrated to be safe and effective in human or animal trials. In some instances, we may be unable to further develop these potential products because of perceived regulatory and commercial risks. Even if we successfully identify potential products, we may still fail to yield products for development and commercialization for many reasons, including the following:
- •
- competitors may develop alternatives that render our potential products obsolete;
- •
- an outside party may develop a cure for any disease state that is the target indication for any of our planned or approved drug products;
- •
- potential products we seek to develop may be covered by third-party patents or other exclusive rights;
- •
- a potential product may on further study be shown to have harmful side effects or other characteristics that indicate it is unlikely to be
effective or otherwise does not meet applicable regulatory criteria;
- •
- a potential product may not be capable of being produced in commercial quantities at an acceptable cost, or at all; and
- •
- a potential product may not be accepted as safe and effective by physicians, veterinarians, patients, animal owners, key opinion leaders and other decision-makers in the gastrointestinal health market, as applicable.
While we are developing specific formulations, including flavors, methods of administration, new patents and other strategies with respect to our current potential products, we may be unable to
12
prevent competitors from developing substantially similar products and bringing those products to market earlier than we can. If such competing products achieve regulatory approval and commercialization prior to our potential products, our competitive position may be impaired. If we fail to develop and successfully commercialize other potential products, our business and future prospects may be harmed and we will be more vulnerable to any problems that we encounter in developing and commercializing our current potential products.
Mytesi faces significant competition from other pharmaceutical companies, both for its currently approved indication and for planned follow-on indications, and our operating results will suffer if we fail to compete effectively.
The development and commercialization of products for human gastrointestinal health is highly competitive and our success depends on our ability to compete effectively with other products in the market. During the ongoing commercialization of Mytesi for its currently approved indication, and during the future commercialization of Mytesi for any planned follow-on indications, if such follow-on indications receive regulatory approval, we expect to compete with major pharmaceutical and biotechnology companies that operate in the gastrointestinal space, such as Sucampo AG, Takeda Pharmaceuticals, Allergan, Inc., Ironwood Pharmaceuticals, Inc., Synergy Pharmaceuticals Inc., Heron Therapeutics, Inc., Sebela Pharmaceuticals, Inc. and Salix Pharmaceuticals.
Many of our competitors and potential competitors in the human gastrointestinal space have substantially more financial, technical and human resources than we do. Many also have more experience in the development, manufacture, regulation and worldwide commercialization of human gastrointestinal health products.
For these reasons, we cannot be certain that we and Mytesi can compete effectively.
We may be unable to obtain, or obtain on a timely basis, regulatory approval for our existing or future human or animal prescription drug product candidates under applicable regulatory requirements, which would harm our operating results.
The research, testing, manufacturing, labeling, approval, sale, marketing and distribution of human and animal health products are subject to extensive regulation. We are typically not permitted to market our prescription drug product candidates in the United States until we receive approval of the product from the FDA through the filing of an NDA or NADA, as applicable. To gain approval to market a prescription drug, we must provide the FDA with safety and efficacy data from pivotal trials that adequately demonstrate that our prescription drug product candidates are safe and effective for the intended indications. Likewise, to gain approval to market an animal prescription drug for a particular species, we must provide the FDA with safety and efficacy data from pivotal trials that adequately demonstrate that our prescription drug product candidates are safe and effective in the target species (e.g. dogs) for the intended indications. In addition, we must provide manufacturing data evidencing that we can produce our product candidates in accordance with cGMP. For the FDA, we must also provide data from toxicology studies, also called target animal safety studies, and in some cases environmental impact data. In addition to our internal activities, we will partially rely on contract research organizations ("CROs"), and other third parties to conduct our toxicology studies and for certain other product development activities. The results of toxicology studies, other initial development activities, and/or any previous studies in humans or animals conducted by us or third parties may not be predictive of future results of pivotal trials or other future studies, and failure can occur at any time during the conduct of pivotal trials and other development activities by us or our CROs. Our pivotal trials may fail to show the desired safety or efficacy of our prescription drug product candidates despite promising initial data or the results in previous human or animal studies conducted by others. Success of a prescription drug product candidate in prior animal studies, or in the treatment of humans, does not ensure success in subsequent studies. Clinical trials in humans and pivotal trials in animals
13
sometimes fail to show a benefit even for drugs that are effective because of statistical limitations in the design of the trials or other statistical anomalies. Therefore, even if our studies and other development activities are completed as planned, the results may not be sufficient to obtain a required regulatory approval for a product candidate.
Regulatory authorities can delay, limit or deny approval of any of our prescription drug product candidates for many reasons, including:
- •
- if they disagree with our interpretation of data from our pivotal studies or other development efforts;
- •
- if we are unable to demonstrate to their satisfaction that our product candidate is safe and effective for the target indication and, if
applicable, in the target species;
- •
- if they require additional studies or change their approval policies or regulations;
- •
- if they do not approve of the formulation, labeling or the specifications of our current and future product candidates; and
- •
- if they fail to approve the manufacturing processes of our third-party contract manufacturers.
Further, even if we receive a required approval, such approval may be for a more limited indication than we originally requested, and the regulatory authority may not approve the labeling that we believe is necessary or desirable for successful commercialization.
Any delay or failure in obtaining any necessary regulatory approval for the intended indications of our human or animal product candidates would delay or prevent commercialization of such product candidates and would harm our business and our operating results.
The results of our earlier studies of Mytesi may not be predictive of the results in any future clinical trials and species-specific formulation studies, respectively, and we may not be successful in our efforts to develop or commercialize line extensions of Mytesi.
Our product pipeline includes a number of potential indications of Mytesi, our lead prescription product. The results of our studies and other development activities and of any previous studies conducted by us or third parties may not be predictive of future results of these clinical studies and formulation studies, respectively. Failure can occur at any time during the conduct of these trials and other development activities. Even if our formulation/clinical studies and other development activities are completed as planned, the results may not be sufficient to pursue a particular line extension for Mytesi. Further, even if we obtain promising results from our clinical trials, we may not successfully commercialize any line extension. Because line extensions are developed for a particular market, we may not be able to leverage our experience from the commercial launch of Mytesi in new markets. If we are not successful in developing and successfully commercializing these line extension products, we may not be able to grow our revenue and our business may be harmed.
Development of prescription drug products is inherently expensive, time-consuming and uncertain, and any delay or discontinuance of our current or future pivotal trials would harm our business and prospects.
Development of prescription drug products for human and animal gastrointestinal health remains an inherently lengthy, expensive and uncertain process, and our development activities may not be successful. We do not know whether our current or planned pivotal trials for any of our product candidates will begin or conclude on time, and they may be delayed or discontinued for a variety of reasons, including if we are unable to:
- •
- address any safety concerns that arise during the course of the studies;
14
- •
- complete the studies due to deviations from the study protocols or the occurrence of adverse events;
- •
- add new study sites;
- •
- address any conflicts with new or existing laws or regulations; or
- •
- reach agreement on acceptable terms with study sites, which can be subject to extensive negotiation and may vary significantly among different sites.
Further, we may not be successful in developing new indications for Mytesi and may be subject to the same regulatory regime as prescription drug products in jurisdictions outside the United States. Any delays in completing our development efforts will increase our costs, delay our development efforts and approval process and jeopardize our ability to commence product sales and generate revenue. Any of these occurrences may harm our business, financial condition and prospects. In addition, factors that may cause a delay in the commencement or completion of our development efforts may also ultimately lead to the denial of regulatory approval of our product candidates which, as described above, would harm our business and prospects.
We will partially rely on third parties to conduct our development activities. If these third parties do not successfully carry out their contractual duties, we may be unable to obtain regulatory approvals or commercialize our current or future human or animal product candidates on a timely basis, or at all.
We will partially rely upon CROs to conduct our toxicology studies and for other development activities. We intend to rely on CROs to conduct one or more of our planned pivotal trials. These CROs are not our employees, and except for contractual duties and obligations, we have limited ability to control the amount or timing of resources that they devote to our programs or manage the risks associated with their activities on our behalf. We are responsible for ensuring that each of our studies is conducted in accordance with the development plans and trial protocols presented to regulatory authorities. Any deviations by our CROs may adversely affect our ability to obtain regulatory approvals, subject us to penalties or harm our credibility with regulators. The FDA and foreign regulatory authorities also require us and our CROs to comply with regulations and standards, commonly referred to as good clinical practices ("GCPs"), or good laboratory practices ("GLPs"), for conducting, monitoring, recording and reporting the results of our studies to ensure that the data and results are scientifically valid and accurate.
Agreements with CROs generally allow the CROs to terminate in certain circumstances with little or no advance notice. These agreements generally will require our CROs to reasonably cooperate with us at our expense for an orderly winding down of the CROs' services under the agreements. If the CROs conducting our studies do not comply with their contractual duties or obligations, or if they experience work stoppages, do not meet expected deadlines, or if the quality or accuracy of the data they obtain is compromised, we may need to secure new arrangements with alternative CROs, which could be difficult and costly. In such event, our studies also may need to be extended, delayed or terminated as a result, or may need to be repeated. If any of the foregoing were to occur, regulatory approval, if required, and commercialization of our product candidates may be delayed and we may be required to expend substantial additional resources.
Even if we obtain regulatory approval for planned follow-on indications of Mytesi, or for Canalevia or our other product candidates, they may never achieve market acceptance. Further, even if we are successful in the ongoing commercialization of Mytesi, we may not achieve commercial success.
If we obtain necessary regulatory approvals for planned follow-on indications of Mytesi or for Canalevia or our other product candidates, such products may still not achieve market acceptance and
15
may not be commercially successful. Market acceptance of Mytesi and Canalevia, and any of our other products depends on a number of factors, including:
- •
- the safety of our products as demonstrated in our target human and animal studies;
- •
- the indications for which our products are approved or marketed;
- •
- the potential and perceived advantages over alternative treatments or products, including generic medicines and competing products currently
prescribed by physicians or veterinarians, as applicable, and, in the case of animal products, products approved for use in humans that are used extra-label in animals;
- •
- the acceptance by physicians, veterinarians, companion animal owners and production animal owners, as applicable, of our products as safe and
effective;
- •
- the cost in relation to alternative treatments and willingness on the part of physicians, veterinarians, patients and animal owners, as
applicable, to pay for our products;
- •
- the prevalence and severity of any adverse side effects of our products;
- •
- the relative convenience and ease of administration of our products; and
- •
- the effectiveness of our sales, marketing and distribution efforts.
Any failure by Mytesi, Canalevia and Equilevia or any of our other products to achieve market acceptance or commercial success would harm our financial condition and results of operations.
Human and animal gastrointestinal health products are subject to unanticipated post-approval safety or efficacy concerns, which may harm our business and reputation.
The success of our commercialization efforts will depend upon the perceived safety and effectiveness of human and animal gastrointestinal health products, in general, and of our products, in particular. Unanticipated safety or efficacy concerns can subsequently arise with respect to approved prescription drug products, such as Mytesi, or non-prescription products, such as Neonorm, which may result in product recalls or withdrawals or suspension of sales, as well as product liability and other claims. Any safety or efficacy concerns, or recalls, withdrawals or suspensions of sales of our products could harm our reputation and business, regardless of whether such concerns or actions are justified.
Future federal and state legislation may result in increased exposure to product liability claims, which could result in substantial losses.
Under current federal and state laws, companion and production animals are generally considered to be the personal property of their owners and, as such, the owners' recovery for product liability claims involving their companion and production animals may be limited to the replacement value of the animal. Companion animal owners and their advocates, however, have filed lawsuits from time to time seeking non-economic damages such as pain and suffering and emotional distress for harm to their companion animals based on theories applicable to personal injuries to humans. If new legislation is passed to allow recovery for such non-economic damages, or if precedents are set allowing for such recovery, we could be exposed to increased product liability claims that could result in substantial losses to us if successful. In addition, some horses can be worth millions of dollars or more, and product liability for horses may be very high. While we currently have product liability insurance, such insurance may not be sufficient to cover any future product liability claims against us.
16
If we fail to retain current members of our senior management, or to identify, attract, integrate and retain additional key personnel, our business will be harmed.
Our success depends on our continued ability to attract, retain and motivate highly qualified management and scientific personnel. We are highly dependent upon our senior management, particularly Lisa A. Conte, our president and Chief Executive Officer. The loss of services of any of our key personnel would cause a disruption in our ability to develop our current or future product pipeline and commercialize our products and product candidates. Although we have offer letters with these key members of senior management, such agreements do not prohibit them from resigning at any time. We currently do not maintain "key man" life insurance on any of our senior management team. The loss of Ms. Conte or other members of our current senior management could adversely affect the timing or outcomes of our current and planned studies, as well as the prospects for commercializing our products.
In addition, competition for qualified personnel in the human and animal gastrointestinal health fields is intense, because there are a limited number of individuals who are trained or experienced in the field. We will need to hire additional personnel as we expand our product development and commercialization activities. Even if we are successful in hiring qualified individuals, as we are a growing organization, we do not have a track record for integrating and retaining individuals. If we are not successful in identifying, attracting, integrating or retaining qualified personnel on acceptable terms, or at all, our business will be harmed.
We are dependent on two suppliers for the raw material used to produce the active pharmaceutical ingredient in Mytesi and Canalevia and the botanical extract in Neonorm and Equilevia. The termination of either of these contracts would result in a disruption to product development and our business will be harmed.
The raw material used to manufacture Mytesi, Canalevia, Neonorm and Equilevia is crude plant latex ("CPL"), derived from the Croton lechleri tree, which is found in countries in South America, principally Peru. The ability of our contract suppliers to harvest CPL is governed by the terms of their respective agreements with local government authorities. Although CPL is available from multiple suppliers, we only have contracts with two suppliers to obtain CPL and arrange the shipment to our contract manufacturer. Accordingly, if our contract suppliers do not or are unable to comply with the terms of our respective agreements, and we are not able to negotiate new agreements with alternate suppliers on terms that we deem commercially reasonable, it may harm our business and prospects. The countries from which we obtain CPL could change their laws and regulations regarding the export of the natural products or impose or increase taxes or duties payable by exporters of such products. Restrictions could be imposed on the harvesting of the natural products or additional requirements could be implemented for the replanting and regeneration of the raw material. Such events could have a significant impact on our cost and ability to produce Mytesi, Canalevia, Neonorm, Equilevia and anticipated line extensions.
We are dependent upon third-party contract manufacturers, both for the supply of the active pharmaceutical ingredient in Mytesi and Canalevia and the botanical extract in Neonorm and Equilevia, as well as for the supply of finished products for commercialization.
We have contracted with third parties for the formulation of API and botanical extract into finished products for our studies. We have also entered into memorandums of understanding with Indena S.p.A. for the manufacture of CPL received from our suppliers into the API in Canalevia to support our regulatory filings, as well as the botanical extract in Neonorm and agreed to negotiate a commercial supply agreement. Indena S.p.A. has never manufactured either such ingredient to commercial scale. Glenmark is the current manufacturer of crofelemer, the active API in Canalevia, for Mytesi, and the manufacturer on file for the NDA to which we have a right of reference. As announced in October of 2015, we have entered an agreement with Patheon, a provider of drug
17
development and delivery solutions, under which Patheon provides enteric-coated tablets to us. We also may contract with additional third parties for the formulation and supply of finished products, which we will use in our planned studies and commercialization efforts.
We will be dependent upon our contract manufacturers for the supply of the API in Mytesi and Canalevia. We currently have sufficient quantities of the botanical extract used in Neonorm and Equilevia to support initial commercialization of Neonorm and Equilevia. However, we will require additional quantities of the botanical extract if our ongoing commercialization of Neonorm or our commercial launch of Equilevia is successful. If we are not successful in reaching agreements with third parties on terms that we consider commercially reasonable for manufacturing and formulation, or if our contract manufacturer and formulator are not able to produce sufficient quantities or quality of API, botanical extract or finished product under their agreements, it could delay our plans and harm our business prospects.
The facilities used by our third-party contractors are subject to inspections, including by the FDA, and other regulators, as applicable. We also depend on our third-party contractors to comply with cGMP. If our third-party contractors do not maintain compliance with these strict regulatory requirements, we and they will not be able to secure or maintain regulatory approval for their facilities, which would have an adverse effect on our operations. In addition, in some cases, we also are dependent on our third-party contractors to produce supplies in conformity to our specifications and maintain quality control and quality assurance practices and not to employ disqualified personnel. If the FDA or a comparable foreign regulatory authority does not approve the facilities of our third-party contractors if so required, or if it withdraws any such approval in the future, we may need to find alternative manufacturing or formulation facilities, which could result in delays in our ability to develop or commercialize our human and animal products, if at all. We and our third-party contractors also may be subject to penalties and sanctions from the FDA and other regulatory authorities for any violations of applicable regulatory requirements. The USDA and the European Medicines Agency (the "EMA"), employ different regulatory standards than the FDA, so we may require multiple manufacturing processes and facilities for the same human or animal product candidate or any approved product. We are also exposed to risk if our third-party contractors do not comply with the negotiated terms of our agreements, or if they suffer damage or destruction to their facilities or equipment.
If we are unable to establish sales capabilities on our own or through third parties, we may not be able to market and sell our current or future human or animal products and product candidates, if approved, and generate product or other revenue.
We currently have limited sales, marketing or distribution capabilities, and prior to Napo's launch of Mytesi for the symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy, and our launch of Neonorm for preweaned dairy calves, we had no experience in the sale, marketing and distribution of human or animal health products. There are significant risks involved in building and managing a sales organization, including our potential inability to attract, hire, retain and motivate qualified individuals, generate sufficient sales leads, provide adequate training to sales and marketing personnel and effectively oversee a geographically dispersed sales and marketing team. Any failure or delay in the development of our internal sales, marketing and distribution capabilities and entry into adequate arrangements with distributors or other partners would adversely impact the commercialization of Mytesi, Neonorm, Equilevia and, if approved, Canalevia. If we are not successful in commercializing Mytesi, Neonorm, Equilevia, Canalevia or any of our other line extension products, either on our own or through one or more distributors, or in generating upfront licensing or other fees, we may never generate significant revenue and may continue to incur significant losses, which would harm our financial condition and results of operations.
18
Changes in distribution channels for animal health prescription drugs may make it more difficult or expensive to distribute our animal health prescription drug products.
In the United States, animal owners typically purchase their animal health prescription drugs from their local veterinarians who also prescribe such drugs. There is a trend, however, toward increased purchases of animal health prescription drugs from Internet-based retailers, "big-box" retail stores and other over-the-counter distribution channels, which follows an emerging shift in recent years away from the traditional veterinarian distribution channel. It is also possible that animal owners may come to rely increasingly on Internet-based animal health information rather than on their veterinarians. We currently expect to market our animal health prescription drugs directly to veterinarians, so any reduced reliance on veterinarians by animal owners could harm our business and prospects by making it more difficult or expensive for us to distribute our animal health prescription drug products.
Legislation has been or may be proposed in various states that would require veterinarians to provide animal owners with written prescriptions and disclosures that the animal owner has the right to fill the prescriptions through other means. If enacted, such legislation could lead to a reduction in the number of animal owners who purchase their animal health pharmaceuticals directly from veterinarians, which also could harm our business.
Consolidation of our customers could negatively affect the pricing of our animal health products.
Veterinarians will be our primary customers for our prescription animal health drug products. In recent years, there has been a trend towards the consolidation of veterinary clinics and animal hospitals. If this trend continues, these large clinics and hospitals could attempt to leverage their buying power to obtain favorable pricing from us and other animal health product companies. Any downward pressure on the prices of any of our animal health products could harm our operating results and financial condition.
We will need to increase the size of our organization and may not successfully manage such growth.
As of July 31, 2018, we had 46 employees. Our ability to manage our growth effectively will require us to hire, train, retain, manage and motivate additional employees and to implement and improve our operational, financial and management systems. These demands also may require the hiring of additional senior management personnel or the development of additional expertise by our senior management personnel. If we fail to expand and enhance our operational, financial and management systems in conjunction with our potential future growth, it could harm our business and operating results.
Research and development with respect to our animal health products and product candidates relies on evaluations in animals, which is controversial and may become subject to bans or additional regulations.
The evaluation of our animal health products and product candidates in target animals is required to develop, formulate and commercialize our animal health products and product candidates. Although our animal testing will be subject to GLPs and GCPs, as applicable, animal testing in the human pharmaceutical industry and in other industries continues to be the subject of controversy and adverse publicity. Some organizations and individuals have sought to ban animal testing or encourage the adoption of additional regulations applicable to animal testing. To the extent that such bans or regulations are imposed, our research and development activities with respect to animal health products, and by extension our operating results and financial condition, could be harmed. In addition, negative publicity about animal practices by us or in our industry could harm our reputation among potential customers.
19
If approved, our animal health prescription drug product candidates may be marketed in the United States only in the target animals and for the indications for which they are approved, and if we want to expand the approved animals or indications, it will need to obtain additional approvals, which may not be granted.
If our animal health prescription drug product candidates are approved by regulatory authorities, we may market or advertise them only in the specific species and for treatment of the specific indications for which they were approved, which could limit use of the products by veterinarians and animal owners. Under the Animal Medicinal Drug Use Clarification Act of 1994, veterinarians are permitted to prescribe extra-label uses of certain approved animal drugs and approved human drugs for animals under certain conditions. While veterinarians may in the future prescribe and use human-approved products or use our products for extra-label uses, we may not promote our animal health products for extra-label uses. We note that extra-label uses are uses for which the product has not received approval. If the FDA determines that any of our marketing activities constitute promotion of an extra-label use, we could be subject to regulatory enforcement, including seizure of any misbranded or mislabeled drugs, and civil or criminal penalties, any of which could have an adverse impact on our reputation and expose us to potential liability. We will continue to spend resources ensuring that our promotional claims for our animal health products and product candidates remain compliant with applicable FDA laws and regulations, including materials we post or link to on our website. For example, in 2012, our Chief Executive Officer received an "untitled letter" from the FDA while at Napo regarding preapproval promotion statements constituting misbranding of crofelemer, which was then an investigational drug. These statements were included in archived press releases included on Napo's website. Napo was required to expend time and resources to revise its website to remove the links in order to address the concerns raised in the FDA's letter.
If our human or animal prescription drug product candidates are approved by regulatory authorities, the misuse or extra-label use of such products may harm our reputation or result in financial or other damages.
If our human or animal prescription drug product candidates are approved by regulatory authorities, there may be increased risk of product liability if physicians, veterinarians, patients, animal owners or others, as applicable, attempt to use such products extra-label, including the use of our products for indications or in species for which they have not been approved. Furthermore, the use of an approved human or animal drug for indications other than those indications for which such products have been approved may not be effective, which could harm our reputation and lead to an increased risk of litigation. If we are deemed by a governmental or regulatory agency to have engaged in the promotion of any approved human or animal product for extra-label use, such agency could request that we modify our training or promotional materials and practices and we could be subject to significant fines and penalties, and the imposition of these sanctions could also affect our reputation and position within the gastrointestinal health industry. Any of these events could harm our reputation and our operating results.
We may not maintain the benefits associated with MUMS designation, including market exclusivity.
Although we have received MUMS designation for Canalevia for the treatment of CID in dogs, we may not maintain the benefits associated with MUMS designation. MUMS designation is a status similar to "orphan drug" status for human drugs. When we were granted MUMS designation for Canalevia for the indication of CID in dogs, we became eligible for incentives to support the approval or conditional approval of the designated use. This designation does not allow us to commercialize a product until such time as we obtain approval or conditional approval of the product.
Because Canalevia has received MUMS designation for the identified particular intended use, we are eligible to obtain seven years of exclusive marketing rights upon approval (or conditional approval) of Canalevia for that intended use and become eligible for grants to defray the cost of our clinical work. Each designation that is granted must be unique, i.e., only one designation can be granted for a
20
particular API in a particular dosage form for a particular intended use. The intended use includes both the target species and the disease or condition to be treated.
At some point, we could lose MUMS designation. The basis for a lost designation can include but is not limited to, our failure to engage with due diligence in moving forward with a non-conditional approval, or a competing product has received conditional approval or approval prior to our product candidate for the same indication or species. In addition, MUMS designation may be withdrawn for a variety of reasons such as where the FDA determines that the request for designation was materially defective, or if the manufacturer is unable to assure sufficient quantity of the prescription drug product to meet the needs of animals with the rare disease or condition. If this designation is lost, it could have a negative impact on the product and us, which includes but is not limited to, market exclusivity related to MUMS designation, or eligibility for grants as a result of MUMS designation.
The market for our human or animal products, and the gastrointestinal health market as a whole, is uncertain and may be smaller than we anticipate, which could lead to lower revenue and harm our operating results.
It is very difficult to estimate the commercial potential of any of our human or animal products because the gastrointestinal health market continues to evolve and it is difficult to predict the market potential for our products. The market will depend on important factors such as safety and efficacy compared to other available treatments, changing standards of care, preferences of physicians and veterinarians, as applicable, the willingness of patients and companion and production animal owners, as applicable, to pay for such products, and the availability of competitive alternatives that may emerge either during the product development process or after commercial introduction. If the market potential for our human or animal products is less than we anticipate due to one or more of these factors, it could negatively impact our business, financial condition and results of operations. Further, the willingness of patients and companion and production animal owners to pay for our products may be less than we anticipate, and may be negatively affected by overall economic conditions. Moreover, with respect to our animal health products, the current penetration of animal insurance in the United States is low, animal owners are likely to have to pay out-of-pocket, and such owners may not be willing or able to pay for our products.
Insurance coverage for Mytesi for its current approved indication could decrease or end, or Mytesi might not receive insurance coverage for any approved follow-on indications, which could lead to lower revenue and harm our operating results.
For its current approved indication, Mytesi is currently covered by all of the top 10 commercial insurance plans, representing more than 245 million U.S. lives. In 50% of these plans it is currently on Tier 3 with no restrictions, and in 50% it is currently on Tier 3 with a prior authorization required. In the top 10 Managed Medicare plans, which represent 24 million covered lives, Mytesi is currently covered on 10% of plans. Mytesi is currently covered on Medicaid in all 50 states. However, the nature or extent of coverage for Mytesi by any of these plans or programs could change or be terminated, or Mytesi might not receive insurance coverage for any approved follow-on indications. Either outcome could lead to significantly lower revenue and significantly harm our operating results.
We may engage in future acquisitions that increase our capital requirements, dilute our stockholders, cause us to incur debt or assume contingent liabilities and subject us to other risks.
We may evaluate various strategic transactions, including licensing or acquiring complementary products, technologies or businesses. Any potential acquisitions may entail numerous risks, including increased operating expenses and cash requirements, assimilation of operations and products, retention of key employees, diversion of our management's attention and uncertainties in our ability to maintain key business relationships of the acquired entities. In addition, if we undertake acquisitions, we may
21
issue dilutive securities, assume or incur debt obligations, incur large one-time expenses and acquire intangible assets that could result in significant future amortization expense. Moreover, we may not be able to locate suitable acquisition opportunities and this inability could impair our ability to grow or obtain access to technology or products that may be important to the development of our business.
Certain of the countries in which we plan to commercialize our products in the future are developing countries, some of which have potentially unstable political and economic climates.
We may commercialize our products in jurisdictions that are developing and emerging countries. This may expose us to the impact of political or economic upheaval, and we could be subject to unforeseen administrative or fiscal burdens. At present, we are not insured against the political and economic risks of operating in these countries. Any significant changes to the political or economic climate in any of the developing countries in which we operate or plan to sell products either now or in the future may have a substantial adverse effect on our business, financial condition, trading performance and prospects.
Fluctuations in the exchange rate of foreign currencies could result in currency transactions losses.
As we expand our operations, we expect to be exposed to risks associated with foreign currency exchange rates, since our contract manufacturers are in foreign countries. We will also be further affected by fluctuations in exchange rates in the future to the extent that sales are denominated in currencies other than U.S. dollars. We do not currently employ any hedging or other strategies to minimize this risk, although we may seek to do so in the future.
There are other gastrointestinal-focused human pharmaceutical companies, and we face competition in the marketplaces in which we operate or plan to operate.
Our commercial success in the human drug arena remains dependent on maintaining or establishing a competitive position in the market for the current, approved specialty indication of Mytesi as well as for planned Mytesi follow-on indications. In the IBS-D market in particular, several competitors have commercially available products approved for our planned IBS-D indication. The availability of our competitors' products could limit the demand, and the price we are able to charge, for any drug candidate we develop. The inability to compete with existing or subsequently introduced drug candidates would have a material adverse impact on our business, financial condition and prospects.
Our obligations to CVP are secured by a security interest in substantially all of our veterinary related assets, so if we default on those obligations, CVP could foreclose on our assets.
Our obligations under the secured promissory notes (the "CVP Notes") issued to Chicago Venture Partners, L.P. ("CVP") are secured by a security interest in substantially all of our veterinary related assets, including intellectual property, as provided in the Security Agreement, dated June 29, 2017, between the Company and CVP, the Security Agreement, dated December 8, 2017, between the Company and CVP, the Security Agreement dated February 26, 2018 between the Company and CVP and the Security Agreement dated March 21, 2018 between the Company and CVP. As a result, if we default on our obligations under the loan and security agreement, CVP could foreclose on its security interests and liquidate some or all of these assets, which would harm our veterinary related business, financial condition and results of operations and could require us to reduce or cease operations.
22
Napo's obligations to the holders of the Kingdon Notes are secured by a security interest in substantially all of Napo's assets, so if we default on those obligations, the convertible note holders could foreclose on Napo's assets.
Napo's obligations under the convertible promissory notes (the "Kingdon Notes") issued pursuant to the Amended and Restated Note Purchase Agreement, dated March 31, 2017, in the amount of $10 million, by and among Kingdon Associates, M. Kingdon Offshore Master Fund L.P., Kingdon Family Partnership, L.P. and Kingdon Credit Master Fund L.P. (collectively, the "Kingdon Purchasers") and Napo and the related transaction documents are secured by a security interest in substantially all of Napo's assets, including Napo intellectual property. As a result, if we default under our obligations under the Kingdon Notes or the transaction documents, the holders of such Kingdon Notes, acting through their appointed agent, could foreclose on their security interests and liquidate some or all of these assets, which would harm our business, financial condition and results of operations and could require us to reduce or cease operations.
Risks Related to Intellectual Property
We cannot be certain that our patent strategy will be effective to protect against competition
Our commercial success depends in large part on obtaining and maintaining patent, trademark and trade secret and other exclusivity protection of our human or animal products, both prescription and non-prescription, our current human or animal product candidates and any future human or animal product candidates, and their respective components, formulations, methods used to manufacture them and methods of treatment, as well as successfully defending our patents and other intellectual property rights against third-party challenges. Our ability to stop unauthorized third parties from making, using, selling, offering to sell or importing our products or our product candidates is dependent upon the extent to which we have rights under valid and enforceable patents, trade secrets and other similar intellectual property that cover these activities. The patent prosecution process is expensive and time-consuming, and we may not be able to prepare, file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of inventions made in the course of development and commercialization activities in time to obtain patent protection on them.
We have a portfolio of United States and foreign issued patents and pending applications related to our products and product candidates. We have five issued United States patents listed in the FDA's Orange Book for Mytesi of which three are in force, as the expiration date of US 7,341,744, has been adjusted to 2019. We plan to rely on US 7,341,744, as protection for Canalevia. We plan to rely on certain of these issued patents as protection for Canalevia. The strength of patents in the field of pharmaceuticals and animal health involves complex legal and scientific questions and can be uncertain. We cannot be certain that pending applications will issue as patents. For those patents that are already issued and even if other patents do successfully issue, third parties may challenge their validity, enforceability or scope, which may result in such patents being narrowed, invalidated or held unenforceable. Furthermore, even if they are unchallenged, our patents may not adequately protect our intellectual property or prevent others from designing around their claims. If the patents we have are not maintained or their scope is significantly narrowed or if we are not able to obtain issued patents from pending applications, our business and prospects would be harmed.
Recent patent reform legislation could increase the uncertainties and costs surrounding the prosecution of any patent applications and the enforcement or defense of any patents that issue. On September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications are prosecuted, redefine prior art, may affect patent litigation, and switch the U.S. patent system from a "first-to-invent" system to a "first-to-file" system.
23
Under a "first-to-file" system, assuming the other requirements for patentability are met, the first inventor to file a patent application generally will be entitled to the patent on an invention regardless of whether another inventor had made the invention earlier. The USPTO has developed new regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act, and in particular, the first-to-file provisions, became effective on March 16, 2013. Among some of the other changes to the patent laws are changes that limit where a patentee may file a patent infringement suit and that provide opportunities for third parties to challenge any issued patent in the USPTO. The Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our patents and any other patents that issue, all of which could harm our business and financial condition.
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic maintenance and annuity fees on any issued patent and, in certain jurisdictions, pending applications, are due to be paid to the USPTO and foreign patent agencies in several stages over the lifetime of the patent. The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a patent or patent application include failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. If we fail to maintain the patents and patent applications covering our prescription drug products, prescription drug product candidates and non-prescription products, our competitors might be able to enter the market, which would harm our business.
Third parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, which would be costly, time-consuming and, if successfully asserted against us, delay or prevent the development and commercialization of our current or future products and product candidates.
Our research, development and commercialization activities may infringe or otherwise violate or be claimed to infringe or otherwise violate patents owned or controlled by other parties. There may be patents already issued of which we are unaware that might be infringed by a product or one of our current or future prescription drug product candidates or non-prescription products. Moreover, it is also possible that patents may exist that we are aware of, but that we do not believe are relevant to our current or future prescription drug product candidates or non-prescription products, which could nevertheless be found to block our freedom to market these products. Because patent applications can take many years to issue and may be confidential for 18 months or more after filing, there may be applications now pending of which we are unaware and which may later result in issued patents that may be infringed by our current or future prescription drug product candidates or non-prescription products. We cannot be certain that our products, current or future prescription drug product candidates or non-prescription products will not infringe these or other existing or future third-party patents. In addition, third parties may obtain patents in the future and claim that use of our technologies infringes upon these patents.
To the extent we become subject to future third-party claims against us or our collaborators, we could incur substantial expenses and, if any such claims are successful, we could be liable to pay substantial damages, including treble damages and attorney's fees if we or our collaborators are found
24
to be willfully infringing a third party's patents. If a patent infringement suit were brought against us or our collaborators, we or they could be forced to stop or delay research, development, manufacturing or sales of the human or animal prescription drug or non-prescription product that is the subject of the suit. Even if we are successful in defending such claims, infringement and other intellectual property claims can be expensive and time-consuming to litigate and divert management's attention from our business and operations. As a result of or in order to avoid potential patent infringement claims, we or our collaborators may be compelled to seek a license from a third party for which we would be required to pay license fees or royalties, or both. Moreover, these licenses may not be available on acceptable terms, or at all. Even if we or our collaborators were able to obtain such a license, the rights may be nonexclusive, which could allow our competitors access to the same intellectual property. Any of these events could harm our business and prospects.
Our proprietary position depends upon patents that are formulation or method-of-use patents, which do not prevent a competitor from using the same human or animal drug for another use.
Composition-of-matter patents on the API in prescription drug products are generally considered to be the strongest form of intellectual property protection because such patents provide protection without regard to any particular method of use or manufacture or formulation of the API used. The composition-of-matter patents for crofelemer, the API in Mytesi and Canalevia, have expired, and the issued patents and applications relevant to our products and product candidates cover formulations and methods of use for crofelemer.
Method-of-use patents protect the use of a product for the specified method and formulation patents cover formulations of the API or botanical extract. These types of patents do not prevent a competitor from developing or marketing an identical product for an indication that is outside the scope of the patented method or from developing a different formulation that is outside the scope of the patented formulation. Moreover, with respect to method-of-use patents, even if competitors do not actively promote their product for our targeted indications or uses for which we may obtain patents, physicians may recommend that patients use our products extra-label, and veterinarians may recommend that animal owners use these products extra-label, or animal owners may do so themselves. Although extra-label use may infringe or contribute to the infringement of method-of-use patents, the practice is common and such infringement is difficult to prevent or prosecute.
We may be involved in lawsuits to protect or enforce our patents, which could be expensive, time-consuming and unsuccessful, and third parties may challenge the validity or enforceability of our patents and they may be successful.
We intend to rely upon a combination of regulatory exclusivity periods, patents, trade secret protection, and confidentiality agreements to protect the intellectual property related to Mytesi, our current prescription drug product candidates, non-prescription products and our development programs.
If the breadth or strength of protection provided by any patents, patent applications or future patents we may own, license, or pursue with respect to any of our current or future product candidates or products is threatened, it could threaten our ability to commercialize any of our current or future human or animal product candidates or products. Further, if we encounter delays in our development efforts, the period of time during which we could market any of our current or future product candidates or products under any patent protection we obtain would be reduced.
Given the amount of time required for the development, testing and regulatory review of new product candidates or products, patents protecting such candidates might expire before or shortly after such product candidates or products are commercialized. Patent term extension has been applied for US 7,341,744 to account for regulatory delays in obtaining human marketing approval for crofelemer. The FDA and the USPTO have confirmed that US 7,341,744 is eligible for an extension of 1075 days
25
and we await issuance of the patent term extension certificate. With respect to requests for patent term extensions, the applicable authorities, including the USPTO and the FDA, and any equivalent regulatory authority in other countries, may not agree with our assessment of whether such extensions are available, and may refuse to grant extensions to patents, or may grant more limited extensions than requested. If this occurs, our competitors may take advantage of our investment in development and trials by referencing our clinical and preclinical data and launch their product earlier than might otherwise be the case.
Even where laws provide protection or we are able to obtain patents, costly and time-consuming litigation may be necessary to enforce and determine the scope of our proprietary rights, and the outcome of such litigation would be uncertain. Moreover, any actions we may bring to enforce our intellectual property against our competitors could provoke them to bring counterclaims against us, and some of our competitors have substantially greater intellectual property portfolios than we have. To counter infringement or unauthorized use of any patents we may obtain, we may be required to file infringement claims, which can be expensive and time-consuming to litigate. In addition, if we or one of our future collaborators were to initiate legal proceedings against a third party to enforce a patent covering one of our products, current product candidates, or one of our future products, the defendant could counterclaim that the patent is invalid or unenforceable. In patent litigation in the United States, defendant counterclaims alleging invalidity or unenforceability are commonplace and challenges to validity of patents in certain foreign jurisdictions is common as well. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness, non-enablement or lack of statutory subject matter. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant material information from the USPTO, or made a materially misleading statement, during prosecution. Under the Hatch-Waxman Act, a competitor seeking to market a generic form of Mytesi before the expiration of any of the patents listed in the FDA's Orange Book for Mytesi could file (and could have filed after December 31, 2016) an ANDA with a certification under 21 U.S.C. § 3559j)(2)(A)(iv) that each of these patents (except for those which the ANDA filer states it will market only after its expiration) is either invalid, unenforceable or not infringed. We may assert the patents in Hatch-Waxman litigation against the party filing the ANDA to keep the competing product off of the market until the patents expire but there is a risk that we will not succeed. The party filing the ANDA may also counterclaim in the litigation that the patents are not valid or unenforceable, and the court may find one or more claims of our patents invalid or unenforceable. If this occurs, a competing generic product could be marketed prior to expiration of our patents listed in the Orange Book, which would harm our business.
Third parties may also raise similar validity claims before the USPTO in post-grant proceedings such as ex parte reexaminations, inter partes review, or post-grant review, or oppositions or similar proceedings outside the United States, in parallel with litigation or even outside the context of litigation. The outcome following legal assertions of invalidity and unenforceability is unpredictable. If a defendant were to prevail on a legal assertion of invalidity or unenforceability, we would lose at least part, and perhaps all, of any future patent protection on one or more of our products or our current or future product candidates. Such a loss of patent protection could harm our business. We cannot be certain that there is no invalidating prior art, of which we and the patent examiner were unaware during prosecution or other basis for a finding of invalidity. Litigation proceedings may fail and, even if successful, may result in substantial costs and distract our management and other employees. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. In addition, there could be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be unsuccessful, it could have an adverse effect on the price of our common stock. Finally, we may not be able to prevent, misappropriation of our trade secrets or confidential
26
information, particularly in countries where the laws may not protect those rights as fully as in the United States.
If we are unable to prevent disclosure of our trade secrets or other confidential information to third parties, our competitive position may be impaired.
We also rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable or for which we have not filed patent applications, processes for which patents are difficult to enforce and other elements of our product development processes that involve proprietary know-how, information or technology that is not covered by patents. Although we require all of our employees to assign their inventions to us, and endeavor to execute confidentiality agreements with all of our employees, consultants, advisors and any third parties who have access to our proprietary know-how, information or technology, we cannot be certain that we have executed such agreements with all parties who may have helped to develop our intellectual property or had access to our proprietary information, or that our agreements will not be breached. We cannot guarantee that our trade secrets and other confidential proprietary information will not be disclosed or that competitors will not otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. If we are unable to prevent disclosure of our intellectual property to third parties, we may not be able to maintain a competitive advantage in our market, which would harm our business.
Any disclosure to or misappropriation by third parties of our confidential proprietary information could enable competitors to quickly duplicate or surpass our technological achievements, and erode our competitive position in our market.
Changes in U.S. patent law could diminish the value of patents in general, thereby impairing our ability to protect our products.
As is the case with other human or animal pharmaceutical product companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the human and animal health industries involves both technological and legal complexity. Therefore, obtaining and enforcing patents is costly, time-consuming and inherently uncertain. In addition, the United States has recently enacted and implemented wide-ranging patent reform legislation. The U.S. Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. In addition to increasing uncertainty with regard to our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by the U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce patents that we have licensed or that we might obtain in the future.
We may not be able to protect our intellectual property rights throughout the world, which could impair our business.
Filing, prosecuting and defending patents on human and animal drug products, product candidates and non-prescription products throughout the world would be prohibitively expensive. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories where we may obtain patent protection, but where patent enforcement is not as strong as that in the United States. These products may compete with our products in jurisdictions where we do not have any issued or licensed patents and any future patent claims or other intellectual property rights may not be effective or sufficient to prevent them from so competing.
27
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to animal health products, which could make it difficult for us to stop the infringement of our future patents, if any, or patents we have in licensed, or marketing of competing products in violation of our proprietary rights generally. Further, the laws of some foreign countries do not protect proprietary rights to the same extent or in the same manner as the laws of the United States. As a result, we may encounter significant problems in protecting and defending our intellectual property both in the United States and abroad. Proceedings to enforce our future patent rights, if any, in foreign jurisdictions could result in substantial cost and divert our efforts and attention from other aspects of our business.
Our business could be harmed if we fail to obtain certain registered trademarks in the United States or in other countries.
Our registered and pending U.S. trademarks include NEONORM®, MYTESI®, NAPO®, Napo Logo®, CANALEVIA, EQUILEVIA, JAGUAR ANIMAL HEALTH, and the Jaguar Animal Health logo. We also own pending applications for the CANALEVIA mark in a number of foreign countries. We have not yet filed applications for our company name or our logo in the U.S. During trademark registration proceedings, we may receive rejections of our trademark applications. If so, we will have an opportunity to respond, but we may be unable to overcome such rejections. In addition, the USPTO and comparable agencies in many foreign jurisdictions may permit third parties to oppose pending trademark applications and to seek to cancel registered trademarks. If opposition or cancellation proceedings are filed against any of our trademark applications or any registered trademarks, our trademarks may not survive such proceedings. Moreover, any name we propose to use with our prescription drug product candidates in the United States, including CANALEVIA, must be approved by the FDA, regardless of whether we have registered or applied to register as a trademark. The FDA typically conducts a review of proposed prescription drug product names, including an evaluation of potential for confusion with other product names. If the FDA objects to any of our proposed proprietary product names, we may be required to expend significant additional resources in an effort to identify a suitable substitute name that would qualify under applicable trademark laws, not infringe the existing rights of third parties and be acceptable to the FDA.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties.
We have received confidential and proprietary information from third parties. In addition, we employ individuals who were previously employed at other biotechnology, pharmaceutical or animal health companies. We may be subject to claims that we or our employees, consultants or independent contractors have inadvertently or otherwise improperly used or disclosed confidential information of these third parties or our employees' former employers. Litigation may be necessary to defend against any such claims. Even if we were successful in defending against any such claims, such litigation could result in substantial cost and be a distraction to our management and employees.
Risks Related to Government Regulation
Even if we receive any of the required regulatory approvals for our current or future prescription drug product candidates and non-prescription products, we will be subject to ongoing obligations and continued regulatory review, which may result in significant additional expense.
If the FDA or any other regulatory body approves any of our current or future prescription drug product candidates, or if necessary, our non-prescription products, the manufacturing processes, clinical development, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion
28
and recordkeeping for the product may be subject to extensive and ongoing regulatory requirements. These requirements could include, but are not limited to, submissions of efficacy and safety and other post-marketing information and reports, establishment registration, and product listing, compliance with new rules promulgated under the FSMA, as well as continued compliance with cGMP, GLP and GCP for any studies that we conduct post-approval. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with our contract manufacturers or manufacturing processes, or failure to comply with regulatory requirements, are reportable events to the FDA and may result in, among other things:
- •
- restrictions on the marketing or manufacturing of the product, withdrawal of the product from the market, revised labeling, or voluntary or
involuntary product recalls;
- •
- additional clinical studies, fines, warning letters or holds on target animal studies;
- •
- refusal by the FDA, or other regulators to approve pending applications or supplements to approved applications filed by us or our strategic
collaborators related to the unknown problems, or suspension or revocation of the problematic product's license approvals;
- •
- product seizure or detention, or refusal to permit the import or export of products; and
- •
- injunctions and/or the imposition of civil or criminal penalties.
The FDA or other regulatory agency's policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates or require certain changes to the labeling or additional clinical work concerning safety and efficacy of the product candidates. We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the United States or abroad. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained and we may not achieve or sustain profitability, which would harm our business. In addition, failure to comply with these regulatory requirements could result in significant penalties.
In addition, from time to time, we may enter into consulting and other financial arrangements with veterinarians, who prescribe or recommend our products, once approved. As a result, we may be subject to state, federal and foreign healthcare and/or veterinary medicine laws. If our financial relationships with veterinarians are found to be in violation of such laws that apply to us, we may be subject to penalties.
The issuance by the FDA of protocol concurrences for our pivotal studies does not guarantee ultimate approval of our NADA.
A pivotal study protocol is submitted to the FDA by a drug sponsor for purposes of obtaining FDA review of the protocol. Prior FDA review of the protocol for a pivotal study makes it more likely that the study design will generate information the sponsor needs to demonstrate to the satisfaction of the FDA whether the drug is safe and effective for its intended use. It creates an expectation by the sponsor that the FDA should not later alter its perspectives on these issues unless public or animal health concerns appear that were not recognized at the time of protocol assessment. Even if the FDA issues a protocol concurrence, ultimate approval of an NADA by the FDA is not guaranteed because a final determination that the agreed-upon protocol satisfies a specific objective, such as the demonstration of efficacy, or supports an approval decision, will be based on a complete review of all the data submitted to the FDA including the outcome of the study for which protocol concurrence was received.
29
Any of our current or future prescription drug product candidates or non-prescription products may cause or contribute to adverse medical events that we would be required to report to regulatory authorities and, if we fail to do so, we could be subject to sanctions that would harm our business.
If we are successful in commercializing any of our current or future prescription drug product candidates or non-prescription products, certain regulatory authorities will require that we report certain information about adverse medical events if those products may have caused or contributed to those adverse events. The timing of our obligation to report would be triggered by the date we become aware of the adverse event as well as the nature of the event. We may fail to report adverse events we become aware of within the prescribed timeframe. We may also fail to appreciate that we have become aware of a reportable adverse event, especially if such event is not reported to us as an adverse event or if it is an adverse event that is unexpected or removed in time from the use of our products. If we fail to comply with our reporting obligations, the regulatory authorities could take action including, but not limited to, criminal prosecution, seizure of our products, facility inspections, removal of our products from the market, recalls of certain lots or batches, or cause a delay in approval or clearance of future products.
Legislative or regulatory reforms with respect to animal health may make it more difficult and costly for us to obtain regulatory clearance or approval of any of our current or future product candidates and to produce, market, and distribute our products after clearance or approval is obtained.
From time to time, legislation is drafted and introduced in the U.S. Congress or other jurisdictions in which we intend to operate that could significantly change the statutory provisions governing the testing, regulatory clearance or approval, manufacture, and marketing of regulated products. In addition, the FDA's regulations and guidance are often revised or reinterpreted by the FDA and such other regulators in ways that may significantly affect our business and our products and product candidates. Similar changes in laws or regulations can occur in other countries. Any new regulations or revisions or reinterpretations of existing regulations in the United States or in other countries may impose additional costs or lengthen review times of any of our current or future products and product candidates. We cannot determine what effect changes in regulations, statutes, legal interpretation or policies, when and if promulgated, enacted or adopted may have on our business in the future. Such changes could, among other things, require:
- •
- changes to manufacturing methods;
- •
- additional clinical trials or testing;
- •
- new requirements related to approval to enter the market;
- •
- recall, replacement, or discontinuance of certain products; and
- •
- additional record keeping or the development of certain regulatory required hazard identification plans.
Each of these would likely entail substantial time and cost and could harm our financial results. In addition, delays in receipt of or failure to receive regulatory clearances or approvals for any future products would harm our business, financial condition, and results of operations.
We believe that our non-prescription products are not subject to regulation by regulatory agencies in the United States, but there is a risk that regulatory bodies may disagree with our interpretation, or may redefine the scope of their regulatory reach in the future, which would result in additional expense and could delay or prevent the commercialization of these products.
The FDA retains jurisdiction over all animal prescription drug products however, in many instances, the Federal Trade Commission will exercise primary or concurrent jurisdiction with FDA on
30
non-prescription products as to post marketing claims made regarding the product. On April 22, 1996, the FDA published a statement in the Federal Register, 61 FR 17706, that it believes that the Dietary Supplement and Health Education Act ("DSHEA"), does not apply to animal health supplement products, such as our non-prescription products. Accordingly, the FDA's Center for Veterinary Medicine only regulates those animal supplements that fall within the FDA's definition of an animal drug, animal food or animal feed additive. The Federal Food Drug and Cosmetic Act defines food as "articles used for food or drink for man or other animals and articles used as components of any such article." Animal foods are not subject to pre-market approval and are designed to provide a nutritive purpose to the animals that receive them. Feed additives are defined as those articles that are added to an animal's feed or water as illustrated by the guidance documents. Our non-prescription products are not added to food, are not ingredients in food nor are they added to any animal's drinking water. Therefore, our non-prescription products do not fall within the definition of a food or feed additive. In light of the pronouncement by the FDA that the DSHEA was not intended to apply to animals, the FDA seeks to regulate such supplements as food or food additives depending on the intended use of the product. The intended use is demonstrated by how the article is included in a food, or added to the animals' intake (i.e., through its drinking water). If the intended use of the product does not fall within the proscribed use making the product a food, it cannot be regulated as a food. There is no intent to make our non-prescription products a component of an animal food, either directly or indirectly. A feed additive is a product that is added to a feed for any reason including the top dressing of an already prepared feed. Some additives, such as certain forage, are deemed to be Generally Recognized as Safe, or GRAS, and therefore, not subject to a feed Additive Petition approval prior to use. However, the substances deemed GRAS are generally those that are recognized as providing nutrients as a food does. We do not believe that our non-prescription products fit within this framework either. Finally, a new animal drug refers to drugs intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease in animals. Our non-prescription Neonorm Foal and Neonorm Calf products are chemically distinct from crofelemer and not intended to diagnose, cure, mitigate, treat or prevent disease and therefore, do not fit within the definition of an animal drug. Additionally, because a previously marketed human formulation of the botanical extract in our non-prescription products was regulated as a human dietary supplement subject to the DSHEA (and not regulated as a drug by the FDA), we do not believe that the FDA would regulate the animal formulation used in our non-prescription products in a different manner. We do not believe that our non-prescription products fit the definition of an animal drug, food or food additive and therefore are not regulated by the FDA at this time.
However, despite many such unregulated animal supplements currently on the market, the FDA may choose in the future to exercise jurisdiction over animal supplement products in which case, we may be subject to new animal drug regulations thereby inhibiting our ability to launch or to continue marketing our non-prescription products. In the past, the FDA has redefined or attempted to redefine some non-prescription non-feed products as falling within the definition of drug, feed or feed additive and therefore subjected those products to the relevant regulations. We have not discussed with the FDA its belief that the FDA currently does not exercise jurisdiction over our non-prescription products. Should the FDA assert regulatory authority over our non-prescription products, we would take commercially reasonable steps to address the FDA's concerns, potentially including but not limited to, seeking registration for such products, reformulating such products to further distance such products from regulatory control, or ceasing sale of such products. Further, the Animal and Plant Health Inspection Service, an agency of the USDA, may at some point choose to exercise jurisdiction over certain non-prescription products that are not intended for production animals. We do not believe we are currently subject to such regulation, but could be in the future. If the FDA or other regulatory agencies, such as the USDA, try to regulate our non-prescription products, we could be required to seek regulatory approval for our non-prescription products, which would result in additional expense and could delay or prevent the commercialization of these products.
31
Even if Napo receives the required regulatory approvals for Napo's current or future prescription drug product candidates and non-prescription products, Napo will be subject to ongoing obligations and continued regulatory review, which may result in significant additional expense.
If the FDA or any other regulatory body approves any of Napo's current or future prescription drug product candidates, or if necessary, Napo's non-prescription products, the manufacturing processes, clinical development, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion and recordkeeping for the product is subject to extensive and ongoing regulatory requirements. These requirements could include, but are not limited to, submissions of efficacy and safety and other post-marketing information and reports, establishment registration, and product listing, compliance with new rules promulgated under the FSMA, as well as continued compliance with cGMP, GLP and GCP for any studies that Napo conducts post-approval. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with Napo's contract manufacturers or manufacturing processes, or failure to comply with regulatory requirements, are reportable events to the FDA and may result in, among other things:
- •
- restrictions on the marketing or manufacturing of the product, withdrawal of the product from the market, revised labeling, or voluntary or
involuntary product recalls;
- •
- additional clinical studies fines, warning letters or holds on studies;
- •
- refusal by the FDA, or other regulators to approve pending applications or supplements to approved applications filed by Napo or Napo's
strategic collaborators related to the unknown problems, or suspension or revocation of the problematic product's license approvals;
- •
- product seizure or detention, or refusal to permit the import or export of products; and
- •
- injunctions or the imposition of civil or criminal penalties.
The FDA or other regulatory agency's policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of Napo's product candidates or require certain changes to the labeling or require additional clinical work concerning safety and efficacy of the product candidates. Napo cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the United States or abroad. If Napo is slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if Napo is not able to maintain regulatory compliance, Napo may lose any marketing approval that Napo may have obtained and Napo may not achieve or sustain profitability, which would harm Napo's business. In addition, failure to comply with these regulatory requirements could result in significant penalties.
In addition, from time to time, Napo may enter into consulting and other financial arrangements with physicians and other healthcare practitioners, who prescribe or recommend Napo's products, once approved. As a result, Napo may be subject to state, federal and foreign healthcare laws, including but not limited to anti-kickback laws. If Napo's financial relationships with physicians or other healthcare practitioners are found to be in violation of such laws that apply to Napo, Napo may be subject to penalties.
Any of Napo's current or future prescription drug product candidates may cause or contribute to adverse medical events that Napo would be required to report to regulatory authorities and, if Napo fails to do so, Napo could be subject to sanctions that would harm Napo's business.
If Napo is successful in commercializing any of Napo's current or future prescription drug product candidates, certain regulatory authorities will require that Napo report certain information about adverse medical events if those products may have caused or contributed to those adverse events. The timing of Napo's obligation to report would be triggered by the date Napo becomes aware of the
32
adverse event as well as the nature of the event. Napo may fail to report adverse events Napo becomes aware of within the prescribed timeframe. Napo may also fail to appreciate that Napo has become aware of a reportable adverse event, especially if it is not reported to Napo as an adverse event or if it is an adverse event that is unexpected or removed in time from the use of Napo's products. If Napo fails to comply with Napo's reporting obligations, the regulatory authorities could take action including, but not limited to, criminal prosecution, seizure of Napo's products, facility inspections, removal of Napo's products from the market, recalls of certain lots or batches, or cause a delay in approval or clearance of future products.
Legislative or regulatory reforms make it more difficult and costly for Napo to obtain regulatory clearance or approval of any of Napo's current or future product candidates and to produce, market, and distribute Napo's products after clearance or approval is obtained.
From time to time, legislation is drafted and introduced in the U.S. Congress or other jurisdictions in which Napo intends to operate that could significantly change the statutory provisions governing the testing, regulatory clearance or approval, manufacture, and marketing of regulated products. In addition, the FDA's regulations and guidance are often revised or reinterpreted by the FDA and such other regulators in ways that may significantly affect Napo's business and Napo's products and product candidates. Similar changes in laws or regulations can occur in other countries. Any new regulations or revisions or reinterpretations of existing regulations in the United States or in other countries may impose additional costs or lengthen review times of any of Napo's current or future products and product candidates. Napo cannot determine what effect changes in regulations, statutes, legal interpretation or policies, when and if promulgated, enacted or adopted may have on Napo's business in the future. Such changes could, among other things, require:
- •
- changes to manufacturing methods;
- •
- additional clinical trials or testing;
- •
- new requirements related to approval to enter the market;
- •
- recall, replacement, or discontinuance of certain products; and
- •
- additional record keeping or the development of certain regulatory required hazard identification plans.
Each of these would likely entail substantial time and cost and could harm Napo's financial results. In addition, delays in receipt of or failure to receive regulatory clearances or approvals for any future products would harm Napo's business, financial condition, and results of operations.
We and our employees and contractors are subject, directly or indirectly, to federal, state and foreign healthcare fraud and abuse laws, including false claims laws. If we are unable to comply, or have not fully complied, with such laws, we could face substantial penalties.
Our operations are subject to various federal, state and foreign fraud and abuse laws. These laws may constrain our operations, including the financial arrangements and relationships through which we market, sell and distribute our products.
U.S. federal and state laws that affect our ability to operate include, but are not limited to:
- •
- The federal Anti-Kickback Statute, which prohibits, among other things, persons or entities from knowingly and willfully soliciting, receiving, offering or paying any remuneration (including any kickback, bribe, or rebate), directly or indirectly, overtly or covertly, in cash or in kind in return for, the purchase, recommendation, leasing or furnishing of an item or service reimbursable under a federal healthcare program, such as the Medicare or Medicaid programs;
33
- •
- Federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from
knowingly presenting, or causing to be presented, claims for payment or approval from Medicare, Medicaid, or other government payers that are false or fraudulent;
- •
- Section 242 of HIPAA codified at 18 U.S.C. § 1347, which created new federal criminal statutes that prohibit a person
from knowingly and willfully executing a scheme or from making false or fraudulent statements to defraud any healthcare benefit program (i.e., public or private);
- •
- Federal transparency laws, including the so-called federal "sunshine" law, which requires the tracking and disclosure to the federal government
by pharmaceutical manufacturers of direct and indirect payments and other transfers of value to physicians and teaching hospitals as well as ownership and investment interests that are held by
physicians and their immediate family members; and
- •
- State law equivalents of each of these federal laws, such as anti-kickback and false claim laws that may apply to items or services reimbursed by any third-party payer, including commercial insurers; state laws that require pharmaceutical companies to comply with their industry's voluntary compliance guidelines and the applicable compliance guidance promulgated by the federal government or otherwise restrict certain payments that may be made to healthcare providers and other potential referral sources; state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures; state laws that prohibit giving gifts to licensed healthcare professionals; and state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts in certain circumstances, such as specific disease states.
In particular, activities and arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, waste, and other abusive practices. These laws and regulations may restrict or prohibit a wide range of activities or other arrangements related to the development, marketing or promotion of products, including pricing and discounting of products, provision of customer incentives, provision of reimbursement support, other customer support services, provision of sales commissions or other incentives to employees and independent contractors and other interactions with healthcare practitioners, other healthcare providers and patients.
Because of the breadth of these laws and the narrow scope of the statutory or regulatory exceptions and safe harbors available, our business activities could be challenged under one or more of these laws. Relationships between pharmaceutical manufacturers and health care providers are an area of heightened scrutiny by the government. We engage in various activities, including the frequent conduct of speaker programs to educate physicians, the provision of reimbursement advice and support to customers, and the provision of customer and patient support services, that have been the subject of government scrutiny and enforcement action within the pharmaceutical industry. Government expectations and industry best practices for compliance continue to evolve and past activities may not always be consistent with current industry best practices. Further, there is a lack of government guidance as to whether various industry practices comply with these laws, and government interpretations of these laws continue to evolve, all of which creates compliance uncertainties. Any non-compliance could result in regulatory sanctions, criminal or civil liability and serious harm to our reputation. Although we have a compliance program designed to ensure that our employees' and commercial partners' activities and interactions with healthcare professionals and patients are appropriate, ethical, and consistent with all applicable laws, regulations, guidelines, policies and standards, it is not always possible to identify and deter misconduct, and the precautions we take to detect and prevent this activity may not be effective in preventing such conduct, mitigating risks, or
34
reducing the chance of governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations.
If a government entity opens an investigation into possible violations of any of these laws (which may include the issuance of subpoenas), we would have to expend significant resources to defend ourselves against the allegations. Allegations that we, our officers, or our employees violated any one of these laws can be made by individuals called "whistleblowers" who may be our employees, customers, competitors or other parties. Government policy is to encourage individuals to become whistleblowers and file a complaint in federal court alleging wrongful conduct. The government is required to investigate all of these complaints and decide whether to intervene. If the government intervenes and we are required to pay money back to the government, the whistleblower, as a reward, is awarded a percentage. If the government declines to intervene, the whistleblower may proceed on her own and, if she is successful, she will receive a percentage of any judgment or settlement amount the company is required to pay. The government may also initiate an investigation on its own. If any such actions are instituted against us, those actions could have a significant impact on our business, including the imposition of significant fines, and other sanctions that may materially impair our ability to run a profitable business. In particular, if our operations are found to be in violation of any of the laws described above or if we agree to settle with the government without admitting to any wrongful conduct or if we are found to be in violation of any other governmental regulations that apply to us, we, our officers and employees may be subject to sanctions, including civil and criminal penalties, damages, fines, exclusion from participation in government health care programs, such as Medicare and Medicaid, imprisonment, the curtailment or restructuring of our operations and the imposition of a corporate integrity agreement, any of which could adversely affect our business, results of operations and financial condition.
Risks Related to Our Common Stock
Our failure to meet the continued listing requirements of The NASDAQ Capital Market could result in a delisting of our common stock.
Our common stock is listed on The NASDAQ Capital Market, which imposes, among other requirements a minimum bid requirement. The closing bid price for our common stock must remain at or above $1.00 per share to comply with NASDAQ's minimum bid requirement for continued listing. If the closing bid price for our common stock is less than $1.00 per share for 30 consecutive business days, NASDAQ may send us a notice stating that we will be provided a period of 180 days to regain compliance with the minimum bid requirement or else NASDAQ may make a determination to delist our common stock.
The delisting of our common stock from NASDAQ may make it more difficult for us to raise capital on favorable terms in the future. Such a delisting would likely have a negative effect on the price of our common stock and would impair your ability to sell or purchase our common stock when you wish to do so. Further, if we were to be delisted from The NASDAQ Capital Market, our common stock would cease to be recognized as covered securities and we would be subject to regulation in each state in which it offers its securities.
35
We have a material weakness in our internal control over financial reporting related to the accounting for income taxes, and if we fail to remediate the material weakness, or experience any additional material weaknesses in the future or otherwise fail to maintain an effective system of internal controls in the future, we may not be able to accurately report our financial condition or results of operations which may adversely affect investor confidence in us and, as a result, the value of our common stock.
Our management is responsible for establishing and maintaining adequate internal control over our financial reporting, as defined in Rule 13a-15(f) under the Securities Exchange Act of 1934, as amended (the "Exchange Act").
Preparing our consolidated financial statements involves a number of complex manual and automated processes, which are dependent upon individual data input or review and require significant management judgment. One or more of these elements may result in errors that may not be detected and could result in a material misstatement of our consolidated financial statements. If we fail to maintain the adequacy of our internal controls over financial reporting, our business and operating results may be harmed and we may fail to meet our financial reporting obligations. If material weaknesses in our internal control are discovered or occur, our consolidated financial statements may contain material misstatements and we could be required to restate our financial results.
In connection with the audit of our financial statements as of and for the year ended December 31, 2017, we identified a material weakness in our internal control over financial reporting, as defined in the standards established by the Public Company Accounting Oversight Board of the United States. Our management has determined that we had a material weakness in our internal control over financial reporting as of December 31, 2017 because we did not adequately and timely review the accounting for income taxes. While the Company utilizes the assistance of an external income tax specialist to prepare its annual tax provision, management has concluded there to be a material weakness in the design of the Company's income tax controls in that the Company's policy that governs the data validation controls over data provided to and received from the external income tax specialist and the management review controls were not designed with appropriate levels of precision and were not undertaken in a timely manner.
We are enhancing our internal controls, processes and related documentation necessary to remediate our material weakness. We may not be able to complete our remediation, evaluation and testing in a timely fashion. If we are unable to remediate this material weakness, or if we identify one or more other material weaknesses in our internal control over financial reporting, we will continue to be unable to conclude that our internal controls are effective. If we are unable to confirm that our internal control over financial reporting is effective we could lose investor confidence in the accuracy and completeness of our financial reports, which could cause the price of our common stock to decline.
If our shares become subject to the penny stock rules, it would become more difficult to trade our shares.
The SEC has adopted rules that regulate broker-dealer practices in connection with transactions in penny stocks. Penny stocks are generally equity securities with a price of less than $5.00, other than securities registered on certain national securities exchanges or authorized for quotation on certain automated quotation systems, provided that current price and volume information with respect to transactions in such securities is provided by the exchange or system. If we do not retain a listing on The NASDAQ Capital Market and if the price of our common stock is less than $5.00, our common stock will be deemed a penny stock. The penny stock rules require a broker-dealer, before a transaction in a penny stock not otherwise exempt from those rules, to deliver a standardized risk disclosure document containing specified information. In addition, the penny stock rules require that before effecting any transaction in a penny stock not otherwise exempt from those rules, a broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive (i) the purchaser's written acknowledgment of the receipt of a risk disclosure statement;
36
(ii) a written agreement to transactions involving penny stocks and (iii) a signed and dated copy of a written suitability statement. These disclosure requirements may have the effect of reducing the trading activity in the secondary market for our common stock, and therefore stockholders may have difficulty selling their shares.
The price of our common stock could be subject to volatility related or unrelated to our operations, and purchasers of our common stock could incur substantial losses.
The trading price of our common stock could be subject to wide fluctuations in response to various factors, some of which are beyond our control. These factors include those discussed previously in this "Risk Factors" section of this report and others, such as:
- •
- delays in the commercialization of Mytesi, Neonorm, Canalevia, Equilevia or our other current or future prescription drug product candidates
and non-prescription products;
- •
- any delays in, or suspension or failure of, our current and future studies;
- •
- announcements of regulatory approval or disapproval of any of our current or future product candidates or of regulatory actions affecting our
company or our industry;
- •
- manufacturing and supply issues that affect product candidate or product supply for our studies or commercialization efforts;
- •
- quarterly variations in our results of operations or those of our competitors;
- •
- changes in our earnings estimates or recommendations by securities analysts;
- •
- the payment of licensing fees or royalties in shares of our common stock;
- •
- announcements by us or our competitors of new prescription drug products or product candidates or non-prescription products, significant
contracts, commercial relationships, acquisitions or capital commitments;
- •
- announcements relating to future development or license agreements including termination of such agreements;
- •
- adverse developments with respect to our intellectual property rights or those of our principal collaborators;
- •
- commencement of litigation involving us or our competitors;
- •
- any major changes in our board of directors or management;
- •
- new legislation in the United States relating to the prescription, sale, distribution or pricing of gastrointestinal health products;
- •
- product liability claims, other litigation or public concern about the safety of our prescription drug product or product candidates and
non-prescription products or any such future products;
- •
- market conditions in the human or animal industry, in general, or in the gastrointestinal health sector, in particular, including performance
of our competitors; and
- •
- general economic conditions in the United States and abroad.
In addition, the stock market, in general, or the market for stocks in our industry, in particular, may experience broad market fluctuations, which may adversely affect the market price or liquidity of our common stock. Any sudden decline in the market price of our common stock could trigger securities class-action lawsuits against us. If any of our stockholders were to bring such a lawsuit against us, we could incur substantial costs defending the lawsuit and the time and attention of our
37
management would be diverted from our business and operations. We also could be subject to damages claims if we were found to be at fault in connection with a decline in our stock price.
No active market for our common stock exists or may develop, and you may not be able to resell our common stock when you wish to sell them or at a price that you consider attractive or satisfactory.
Prior to our initial public offering in May 2015, there was no public market for shares of our common stock. The listing of our common stock on The NASDAQ Capital Market does not assure that a meaningful, consistent and liquid trading market exists. Although our common stock is listed on The NASDAQ Capital Market, trading volume in our common stock has been limited and an active trading market for our shares may never develop or be sustained. If an active market for our common stock does not develop, you may be unable to sell your shares when you wish to sell them or at a price that you consider attractive or satisfactory. The lack of an active market may also adversely affect our ability to raise capital by selling securities in the future, or impair our ability to license or acquire other product candidates, businesses or technologies using our shares as consideration.
Holders of the pre-funded warrants will not have rights of common stockholders until such warrants are exercised.
The pre-funded warrants being offered do not confer any rights of common stock ownership on their holders, such as voting rights or the right to receive dividends, but rather merely represent the right to acquire shares of common stock at a fixed price.
If securities or industry analysts do not publish research or reports about our company, or if they issue adverse or misleading opinions regarding us or our stock, our stock price and trading volume could decline.
The trading market for our common stock depends in part on the research and reports that industry or financial analysts publish about us or our business. We do not influence or control the reporting of these analysts. If one or more of the analysts who do cover us downgrade or provide a negative outlook on our company or our industry, or the stock of any of our competitors, the price of our common stock could decline. If one or more of these analysts ceases coverage of our company, we could lose visibility in the market, which in turn could cause the price of our common stock to decline.
You may be diluted by conversions of outstanding convertible notes, Series A Preferred shares, and exercises of outstanding options and warrants.
As of June 30, 2018, we had (i) 3,314,955 shares of common stock issuable upon conversion of outstanding preferred stock as of June 30, 2018 with a weighted-average conversion price of $2.775 per share; (ii) 2,870,138 shares of common stock issuable upon exercise of outstanding options as of June 30, 2018 with a weighted-average exercise price of $14.85 per share; (iii) 209,531 shares of common stock issuable upon exercise of outstanding inducement options as of June 30, 2018 with a weighted-average exercise price of $1.75 per share; (iv) 941,347 shares of common stock issuable upon exercise of outstanding warrants as of June 30, 2018 with a weighted-average exercise price of $4.70 per share; (v) 185,417 shares of common stock issuable upon exercise of outstanding warrants as of June 30, 2018 with a weighted-average exercise price of $0.90 per share; (vi) 33,918 shares of common stock issuable upon exercise of outstanding warrants as of June 30, 2018 with a weighted-average exercise price of $1.23 per share; (vii) up to 720,721 shares of common stock issuable upon conversion of outstanding convertible promissory notes in the aggregate principal amount of $10,000,000 issued after June 30, 2018, convertible at a price of $13.875 per share; (viii) 38,675 shares of common stock issuable pursuant to a convertible promissory note issued to Chicago Venture Partners, L.P. ("CVP"), in the aggregate principal amount of $580,127, convertible at a price of $15 per share of common stock; (ix) 670,586 shares of common stock, issuable upon exercise of a warrant issued on August 28, 2018 to Pacific Capital Management, LLC with an exercise price that is the lower of (i) $0.85 per share and
38
(ii) the average of the closing sales price of the common stock for the 30 consecutive trading days commencing on September 4, 2018; (x) 185,417 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to L2 Capital, LLC with an exercise price of $0.90 per share; (xi) 33,918 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to Charles C. Conte with an exercise price of $1.23 per share; (xii) up to 706,765 shares of common stock issuable upon conversion of a convertible promissory note delivered to L2 Capital, LLC on September 11, 2018 in the aggregate principal amount of $445,000 with an exercise price of $0.85 per share; and (xiii) up to 176,691 shares of common stock issuable upon conversion of a convertible promissory note delivered to Charles C. Conte on September 11, 2018 in the aggregate principal amount of $111,250 with an exercise price of $0.85 per share.
The exercise of such options and warrants or conversion of the convertible promissory notes and preferred stock will result in further dilution of your investment. In addition, you may experience additional dilution if we issue common stock in the future. As a result of this dilution, you may receive significantly less in net tangible book value than the full purchase price you paid for the shares in the event of liquidation.
If shares of our non-voting common stock are converted into shares of our voting common stock, your voting power will be diluted.
As of June 30, 2018, we had 8,736,579 shares of voting common stock and 2,686,749 shares of voting common stock issuable upon conversion of non-voting common stock outstanding. Generally, holders of our non-voting common stock have no voting power (other than in connection with a change of control of our company) and have no right to participate in any meeting of stockholders or to have notice thereof. However, shares of our non-voting common stock that are converted into voting common stock will have all the voting rights of the voting common stock. Shares of our non-voting common stock are convertible into shares of our voting common stock (i) at the option of the respective holders thereof, at any time or (ii) automatically, without any payment of additional consideration by the holder thereof, (x) upon a transfer of such shares to any person or entity that is neither an affiliate of Nantucket nor an investment fund, investment vehicle or other account, that is, directly or indirectly, managed or advised by Nantucket or any of its affiliates pursuant to a sale of such stock to a third-party for cash, or (y) upon the subsequent release or transfer of such shares to the registered pre-Merger legacy stockholders of Napo's outstanding shares of common stock as of July 31, 2017 (the "Napo Legacy Stockholders"). Upon conversion of any non-voting common stock, your voting power will be diluted in proportion to the decrease in your ownership of the total outstanding voting common stock.
We will have broad discretion to use the net proceeds from this offering, and may use them in ways that do not enhance our operating results or the market price of our common stock.
Our management will have broad discretion regarding the use of the net proceeds from this offering, and we could spend the net proceeds in ways our stockholders may not agree with or that do not yield a favorable return, if at all. We intend to use the net proceeds from this offering for general corporate and working capital purposes. We may also use a portion of the net proceeds to acquire additional product candidates or complementary assets or businesses; however, we currently have no agreements or commitments to complete any such transaction. Our use of these proceeds may differ substantially from our current plans. If we do not invest or apply the net proceeds from this offering in ways that improve our operating results or our prospects, our stock price could decline.
39
Purchasers in this offering will experience immediate and substantial dilution in the book value of their investment.
The public offering price per share of our voting common stock will be substantially higher than the net tangible book value per share of our voting common stock immediately after the offering. At the public offering price of $ per share, purchasers of our voting common stock will incur an estimated immediate dilution of $ per share in the net tangible book value of their purchased shares. Conversely, the shares of voting common stock that our existing stockholders currently own will receive an increase in net tangible book value of per share. For a further description of the dilution that you will experience immediately after this offering, see the section in this prospectus titled "Dilution."
Provisions in our charter documents and under Delaware law could discourage a takeover that stockholders may consider favorable and may lead to entrenchment of management.
Our third amended and restated certificate of incorporation, as amended, and amended and restated bylaws contain provisions that could delay or prevent changes in control or changes in our management without the consent of our board of directors. These provisions to include the following:
- •
- a classified board of directors with three-year staggered terms, which may delay the ability of stockholders to change the membership of a
majority of our board of directors;
- •
- no cumulative voting in the election of directors, which limits the ability of minority stockholders to elect director candidates;
- •
- the exclusive right of our board of directors to elect a director to fill a vacancy created by the expansion of the board of directors or the
resignation, death or removal of a director, which prevents stockholders from being able to fill vacancies on our board of directors;
- •
- the ability of our board of directors to authorize the issuance of shares of preferred stock and to determine the terms of those shares,
including preferences and voting rights, without stockholder approval, which could adversely affect the rights of our common stockholders or be used to deter a possible acquisition of our company;
- •
- the ability of our board of directors to alter our bylaws without obtaining stockholder approval;
- •
- the required approval of the holders of at least 75% of the shares entitled to vote at an election of directors to adopt, amend or repeal our
bylaws or repeal the provisions of our third amended and restated certificate of incorporation, as amended, regarding the election and removal of directors;
- •
- a prohibition on stockholder action by written consent, which forces stockholder action to be taken at an annual or special meeting of our
stockholders;
- •
- the requirement that a special meeting of stockholders may be called only by the chairman of the board of directors, the chief executive
officer, the president or the board of directors, which may delay the ability of our stockholders to force consideration of a proposal or to take action, including the removal of directors; and
- •
- advance notice procedures that stockholders must comply with in order to nominate candidates to our board of directors or to propose matters to be acted upon at a stockholders' meeting, which may discourage or deter a potential acquirer from conducting a solicitation of proxies to elect the acquirer's own slate of directors or otherwise attempting to obtain control of us.
These provisions could inhibit or prevent possible transactions that some stockholders may consider attractive.
40
We are also subject to the anti-takeover provisions contained in Section 203 of the Delaware General Corporation Law. Under Section 203, a corporation generally may not engage in a business combination with any holder of 15% or more of its capital stock unless the holder has held the stock for three years or, among other exceptions, the board of directors has approved the transaction.
Our amended and restated bylaws designate the Court of Chancery of the State of Delaware as the sole and exclusive forum for certain actions and proceedings that may be initiated by our stockholders, which could limit our stockholders' ability to obtain a favorable judicial forum for disputes with us or our directors, officers or other employees.
Our amended and restated bylaws provide that, unless we consent in writing to an alternative forum, the Court of Chancery of the State of Delaware will be the sole and exclusive forum for (i) any derivative action or proceeding brought on our behalf, (ii) any action asserting a claim of breach of a fiduciary duty owed by any director, officer or other employee to us or our stockholders, (iii) any action asserting a claim arising pursuant to any provision of the Delaware General Corporation Law, (iv) any action asserting a claim that is governed by the internal affairs doctrine or (v) any action to interpret, apply, enforce or determine the validity of our certificate of incorporation or bylaws. Any person purchasing or otherwise acquiring any interest in any shares of our capital stock shall be deemed to have notice of and to have consented to this provision of our amended and restated bylaws. This choice-of-forum provision may limit our stockholders' ability to bring a claim in a judicial forum that they find favorable for disputes with us or our directors, officers or other employees, which may discourage such lawsuits. Alternatively, if a court were to find this provision of our amended and restated bylaws inapplicable or unenforceable with respect to one or more of the specified types of actions or proceedings, we may incur additional costs associated with resolving such matters in other jurisdictions, which could harm our business and financial condition.
We do not intend to pay dividends on our common stock, and your ability to achieve a return on your investment will depend on appreciation in the market price of our common stock.
We currently intend to invest our future earnings, if any, to fund our growth and not to pay any cash dividends on our common stock. Moreover, so long as Nantucket or any of its affiliates owns any shares of our non-voting common stock, we cannot pay dividends on our common stock or non-voting common stock without obtaining the prior written consent of Nantucket. Because we do not intend to pay dividends and may be required to obtain written consent if we were to do so, your ability to receive a return on your investment will depend on any future appreciation in the market price of our common stock. We cannot be certain that our common stock will appreciate in price.
Our principal stockholders own a significant percentage of our voting stock and will be able to exert significant control over matters subject to stockholder approval.
As of August 31, 2018, our executive officers, directors, holders of 5% or more of our capital stock and their respective affiliates beneficially owned in the aggregate approximately 73.25% of the outstanding shares of our voting common stock. As a result of their stock ownership, these stockholders may have the ability to influence our management and policies, and will be able to significantly affect the outcome of matters requiring stockholder approval such as elections of directors, amendments of our organizational documents or approvals of any merger, sale of assets or other major corporate transaction. This may prevent or discourage unsolicited acquisition proposals or offers for our common stock that you may feel are in your best interest as one of our stockholders.
41
The requirements of being a public company, including compliance with the reporting requirements of the Exchange Act and the requirements of the Sarbanes-Oxley Act, may strain our resources, increase our costs and distract management, and we may be unable to comply with these requirements in a timely or cost-effective manner.
Our initial public offering had a significant, transformative effect on us. Prior to our initial public offering, our business operated as a privately-held company, and we were not required to comply with public reporting, corporate governance and financial accounting practices and policies required of a publicly-traded company. As a publicly-traded company, we incur significant additional legal, accounting and other expenses compared to historical levels. In addition, new and changing laws, regulations and standards relating to corporate governance and public disclosure, including the Dodd-Frank Wall Street Reform and Consumer Protection Act and the rules and regulations thereunder, as well as under the Sarbanes-Oxley Act, the JOBS Act and the rules and regulations of the SEC and The NASDAQ Capital Market, may result in an increase in our costs and the time that our board of directors and management must devote to our compliance with these rules and regulations. These rules and regulations have substantially increased our legal and financial compliance costs and diverted management time and attention from our product development and other business activities.
The Sarbanes-Oxley Act requires, among other things, that we assess the effectiveness of its internal control over financial reporting annually and the effectiveness of our disclosure controls and procedures quarterly. In particular, Section 404 of the Sarbanes-Oxley Act, or Section 404, requires us to perform system and process evaluation and testing of our internal control over financial reporting to allow management to report on, and our independent registered public accounting firm potentially to attest to, the effectiveness of our internal control over financial reporting. We have needed to expend time and resources on documenting our internal control over financial reporting so that we are in a position to perform such evaluation when required. As an "emerging growth company," we expect to avail ourselves of the exemption from the requirement that our independent registered public accounting firm attest to the effectiveness of our internal control over financial reporting under Section 404. However, we may no longer avail itself of this exemption when we cease to be an "emerging growth company." When our independent registered public accounting firm is required to undertake an assessment of our internal control over financial reporting, the cost of our compliance with Section 404 will correspondingly increase. Our compliance with applicable provisions of Section 404 requires that we incur substantial accounting expense and expend significant management time on compliance-related issues as we implement additional corporate governance practices and comply with reporting requirements. Moreover, if we are not able to comply with the requirements of Section 404 applicable to us in a timely manner, or if we or our independent registered public accounting firm identifies deficiencies in our internal control over financial reporting that are deemed to be material weaknesses, the market price of our stock could decline and we could be subject to sanctions or investigations by the SEC or other regulatory authorities, which would require additional financial and management resources.
We are an "emerging growth company" and we cannot be certain if the reduced disclosure requirements applicable to "emerging growth companies" will make our common stock less attractive to investors.
We are an "emerging growth company," as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and we may take advantage of certain exemptions and relief from various reporting requirements that are applicable to other public companies that are not "emerging growth companies." In particular, while we are an "emerging growth company" (i) we will not be required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act, (ii) we will be subject to reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements and (iii) we will not be required to hold nonbinding advisory votes on executive compensation or stockholder approval of any golden parachute payments not previously
42
approved. In addition, the JOBS Act provides that an emerging growth company can delay its adoption of any new or revised accounting standards, but we have irrevocably elected not to avail ourselves of this exemption and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies. In addition, investors may find our common stock less attractive if we rely on the exemptions and relief granted by the JOBS Act. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may decline and/or become more volatile.
We may remain an "emerging growth company" until as late as December 31, 2020 (the fiscal year-end following the fifth anniversary of the closing of our initial public offering, which occurred on May 18, 2015), although we may cease to be an "emerging growth company" earlier under certain circumstances, including (i) if the market value of our common stock that is held by non-affiliates exceeds $700.0 million as of any June 30, in which case we would cease to be an "emerging growth company" as of December 31 of such year, (ii) if our gross revenue exceeds $1.07 billion in any fiscal year or (iii) if we issue more than $1.0 billion of non-convertible debt over a three-year period.
43
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking statements. All statements other than statements of historical facts contained in this prospectus, including statements regarding our future results of operations and financial position, business strategy, prospective products, product approvals, research and development costs, timing of receipt of clinical trial, field study and other study data, and likelihood of success, commercialization plans and timing, other plans and objectives of management for future operations, and future results of current and anticipated products are forward-looking statements. These statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements.
In some cases, you can identify forward-looking statements by terms such as "may," "will," "should," "expect," "plan," "aim," "anticipate," "could," "intend," "target," "project," "contemplate," "believe," "estimate," "predict," "potential" or "continue" or the negative of these terms or other similar expressions. The forward-looking statements in this prospectus are only predictions. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. These forward-looking statements speak only as of the date of this prospectus and are subject to a number of risks, uncertainties and assumptions described under the sections in this prospectus titled "Risk Factors" and elsewhere in this prospectus. Forward-looking statements are subject to inherent risks and uncertainties, some of which cannot be predicted or quantified and some of which are beyond our control. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. Moreover, we operate in a dynamic industry and economy. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties that we may face. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
Unless otherwise indicated, information contained in this prospectus concerning our industry and the markets in which we operate, including our general expectations and market position, market opportunity and market share, is based on information from our own management estimates and research, as well as from industry and general publications and research, surveys and studies conducted by third parties. Management estimates are derived from publicly available information, our knowledge of our industry and assumptions based on such information and knowledge, which we believe to be reasonable. In addition, assumptions and estimates of our and our industry's future performance are necessarily subject to a high degree of uncertainty and risk due to a variety of factors, including those described in "Risk Factors." These and other factors could cause our future performance to differ materially from our assumptions and estimates. See "Special Note Regarding Forward-Looking Statements."
44
We estimate that we will receive net proceeds of approximately $ million from this offering, or approximately $ million if the underwriter exercises its option to purchase additional shares in full, based on an assumed public offering price of $ per share, which is the last reported sale price of our common stock on The Nasdaq Capital Market on September , 2018, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us.
We intend to use the net proceeds from this offering to fund approximately $1.2-1.6 million of non-clinical pipeline and business development activities, and the remainder for the ongoing commercialization of Mytesi as well as for working capital and other general corporate purposes. Pending our use of the net proceeds from this offering, we intend to invest the net proceeds in a variety of capital preservation investments, including short-term, investment grade, interest bearing instruments and U.S. government securities.
This expected use of the net proceeds to us from this offering represents our intentions based upon our current plans and business conditions. The amounts and timing of our actual expenditures may vary significantly depending on numerous factors, including the progress of our research and development efforts, the status of and results from clinical trials, any collaborations that we may enter into with third parties for our programs, any unforeseen cash needs and the factors described in "Risk Factors". Accordingly, our management will have broad discretion in applying the net proceeds from this offering. An investor will not have the opportunity to evaluate the economic, financial or other information on which we base our decisions on how to use the proceeds.
Based on our current plans, we believe that our existing cash, cash equivalents and marketable securities, together with the net proceeds from this offering, will be sufficient to enable us to fund our operating expenses and capital expenditure requirements through to a cash flow positive situation. We have based this estimate on assumptions that may prove to be wrong, and we could use our available capital resources sooner than we currently expect. We will need to raise substantial additional funds before we can expect to commercialize additional products, if approved. We may satisfy our future cash needs through the sale of equity securities, debt financings, working capital lines of credit, corporate collaborations or license agreements, grant funding, interest income earned on invested cash balances or a combination of one or more of these sources.
45
MARKET PRICE OF OUR COMMON STOCK
Our shares of common stock are listed and traded on The NASDAQ Capital Market under the symbol "JAGX"
The following table sets forth, for the periods indicated, the high and low intra-day sale prices in dollars on The NASDAQ Capital Market for our common stock, as adjusted for the 1-for-15 reverse stock split effected on June 1, 2018.
Quarter Ended
|
High | Low | |||||
|---|---|---|---|---|---|---|---|
March 31, 2016 |
$ | 69.00 | $ | 20.25 | |||
June 30, 2016 |
$ | 56.85 | $ | 17.85 | |||
September 30, 2016 |
$ | 33.75 | $ | 16.28 | |||
December 31, 2016 |
$ | 22.95 | $ | 9.15 | |||
March 31, 2017 |
$ | 22.80 | $ | 7.50 | |||
June 30, 2017 |
$ | 15.45 | $ | 7.58 | |||
September 30, 2017 |
$ | 14.85 | $ | 2.55 | |||
December 31, 2017 |
$ | 3.75 | $ | 1.58 | |||
March 31, 2018 |
$ | 6.60 | $ | 1.80 | |||
June 30, 2018 |
$ | 3.00 | $ | 1.40 | |||
On September 10, 2018, the last reported sale price of our common stock on The NASDAQ Capital Market was $0.66 per share. As of June 30, 2018, there were approximately 38 stockholders of record of our common stock. These figures do not reflect the beneficial ownership or shares held in nominee name, nor do they include holders of any restricted stock units or warrants.
46
We have never declared or paid any cash dividends on our capital stock. We intend to retain future earnings, if any, to finance the operation and expansion of our business and do not anticipate paying any cash dividends in the foreseeable future. Any future determination related to dividend policy will be made at the discretion of our board of directors after considering our financial condition, results of operations, capital requirements, business prospects and other factors the board of directors deems relevant, and subject to the restrictions contained in any future financing instruments.
47
You should read this information in conjunction with our financial statements and related notes incorporated by reference in this prospectus and the sections in this prospectus titled "Selected Financial Data."
The following table sets forth our cash and cash equivalents and capitalization as of June 30, 2018, as follows:
- •
- on an actual basis;
- •
- on an as adjusted basis to give further effect to the receipt of the estimated net proceeds from the sale of shares of common stock in this offering at a price of $ per share, and after deducting the estimated underwriting discounts and commissions and estimated expenses payable by us.
You should read this table in conjunction with "Selected Financial Data" included elsewhere in this prospectus and with "Management's Discussion and Analysis of Financial Condition and Results of Operations" in our Annual Report on Form 10-K for the year ended December 31, 2017 and our financial statements and related notes incorporated by reference herein.
The as adjusted information set forth in the table below is illustrative only and will be adjusted based on the actual public offering price and other terms of this offering determined at pricing.
| |
June 30, 2018 |
As adjusted(1) | |||||
|---|---|---|---|---|---|---|---|
| |
(in thousands) |
|
|||||
Cash and cash equivalents |
$ | 2,412 | |||||
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
Series A convertible preferred stock; par value $0.0001 per share: 10,000,000 shares authorized at June 30, 2018 and December 31, 2017; 5,524,926 and 0 issued and outstanding at June 30, 2018 and December 31, 2017; (liquidation preference of $9,9199,002 at June 30, 2018) actual; |
$ | 9,000 | |||||
Stockholders' (deficit) equity: |
|||||||
Common stock: par value $0.0001 per share: 150,000,000 shares and 250,000,000 authorized at June 30, 2018 and December 31, 2017, respectively; 8,736,579 and 4,180,484 shares issued and outstanding at June 30, 2018 and December 31, 2017, respectively actual; shares authorized, shares issued and outstanding, as adjusted |
1 | ||||||
Common stock: non-voting: par value $0.0001 per share: 50,000,000 shares and 250,000,000 authorized at June 30, 2018 and December 31, 2017, respectively; 40,301,237 and 42,617,893 shares issued and outstanding at June 30, 2018 and December 31, 2017, respectively actual; shares authorized, shares issued and outstanding, as adjusted |
4 | ||||||
Additional paid-in capital |
89,772 | ||||||
Accumulated deficit |
(75,759 | ) | |||||
| | | | | | | | |
Total stockholders' equity |
14,018 | ||||||
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
Total capitalization |
|||||||
- (1)
- A $1.00 increase or decrease in the assumed public offering price of $ per share would increase or decrease total stockholders' equity and total capitalization on an as adjusted basis by approximately $ , assuming that the number of shares offered by us, as set forth on the cover
48
page of this prospectus, remains the same and after deducting estimated underwriting discounts and commissions.
The number of shares of our common stock to be outstanding after this offering is based on 8,736,579 shares of voting common stock and 40,301,237 shares of nonvoting common stock outstanding as of June 30, 2018 and assumes the sale and issuance by us of shares of common stock in this offering and excludes, as of June 30, 2018:
- •
- 3,314,955 shares of common stock issuable upon conversion of outstanding preferred stock as of June 30, 2018 with a weighted-average
conversion price of $2.775 per share;
- •
- 2,704,692 shares of common stock issuable upon exercise of outstanding options as of June 30, 2018 with a weighted-average exercise
price of $6.40 per share;
- •
- 402,348 shares of common stock reserved for future issuance under our 2014 Stock Incentive Plan as of June 30, 2018;
- •
- 209,531 shares of common stock issuable upon exercise of outstanding inducement options as of June 30, 2018 with a weighted-average
exercise price of $1.75 per share;
- •
- 392,904 shares of common stock issuable upon exercise of outstanding restricted stock unit awards, or RSUs, as of June 30, 2018;
- •
- 270,761 shares of common stock issuable upon exercise of outstanding warrants as of June 30, 2018 with a weighted-average exercise price of $16.35 per share;
The number of shares of common stock outstanding after the offering also does not include the following:
- •
- 866,524 shares of common stock issued after June 30, 2018;
- •
- Up to 720,721 shares of common stock issuable upon conversion of outstanding convertible promissory notes in the aggregate principal amount of
$10,000,000 issued after June 30, 2018, convertible at a price of $13.875 per share;
- •
- 38,675 shares of common stock issuable pursuant to a convertible promissory note issued to Chicago Venture Partners, L.P. ("CVP"), in
the aggregate principal amount of $580,127, convertible at a price of $15 per share of common stock;
- •
- 670,586 shares of common stock, issuable upon exercise of a warrant issued on August 28, 2018 to Pacific Capital Management, LLC
with an exercise price that is the lower of (i) $0.85 per share and (ii) the average of the closing sales price of the common stock for the 30 consecutive trading days commencing on
September 4, 2018;
- •
- 185,417 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to L2 Capital, LLC with an
exercise price of $0.90 per share;
- •
- 33,918 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to Charles C. Conte with an exercise price
of $1.23 per share;
- •
- Up to 706,765 shares of common stock issuable upon conversion of a convertible promissory note delivered to L2 Capital, LLC on
September 11, 2018 in the aggregate principal amount of $445,000 with an exercise price of $0.85 per share; and
- •
- Up to 176,691 shares of common stock issuable upon conversion of a convertible promissory note delivered to Charles C. Conte on September 11, 2018 in the aggregate principal amount of $111,250 with an exercise price of $0.85 per share.
49
DESCRIPTION OF SECURITIES WE ARE OFFERING
We are offering shares of our common stock or pre-funded warrants to purchase shares of our common stock. The shares of common stock or pre-funded warrants will be issued separately. We are also registering the shares of common stock issuable from time to time upon exercise of the pre-funded warrants offered hereby.
Common Stock
- •
- As of June 30, 2018, we had 8,736,579 shares of voting common stock and 40,301,237 shares of nonvoting common stock outstanding and
5,524,926 shares of preferred stock outstanding. See "Capitalization" beginning on page 48 of this prospectus for a description of the exclusions pertaining to the outstanding share information.
- •
- As of June 30, 2018, we had 38 record holders of common stock.
Voting Rights
The holders of our common stock are entitled to one vote per share on all matters to be voted on by our stockholders. Subject to preferences that may be applicable to any outstanding shares of preferred stock, holders of common stock are entitled to receive ratably such dividends as may be declared by our board of directors out of funds legally available for that purpose. In the event of our liquidation, dissolution or winding up, the holders of common stock are entitled to share ratably in all assets remaining after the payment of liabilities, subject to the prior distribution rights of preferred stock then outstanding. Holders of common stock have no preemptive, conversion or subscription rights. There are no redemption or sinking fund provisions applicable to the common stock.
Dividends
Subject to preferences that may be applicable to any outstanding preferred stock, holders of common stock are entitled to receive dividends, if any, as may be declared from time to time by our board of directors out of legally available funds.
Liquidation
In the event of our liquidation, dissolution or winding up, holders of common stock will be entitled to share ratably in the net assets legally available for distribution to stockholders after the payment of all of our debts and other liabilities and the satisfaction of any liquidation preference granted to the holders of any then outstanding shares of preferred stock.
Rights and Preferences
Holders of common stock have no preemptive, conversion, subscription or other rights, and there are no redemption or sinking fund provisions applicable to the common stock. The rights, preferences, and privileges of the holders of common stock are subject to and may be adversely affected by, the rights of the holders of shares of any series of preferred stock that we may designate in the future.
Fully Paid and Nonassessable
All of our outstanding shares of common stock are, and the shares of common stock to be issued pursuant to this offering, when paid for, will be fully paid and nonassessable.
50
Pre-Funded Warrants
The following summary of certain terms and provisions of pre-funded warrants that are being offered hereby is not complete and is subject to, and qualified in its entirety by, the provisions of the pre-funded warrant, the form of which will be filed as an exhibit to the registration statement of which this prospectus forms a part. Prospective investors should carefully review the terms and provisions of the form of pre-funded warrant for a complete description of the terms and conditions of the pre-funded warrants.
Duration and Exercise Price
Each pre-funded warrant offered hereby will have an initial exercise price per share equal to $0.01. The pre-funded warrants will be immediately exercisable and may be exercised at any time until the pre-funded warrants are exercised in full. The exercise price and number of shares of common stock issuable upon exercise is subject to appropriate adjustment in the event of stock dividends, stock splits, reorganizations or similar events affecting our common stock and the exercise price.
Exercisability
The pre-funded warrants will be exercisable, at the option of each holder, in whole or in part, by delivering to us a duly executed exercise notice accompanied by payment in full for the number of shares of our common stock purchased upon such exercise (except in the case of a cashless exercise as discussed below). A holder (together with its affiliates) may not exercise any portion of the pre-funded warrant to the extent that the holder would own more than 4.99% of the outstanding common stock immediately after exercise, except that upon at least 61 days' prior notice from the holder to us, the holder may increase the amount of ownership of outstanding stock after exercising the holder's pre-funded warrants up to 9.99% of the number of shares of our common stock outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of the pre-funded warrants. Purchasers of pre-funded warrants in this offering may also elect prior to the issuance of the pre-funded warrants to have the initial exercise limitation set at 9.99% of our outstanding common stock. No fractional shares of common stock will be issued in connection with the exercise of a pre-funded warrant. In lieu of fractional shares, we will either pay the holder an amount in cash equal to the fractional amount multiplied by the exercise price or round up to the next whole share.
Cashless Exercise
If, at the time a holder exercises its pre-funded warrants, a registration statement registering the issuance of the shares of common stock underlying the pre-funded warrants under the Securities Act is not then effective or available, then in lieu of making the cash payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the holder may elect instead to receive upon such exercise (either in whole or in part) the net number of shares of common stock determined according to a formula set forth in the pre-funded warrants.
Transferability
Subject to applicable laws, a pre-funded warrant may be transferred at the option of the holder upon surrender of the pre-funded warrant to us together with the appropriate instruments of transfer.
Exchange Listing
There is no trading market available for the pre-funded warrants on any securities exchange or nationally recognized trading system. We do not intend to list the pre-funded warrants on any securities exchange or nationally recognized trading system.
51
Rights as a Stockholder
Except as otherwise provided in the pre-funded warrants or by virtue of such holder's ownership of shares of our common stock, the holders of the pre-funded warrants do not have the rights or privileges of holders of our common stock, including any voting rights, until they exercise their pre-funded warrants.
Fundamental Transaction
In the event of a fundamental transaction, as described in the pre-funded warrants and generally including any reorganization, recapitalization or reclassification of our common stock, the sale, transfer or other disposition of all or substantially all of our properties or assets, our consolidation or merger with or into another person, the acquisition of more than 50% of our outstanding common stock, or any person or group becoming the beneficial owner of 50% of the voting power represented by our outstanding common stock, the holders of the pre-funded warrants will be entitled to receive upon exercise of the pre-funded warrants the kind and amount of securities, cash or other property that the holders would have received had they exercised the pre-funded warrants immediately prior to such fundamental transaction.
52
If you invest in our common stock in this offering, your ownership interest will be immediately diluted to the extent of the difference between the public offering price per share of our common stock and the as adjusted net tangible book value per share of our common stock immediately after this offering.
As of June 30 2018, our historical net tangible book value was $(23.7) million, or $(2.08) per share of common stock. Our historical net tangible book value per share represents the amount of our total tangible assets less total liabilities divided by the number of shares of common stock outstanding as of June 30, 2018.
Our as adjusted net tangible book value as of June 30, 2018 would have been $ million or $ per share of common stock, after giving effect to the sale of the shares of common stock in this offering at the assumed public offering price of $ per share, which was the last reported sale price of our common stock on the NASDAQ Capital Market on September , 2018, after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us. This amount represents an immediate decrease in adjusted net tangible book value of $ per share to our existing stockholders, and an immediate dilution in adjusted net tangible book value of approximately $ per share to new investors purchasing shares of common stock in this offering.
Dilution in net tangible book value per share represents the difference between the amount per share paid by purchasers of our common stock in this offering and the net tangible book value per share of our common stock immediately after this offering. The following table illustrates this dilution:
Assumed public offering price per share |
$ | |||
Historical net tangible book value per share as of June 30, 2018 |
$(2.08) | |||
Decrease in net tangible book value per share attributable to new investors |
||||
| | | | | |
As adjusted net tangible book value per share after this offering |
||||
| | | | | |
Dilution per share to new investors |
$ | |||
| | | | | |
| | | | | |
| | | | | |
The dilution information above is illustrative only and will change based on the actual public offering price and other terms of this offering determined at pricing.
If the underwriter exercises its option to purchase additional shares in full, the as adjusted net tangible book value will increase to $ per share, representing an immediate dilution of $ per share to new investors, assuming that the public offering price will be $ per share, which was the last reported sale price of our common stock on The NASDAQ Capital Market on September , 2018.
The number of shares of our common stock to be outstanding after this offering is based on 8,736,579 shares of voting common stock and 40,301,237 shares of nonvoting common stock outstanding as of June 30, 2018 and assumes the sale and issuance by us of shares of common stock in this offering and excludes, as of June 30, 2018:
- •
- 3,314,955 shares of common stock issuable upon conversion of outstanding preferred stock as of June 30, 2018 with a weighted-average
conversion price of $2.775 per share;
- •
- 2,704,692 shares of common stock issuable upon exercise of outstanding options as of June 30, 2018 with a weighted-average exercise
price of $6.40 per share;
- •
- 402,348 shares of common stock reserved for future issuance under our 2014 Stock Incentive Plan as of June 30, 2018;
53
- •
- 209,531 shares of common stock issuable upon exercise of outstanding inducement options as of June 30, 2018 with a weighted-average
exercise price of $1.75 per share;
- •
- 392,904 shares of common stock issuable upon exercise of outstanding restricted stock unit awards, or RSUs, as of June 30, 2018;
- •
- 270,761 shares of common stock issuable upon exercise of outstanding warrants as of June 30, 2018 with a weighted-average exercise price of $16.35 per share;
The number of shares of common stock outstanding after the offering also does not include the following:
- •
- 866,524 shares of common stock issued after June 30, 2018;
- •
- Up to 720,721 shares of common stock issuable upon conversion of outstanding convertible promissory notes in the aggregate principal amount of
$10,000,000 issued after June 30, 2018, convertible at a price of $13.875 per share ;
- •
- 38,675 shares of common stock issuable pursuant to a convertible promissory note issued to Chicago Venture Partners, L.P. ("CVP"), in
the aggregate principal amount of $580,127, convertible at a price of $15 per share of common stock;
- •
- 670,586 shares of common stock, issuable upon exercise of a warrant issued on August 28, 2018 to Pacific Capital Management, LLC
with an exercise price that is the lower of (i) $0.85 per share and (ii) the average of the closing sales price of the common stock for the 30 consecutive trading days
commencing on September 4, 2018;
- •
- 185,417 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to L2 Capital, LLC with an
exercise price of $0.90 per share;
- •
- 33,918 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to Charles C. Conte with an exercise price
of $1.23 per share;
- •
- Up to 706,765 shares of common stock issuable upon conversion of a convertible promissory note delivered to L2 Capital, LLC on
September 11, 2018 in the aggregate principal amount of $445,000 with an exercise price of $0.85 per share; and
- •
- Up to 176,691 shares of common stock issuable upon conversion of a convertible promissory note delivered to Charles C. Conte on September 11, 2018 in the aggregate principal amount of $111,250 with an exercise price of $0.85 per share.
To the extent any of these outstanding convertible securities are exercised or vest, there will be further dilution to new investors. If all of such outstanding preferred stock had been converted, all options or warrants had been exercised, and all RSUs vested as of September , 2018, the as adjusted net tangible book value after this offering would be $ per share, and total dilution to new investors would be $ per share.
If the underwriter exercises its option to purchase additional shares of common stock in full:
- •
- the percentage of shares of common stock held by existing stockholders will decrease to approximately % of the total number of
shares of common stock outstanding after this offering; and
- •
- the number of shares held by new investors will increase to , or approximately % of the total number of shares of common stock outstanding after this offering.
54
Overview
We are a commercial stage pharmaceutical company focused on developing novel, sustainably derived gastrointestinal products on a global basis. Our wholly-owned subsidiary, Napo, focuses on developing and commercializing proprietary human gastrointestinal pharmaceuticals for the global marketplace from plants used traditionally in rainforest areas. Our Mytesi (crofelemer) product is a first-in-class anti-secretory agent, approved by the FDA for the symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy. The full launch of Mytesi began in April 2018—with the support of a full complement of patient and healthcare practitioner programs—when Napo's direct sales force of 18 sales representatives, a national sales director and one regional sales director became fully deployed.
Jaguar was founded in San Francisco, California as a Delaware corporation on June 6, 2013. The Company was a majority-owned subsidiary of Napo Pharmaceuticals, Inc. ("Napo" or the "Former Parent") until the close of the Company's initial public offering on May 18, 2015. On July 31, 2017, with the merger of Jaguar and Napo, Napo began operating as a wholly-owned subsidiary of Jaguar focused on human health and the ongoing commercialization of, and development of follow-on indications for, Mytesi.
We believe that Jaguar is poised to realize a number of value adding events—including continued commercial growth of Mytesi for the current HIV-diarrhea indication, an expanded pipeline of important human follow-on indications, and a second-generation anti-secretory agent, upon which to build global partnerships. As previously announced, Jaguar, through Napo, now controls commercial rights for Mytesi for all indications, territories and patient populations globally, and crofelemer manufacturing is being conducted at a new, multimillion-dollar commercial manufacturing facility that has been FDA-inspected and approved. Additionally, several of the drug product candidates in Jaguar's Mytesi pipeline are backed by Phase 2 and/or proof of concept evidence from completed human clinical trials. The current approved indication in HIV-related diarrhea is a chronic approval, with a safety package that supports chronic use of Mytesi for follow-on indications as well.
Mytesi
Mytesi is a novel, first-in-class anti-secretory agent which has a basic normalizing effect locally on the gut, and this mechanism of action has the potential to benefit multiple gastrointestinal disorders. Mytesi is in development for multiple possible follow-on indications, including cancer therapy-related diarrhea; orphan-drug indications for infants and children with congenital diarrheal disorders and short bowel syndrome (SBS); supportive care for inflammatory bowel disease (IBD); irritable bowel syndrome (IBS); and as a second-generation anti-secretory agent for use in cholera patients. Mytesi has received orphan-drug designation for SBS.
As of the second quarter of 2018, Napo has a fully deployed direct sales organization consisting of 18 Mytesi field sales representatives strategically positioned to cover U.S. geographies with the highest potential (and a national sales director and one regional sales director). Additionally, in June 2018, we hired Robert J. Griffing, a seasoned industry veteran with a broad range of experience that includes commercializing supportive care and HIV treatments, as chief commercial officer for Napo. With support provided by concomitant marketing, promotional activities, patient empowerment programs and medical education initiatives described below, we expect a proportional response in the number of patients treated with Mytesi.
The goal of Napo's internal sales team is to deliver a frequent and consistent selling message to targeted, high-volume prescribers of antiretroviral therapies (ART) and to gastroenterologists who see large numbers of HIV patients. In December 2017 we released the results of a survey of 350 people
55
living with HIV and AIDS regarding the topic of "Talking to Your Doctor About Symptoms". The survey results show that diarrhea remains prevalent in those living with HIV/AIDS, as 27% of respondents living with HIV/AIDS reported that they currently have diarrhea, while 56% reported that they have had diarrhea in the past. Additionally, the results of a recent Napo-sponsored survey of 271 U.S. board certified gastroenterologists indicate that the number one GI complaint for people living with HIV/AIDS is diarrhea, and 93% of U.S. gastroenterologists see patients with HIV/AIDS in their practice.
Key to the success of our sales representatives in growing Mytesi is differentiating and targeting the right doctors—those HIV specialists who are high prescribers of ART medications and those gastrointestinal doctors who see large populations of people living with HIV/AIDS. The target list of prescribers for our sales reps includes a pool of approximately 3,100 high volume ART prescribing HIV specialists, and gastroenterologists who see the largest number of people living with HIV/AIDs, and we've strategically placed our sales force in the US geographies with the highest potential, including San Francisco, Los Angeles/Palm Springs, Miami/southern Florida, northern Florida, New York, New Jersey, Pennsylvania, Delaware, Maryland, DC, Houston, northern Texas, Chicago, St. Louis, Indianapolis, Kansas City, Alabama, Mississippi, Louisiana, North Carolina/South Carolina and Atlanta. Areas not accessible by direct sales representation are able to be reached with Napo's dedicated telesales representative.
In June 2018, Napo entered into an agreement with RedHill Biopharma, a specialty biopharmaceutical company primarily focused on late clinical-stage development and commercialization of proprietary drugs for gastrointestinal diseases and cancer, to establish a U.S. co-promotion program for Mytesi. RedHill's specialized, GI-focused field sales force is promoting Mytesi to health care practitioners in 36 U.S. territories that contain significant numbers of HIV patients and health care practitioners that are not currently covered by Napo's field sales force. In these geographies, RedHill sales representatives will target gastroenterologists who see large populations of people living with HIV, along with nurse practitioners and physician assistants. RedHill field representatives will also target lower-level prescribers of anti-retroviral infectious disease specialists in regions currently covered by Napo's sales force. Four RedHill inside sales representatives will actively target health care practitioners in other regions not covered by the Napo or RedHill field representatives. We believe this co-promotion program will play a significant role in extending the reach of our commercial efforts into the GI medical community in support of the treatment of people living with HIV (PLWH) with Mytesi. Under the terms of the Agreement, RedHill will be compensated based on performance, and the program can be extended by agreement between the two companies.
Napo has completed the training of 39 health care practitioners (HCPs) and 14 patient advocates as part of the Napo Speakers Bureau program and plans to train an additional eight HCPs in 2018. Medical education presentations led by participating HCPs—a group that includes HIV/AIDS specialists, infectious disease specialists, gastroenterologists, colorectal surgeons, and nurse practitioners—focus on the prevalence and pathophysiology of gastrointestinal consequences of HIV infection and on the latest treatment options for HIV-related diarrhea. Presentations given by patient advocate members provide information to PLWH about the prevalence of diarrhea in PLWH and offer guidance about talking to HCPs regarding diarrhea-related concerns.
As previously announced, Napo has planned 1,400 medical education events consisting of HCP peer-to-peer programs, patient programs, and advisory boards to better understand physician and patient attitudes and behaviors related to non-infectious diarrhea in those living with HIV that are on ART. Through August 24, 2018, 39% of programs have targeted HCP peer-to-peer events and 55% patients.
On July 24, 2018, we announced the results of an analysis conducted to examine whether the rate of HIV-associated diarrhea has changed over time. The analysis of data, sourced from the National
56
Institutes of Health (NIH) clinicaltrials.gov database, revealed that 18% of HIV patients experience diarrhea and the rates have not declined significantly over time. The analysis includes data from 38 U.S. clinical trials from 2008-2016 in more than 21,000 patients. The results were reported at the International AIDS 2018 Conference (AIDS 2018) on Tuesday, July 24 in Amsterdam, Netherlands. The poster is available on the AIDS 2018 website at this link: https://programme.aids2018.org//PAGMaterial/eposters/4900.pdf.
With the introduction of newer antiretroviral (ARV) drug therapy, there has been a reduction in the severity of ARV-induced diarrhea. However, a significant portion of this patient population still suffers from diarrhea caused by HIV enteropathy, which is due to direct and indirect effects of HIV on the intestinal mucosa. Chronic diarrhea remains a significant complaint of people living with HIV/AIDS, particularly those who are older and have lived the virus in their gut for 10+ years. According to data from the U.S. Centers for Disease Control and Prevention, currently more than 50% of people living with HIV are over age 50; by 2020 this figure will increase to 70%.
New crofelemer (Mytesi) data from a supplemental analysis of the ADVENT trial was featured in a poster presentation at the 9th International Aids Society (IAS) Conference on HIV Science held in July 2017 in Paris, France. The presentation was titled Long-Term Crofelemer Use Gives Clinically Relevant Reductions in HIV-Related Diarrhea. IAS features the latest HIV science, including basic, clinical and prevention research, and brings together a broad cross section of HIV professionals from around the world with a focus on implementation—moving scientific advances into practice. The results indicate that at the end of the study period, more than 50% of the patients treated had complete resolution of their diarrhea; and 83% had at least a 50% reduction in diarrhea. Entry criteria required at least 7 watery stools in a week, and the average was 20 (with some patients having as high at 67 stools in a week).
In October 2017, Napo launched a national campaign—called "Keep your pants on... Unless you don't want to"—to highlight the need to recognize and treat diarrhea in PLWH. The campaign, which launched initially to the 10,000 participants in the AIDS Walk Los Angeles event on October 15, 2017, was designed to raise awareness and to engage PLWH in a fun and light way to discuss a topic that can be embarrassing. The campaign integrated live third-party events, including the Greater Palm Springs Pride event taking place in November 2017, with social media on the web, Twitter, and Facebook.
Additionally in Q4 2017, Napo launched a print and digital advertising campaign titled "Enough is Enough" to target PLWH who are tired of planning their lives around diarrhea as well as HIV physicians and gastroenterologists. The campaign is centered around national HIV magazines, local HIV publications, and publications targeting physicians.
As of August 24, 2018, Napo has launched its first integrated social digital campaign targeting patients living with HIV who are interested in information on non-infectious diarrhea (who are on ART) coupled with activating healthcare providers. Phase I of this campaign is focused on contextual and native display ads focused on informing patients of options that may assist them with getting on with their daily lives for the long term and restoring their confidence with relief from HIV-related diarrhea. Phase II will kick off with improving the user experience (UX) once patients land on Mytesi.com coupled with an email marketing campaign to appropriate patients and healthcare providers.
Napo is pursuing AIDS Drug Assistance Program (ADAP) formulary listing in states where Mytesi is not currently on ADAP formulary, and Mytesi is now on the ADAP formulary in 29 states. The ADAP program provides Mytesi free of charge to patients who qualify and copay support for some patients who have insurance coverage.
As we announced April 10, 2018, Napo has signed an agreement with the ADAP Crisis Task Force. The agreement establishes a reduced price that will be provided by Napo ADAPs in all U.S. states and
57
territories for purchases of Mytesi. Formed in 2002, the Task Force negotiates reduced drug prices for all ADAPs. ADAPs provide life-saving HIV treatments to low income, uninsured, and underinsured individuals living with HIV/AIDS in all 50 states and the territories. Task Force membership is currently comprised of representatives from Arizona, California, Florida, Illinois, Massachusetts, New York, North Carolina, Tennessee, Texas, Virginia, and Washington state HIV/AIDS divisions. Per the terms of the agreement, all state ADAPs are guaranteed the same reduced price for the drug. ADAPs provide HIV-related services and approved medications to more than half a million people in the U.S. each year, and we expect this agreement to help further expand the number of patients able to benefit from the novel, first-in-class anti-secretory mechanism of action of Mytesi.
Mytesi is currently reimbursed by Medicaid in all 50 states. It is also currently covered on 100% of the top 10 commercial insurance plan national formularies, representing more than 245 million U.S. lives. Additionally, Napo operates a co-pay coupon program, which helps ensure that the majority of participating patients do not have a Mytesi co-pay greater than $25. Information about the NapoCares Patient Assistance Program, which assists patients with benefit verification, prior authorization, and claims appeals, can be found at mytesi.com/mytesi-savings.html.
Pipeline within a product—crofelemer
According to the World Health Organization, there are nearly 1.7 billion cases of diarrheal disease globally every year. Although not all types of diarrhea are secretory in nature, we view the current, initial approval of Mytesi as the opening of the door to an important pipeline—underscored by the current approval by the FDA of the Chemistry, Manufacturing and Controls (CMC) for this natural product, as well as acknowledgement by the FDA of the safety of the product for chronic use for the approved indication.
Crofelemer is in development for cancer therapy-related diarrhea (CTD). Diarrhea continues to be an area of concern for patients undergoing cancer treatment. Novel targeted agents, such as epidermal growth factor receptor antibodies and tyrosine kinase inhibitors, may block natural chloride secretion regulation pathways in the normal gastrointestinal mucosa, thereby leading to secretory diarrhea. We recognize the importance of supportive care for patients being treated with these cancer-related therapies, analogous to the supportive care of managing diarrhea in people living with HIV/AIDS. An investigator-initiated trial (IIT) titled HALT-D: DiarrHeA Prevention and ProphyLaxis with Crofelemer in HER2 Positive Breast Cancer Patients Receiving Trastuzumab, Pertuzumab, and Docetaxel or Paclitaxel with or without Carboplatin is underway in conjunction with Georgetown University. An IIT titled An Open-Label Study to Characterize the Incidence and Severity of Diarrhea in Patients with Early-Stage HER2+ Breast Cancer Treated with Neratinib and Intensive Loperamide Prophylaxis is underway at the University of California, San Francisco. Diarrhea leading to patient discomfort is also a problem with traditional chemotherapy, radiation, and novel immunotherapy agents, such as checkpoint inhibitors. Napo is aiming to file a protocol for a pivotal trial with the FDA in the second half of 2018, with principal and participating investigators from leading cancer centers in the U.S.
Two investigator-initiated trials of Mytesi are underway in breast cancer patients suffering from CTD, one funded by Genentech-Roche with Herceptin (enrolling patients), and one funded by Puma with neratinib (enrolling patients).
According to data appearing in "Treatment Guidelines for CID" (chemotherapy-induced diarrhea) in the April 2004 issue of Gastroenterology and Endoscopy News, diarrhea is the most common adverse event reported in chemotherapy patients. Approved third-party supportive care products for chemotherapy-induced nausea and vomiting (CINV) include Sustol, Aloxi, Akynzeo and Sancuso. According to Transparency Market Research, sales of therapeutics for the prevention of CINV approximated $620 million in 2013, and sales of such therapeutics are expected to reach $1 billion in 2020.
58
Diarrhea has been reported as the most common side effect of the recently approved CDK 4/6 inhibitor abemaciclib and the pan-HER TKI neratinib, with occurrence ranging from 86% to >95% and grade 3 over 40%, in published studies. Diarrhea in this patient population has the potential to cause dehydration, potential infections, and non-adherence to treatment. A novel anti-diarrheal like Mytesi may hold promise for treating secretory diarrhea—and therefore also support long-term cancer treatment adherence—in this population.
As we announced on January 22, 2018, Napo has accepted a request for support submitted by Dr. Mohamad Miqdady, Chief of Pediatric Gastroenterology, Hepatology and Nutrition at Sheikh Khalifa Medical City (SKMC) in Abu Dhabi, for an investigator-initiated trial of crofelemer, the active pharmaceutical ingredient in Mytesi, for congenital diarrheal disorders (CDDs) in children.
CDDs are a group of rare, chronic intestinal channel diseases, with onset in early infancy, that are characterized by severe, lifelong diarrhea and a lifelong need for nutritional intake either parenterally or with a feeding tube. CDDs are related to specific genetic defects inherited as autosomal recessive traits. The incidence of CDDs is prevalent in regions where consanguineous marriages (related by blood) is part of the culture. CDDs are directly associated with serious secondary conditions including dehydration, metabolic acidosis, and failure to thrive, prompting the need for immediate therapy to prevent death and limit lifelong disability.
SKMC is the Abu Dhabi public health system's flagship institution and the largest hospital in the United Arab Emirates (UAE), consisting of a 586-bed tertiary hospital, 14 outpatient specialty clinics, and the Abu Dhabi Blood Bank, all of which are accredited by Joint Commission International, the oldest and largest healthcare standards-setting and accrediting body in the United States. Dr. Miqdady is American Board certified in Pediatric Gastroenterology, Hepatology and Nutrition, and he is a member of Napo's Scientific Advisory Board.
Napo intends to submit documentation in the first half of 2019 to the FDA for the planned formulation of crofelemer appropriate for feeding tube administration to support this investigation.
As announced on June 5, 2017, Napo has received orphan-drug designation from the FDA for pediatric short bowel syndrome (SBS). The Orphan Drug Act provides for granting special status to a drug or biological product to treat a rare disease or condition upon request of a sponsor. Orphan-drug designation qualifies the sponsor of the drug for various development incentives, including extended exclusivity, tax credits for qualified clinical testing, and relief of filing fees.
Jaguar's and Napo's portfolio development strategy involves meeting with Key Opinion Leaders (KOLs) to identify indications that are potentially high-value because they address important medical needs that are significantly or globally unmet, obtain input on protocol practicality and protocol generation, and then strategically sequencing indication development priorities, second-generation product pipeline development, and partnering goals on a global basis, as well as identifying possible opportunities for a Special Protocol Assessment (SPA) from the FDA. When granted, SPA provides that, upon request, FDA will evaluate within 45 days certain protocols to assess whether they are adequate to meet scientific and regulatory requirements identified by the sponsor. In 2007, under the SPA process, Napo obtained agreement with the FDA for the design of the pivotal study protocol for the currently approved indication of crofelemer (Mytesi) for the symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy. The 2007 SPA agreement was an important milestone for Napo, allowing Napo to address and mitigate regulatory uncertainty prior to the completion of its final Phase 3 trial of crofelemer for its currently approved indication.
In October 2017, Napo established a scientific advisory board for each potential follow-on indication currently planned for Mytesi. Napo has developed relationships with physicians and patient advocates around the world who are recognized specialists and key opinion leaders (KOLs) in the
59
planned Mytesi follow-on indications. The two charts below provide the names, credentials and affiliations of current Napo scientific advisory board members and KOL advisors to Napo.
As we announced on May 10, 2017, Dr. Pravin Chaturvedi, a highly experienced drug development veteran who has spent more than 25 years in the pharmaceutical/biotech industry, has been appointed chair of Napo's scientific advisory boards. From 2006 to 2013, Dr. Chaturvedi served as Napo's Chief Scientific Officer and has remained a scientific adviser to the company since 2014. His track record of successful development includes participating in and/or leading successful development efforts for seven drugs, including crofelemer (Mytesi).
We are confident that our scientific advisory boards will provide expert, actionable input regarding all aspects of development, including trial design, for Mytesi for our follow-on indications—each of which addresses a significant, global, unmet medical need. We also expect that our scientific advisory board members will serve as speakers for our medical education programs, authors on Napo abstracts and publications, and as a resource for media inquiries.
Napo's HIV Scientific Advisory Board has focused primarily on physician education, and community and global awareness regarding the importance and availability of solutions for neglected comorbidities, such as the first-in-class anti-secretory mechanism of action of Mytesi for its currently approved indication.
HIV Physicians Scientific Advisory Board
Napo Scientific Advisory Board (SAB) Members
Dr. Roscoe Moore Jr., DVM, MPH, Ph.D., DSc. |
Former Assistant United States Surgeon General and a Rear Admiral (Retired) in the U.S. Public Health Service. Dr. Moore was involved in the creation of PEPFAR (the U.S. President's Emergency Plan for AIDS Relief) | |
David Asmuth, MD |
Infectious diseases specialist and Professor of Medicine, UC Davis Health |
|
Gary Blick, MD, AAHIVS |
Founder of Health Care Advocates International and BEAT AIDS Project Zimbabwe |
|
Christine Wanke, MD |
Director of the Nutrition and Infection Unit; Associate Chair and Professor, Department of Public Health and Community Medicine; Professor, Department of Medicine, Tufts University School of Medicine; Professor, Sackler School of Biomedical Science; Professor, Friedman School of Nutrition Science and Policy |
|
Pravin Chaturvedi, PhD |
Chair of Napo's Scientific Advisory Boards; 25+ years drug development experience in pharmaceutical/biotech field; Successfully developed crofelemer (Mytesi) (first pivotal adaptive design) |
60
Cancer Therapy-Related Diarrhea Scientific Advisory Board
Lee Schwartzberg, MD, FACP |
Executive Director of the West Cancer Center, a multispecialty oncology practice affiliated with the University of Tennessee; Chief, Division of Hematology/Oncology, the University of Tennessee Health Science Center | |
Eric Roeland, M.D. |
Attending Physician, Center for Palliative Care, Harvard Medical School |
|
Hope Rugo, MD |
Clinical Professor of Medicine, Director Breast Oncology and Clinical Trials Education, Division of Hematology and Oncology, University of California San Francisco |
|
Pravin Chaturvedi, PhD |
Chair of Napo's Scientific Advisory Boards; 25+ years drug development experience in pharmaceutical/biotech field; Successfully developed crofelemer (Mytesi) (first pivotal adaptive design) |
IBD Scientific Advisory Board
Corey Siegel, MD, MS |
Associate Professor of Medicine; Associate Professor of The Dartmouth Institute; Director of the Inflammatory Bowel Disease Center at the Dartmouth-Hitchcock Medical Center | |
Pravin Chaturvedi, PhD |
Chair of Napo's Scientific Advisory Boards; 25+ years drug development experience in pharmaceutical/biotech field; Successfully developed crofelemer (Mytesi) (first pivotal adaptive design) |
Pediatric Indications (SBS and CDD) Scientific Advisory Board
Mohammed Miqdady, MD |
Chief of Pediatric Gastroenterology, Hepatology & Nutrition at Sheikh Khalifa Medical City in Abu Dhabi | |
Christopher Duggan, MD |
Senior Physician, Division of Gastroenterology, Hepatology and Nutrition / Director, Center for Nutrition / Medical Director, Center for Advanced Intestinal Rehabilitation, Boston Children's Hospital; Professor of Pediatrics, Harvard Medical School; Professor in the Departments of Nutrition and Global Health and Population, Harvard TH Chan School of Public Health |
|
Martin Martin, MD |
Professor, Department of Pediatrics, David Geffen School of Medicine at UCLA |
|
Sue Rhee, MD |
Division Chief, Pediatric Gastroenterology, Hepatology and Nutrition Pediatric gastroenterologist and liver specialist, UCSF Benioff Children's Hospital |
|
Pravin Chaturvedi, PhD |
Chair of Napo's Scientific Advisory Boards; 25+ years drug development experience in pharmaceutical/biotech field; Successfully developed crofelemer (Mytesi) (first pivotal adaptive design) |
61
Key Opinion Leader (KOL) Advisors to Napo (on an as-needed basis)
KOL Advisors: Cancer Therapy-Related Diarrhea
Herbert DuPont, MD |
Professor and Director, Center for Infectious Diseases, University of Texas Houston School of Public Health |
KOL Advisors: HIV-Related Diarrhea
Patrick Clay, PharmD |
Consultant | |
Brooks D. Cash, MD, AGAF, FACG, FACP, FASGE |
Division Director, Gastroenterology, Hepatology, and Nutrition Visiting Professor of Medicine, The University of Texas McGovern Medical School |
|
Paulo Pacheco, MD |
Clinical Assistant Professor, Department of Medicine, New York University Langone Health |
|
Elie Schochet, MD, FACS |
Colorectal surgeon, Holy Cross Medical Group |
KOL Advisors: Diarrhea Related to IBD
David Rubin, MD |
Joseph B. Kirsner Professor of Medicine Section Chief, Gastroenterology, Hepatology and Nutrition Co-Director, Digestive Diseases Center, University of Chicago Medicine | |
Charles Bernstein, MD |
Distinguished Professor of Medicine and Bingham Chair in Gastroenterology Research, University of Manitoba |
|
William Sandborn, MD |
Director, Inflammatory Bowel Disease Center Chief, Division of Gastroenterology Professor of Medicine, US San Diego Health |
|
Scott Lee, MD |
Associate Professor of Medicine, Digestive Health Center, University of Washington Medical Center |
|
Edward Loftus, Jr., MD |
Consultant, Division of Gastroenterology and Hepatology, Department of Internal Medicine, Mayo Clinic |
|
Douglas Wolf, MD |
Medical Director of IBD Research at Atlanta Gastroenterology Associates. Clinical Assistant Professor of Medicine, Emory University School of Medicine |
KOL Advisors: Pediatric Indications (SBS and CDD)
Jay Thiagarajah, MD, PhD |
Attending Physician, Division of Gastroenterology, Hepatology and Nutrition, Boston Children's Hospital. Instructor of Pediatrics, Harvard Medical School | |
James Goldenring, M.D., Ph.D. |
Professor of Surgery, Vanderbilt University School of Medicine. Paul W. Sanger Chair in Experimental Surgery. Professor of Cell and Developmental Biology |
62
KOL Advisors: Diarrhea Related to Infectious Diseases
Herbert DuPont, MD |
Professor and Director, Center for Infectious Diseases, University of Texas Houston School of Public Health | |
Pradip Bardhan, MBBS, MD |
Chief Physician at ICDDR,B, Bangladesh |
KOL Advisors: Diarrhea Related to IBS
Anthony Lembo, MD |
Director of the GI Motility and Functional Bowel Disorders Program at Beth Israel Deaconess Medical Center and Associate Professor of Medicine at Harvard Medical School | |
Doug Drossman, MD |
Co-Director Emeritus, UNC Center for Functional GI and Motility Disorders Adjunct Professor of Medicine and Psychiatry, University of North Carolina School of Medicine |
|
William Chey, MD |
Professor of Internal Medicine and Professor of Nutritional Sciences, University of Michigan School of Public Health |
According to a 2017 report from Research and Markets, the combined global market for prescription and OTC gastrointestinal agents is expected to reach $21 billion by 2025. Jaguar estimates that a first-in-class anti-secretory agent should be able to achieve a significant portion of the market share.
Our management team has significant experience in gastrointestinal product development for both humans and animals. Napo was founded 29 years ago to perform drug discovery and development by leveraging the knowledge of traditional healers working in rainforest areas. Ten members of the Jaguar and Napo team have been together for more than 15 years. Dr. Steven King, our executive vice president of sustainable supply, ethnobotanical research and intellectual property, and Lisa Conte, our founder, president and CEO, have worked together for more than 29 years. Together, these dedicated personnel successfully transformed crofelemer, which is extracted from trees growing in the rainforest, to Mytesi, which is a natural, sustainably harvested, FDA-approved drug.
There are significant barriers to entry for Mytesi (crofelemer). Through Napo, we hold an extensive global patent portfolio. At the present time we hold approximately 140 issued worldwide patents, with coverage in many cases that extends until 2031. These issued patents cover multiple indications including HIV-AIDS diarrhea, IBS, IBD, manufacturing, enteric protection from gastric juices, among others. We also have approximately 40 pending patent applications worldwide in the human health areas that are being prosecuted.
Mytesi is the first oral drug approved by the FDA under botanical guidance, which provides another barrier to entry from potential generic competition. The FDA requires that the manufacturer of crofelemer use a validated proprietary bioassay to release the drug substance and drug product of Mytesi. While most generic products are fashioned to meet chemical release specifications that are in the public domain, the specifics of this assay are not publicly available. There is no pathway by which a generic product can be developed for a drug approved under botanical guidance. In addition, Mytesi is not systemically absorbed, so the classic approach of creating a generic drug by matching pharmacokinetic blood levels is not possible. A generic player would have to conduct costly and risky clinical trials.
63
While Jaguar's commercial and development efforts have evolved to focus primarily on Mytesi and human pipeline indications since its merger with Napo, the Company is continuing limited initiatives related to Canalevia, its drug product candidate for treatment of chemotherapy-induced diarrhea ("CID") in dogs, and Equilevia, its non-prescription, personalized, premium product for total gut health in equine athletes. CID in dogs is typically caused by the same mechanism of action as in humans, and hence the work in dogs serves as a preclinical proof of concept for the human cancer therapy related diarrhea.
As previously announced, Jaguar has received MUMS (Minor Use and Minor Species) designation status from the FDA for Canalevia for the indication of chemotherapy-induced diarrhea (CID) in dogs. Jaguar has completed clinical and manufacturing activity for Canalevia for this indication. MUMS designation is modeled on the orphan-drug designation for human drug development and offers possible financial incentives to encourage MUMS drug development, such as the availability of grants to help with the cost of developing the MUMS drug. Additionally, as announced on March 8, 2018, the FDA's Center for Veterinary Medicine (CVM) has indicated that Jaguar's Reasonable Expectation of Effectiveness (RxE) technical section is complete towards conditional approval of Canalevia (crofelemer delayed-release tablets) for treatment of CID in dogs, based on CVM's review of the results of Jaguar's completed pilot study (CANA-001) of Canalevia for this indication. Jaguar has now completed two of the four required technical sections of the Company's application for conditional approval of Canalevia for CID in dogs.
Crofelemer is extracted from the Croton lechleri tree, which we sustainably harvest and manage through programs that we have been developing over the past 28 years. This process has involved working with communities to plant trees, obtaining permits for export, and creating a supply network that is robust and reliable.
We continue to have working relationships with partners that began in the 1990s. Additionally, through the establishment of a nonprofit called the Healing Forest Conservancy (HFC), our team has created a long-term mechanism for benefit sharing that recognizes the intellectual contribution of indigenous populations. This program is intended to contribute to the continued strength and effectiveness of the valued and strategically important relationships we have carefully cultivated over the past 28 years.
Product Pipeline
In addition to our Mytesi (crofelemer) product that is approved by the U.S. FDA for the symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy, we are also developing a pipeline of prescription drug product candidates to address unmet needs in gastrointestinal health through Napo. Mytesi (crofelemer) is a novel, first-in-class anti-secretory agent which has a basic normalizing effect locally on the gut, and this mechanism of action has the potential to benefit multiple disorders. Clinical trials demonstrated that nearly 80% of Mytesi users experienced an improvement in their diarrhea over a four-week period. At week 20 of the pivotal trial, over half the patients had no watery stools, or a 100% decrease, and 83% had at least a 50% decrease in watery stools. Our Mytesi pipeline currently includes prescription drug product candidates for four follow-on indications, several of which are backed by Phase 2 evidence from completed Phase 2 trials. In addition, a second-generation proprietary anti-secretory agent is in development for cholera.
64
Napo Prescription Drug Product Candidates
Product Candidates
|
Indication | Completed Milestones | Current Phase of Development |
Anticipated Near-Term Milestones* |
||||
|---|---|---|---|---|---|---|---|---|
Mytesi |
Cancer therapy-related diarrhea (CTD) | • Two investigator-initiated clinical trials funded by Genentech-Roche & Puma |
Phase 2 | • Pivotal protocol development with KOLs for discussions with FDA |
||||
Mytesi |
Supportive care for IBD |
• Safety |
Phase 2 |
• Protocol development with KOLs for discussions with FDA |
||||
|
• Multiple Phase 2 studies completed in various secretory diarrheas (not IBD patients) |
|||||||
Formulation of crofelemer |
Rare disease indications (SBS & CDD) |
• Phase 1 study |
Phase 2 |
• Formulation/IIT, Abu Dhabi |
||||
|
• Orphan-drug designation for SBS |
• Pursue orphan-drug status for CDD |
||||||
Mytesi |
Irritable Bowel Syndrome—diarrhea predominant (IBS-D) |
• Phase 1 study |
Phase 2 |
• Protocol development with KOLs for discussions with FDA |
||||
|
• Two Phase 2 studies completed |
• Publication of supplemental analysis of Phase 2 data |
||||||
SB-300 (lechlemer) |
Second-generation anti-secretory agent for multiple indications including cholera |
• Animal and human studies in secretory diarrheas; successful cholera trial design for anti-secretory mechanism of action with crofelemer |
Pre IND |
• CMC development for
SB-300
• Pre-clinical and Phase 1/2 in 2019 |
- *
- Clinical trials are funding dependent
Estimated Size of Mytesi Target Markets
We believe the medical need for Mytesi is significant, compelling, and unmet, and that doctors are looking for a drug product with a mechanism of action that is distinct from the options currently available to resolve diarrhea. A growing percentage of HIV patients have lived with the virus in their gut for 10+ years, often causing gut enteropathy and chronic or chronic-episodic diarrhea. According to data from the U.S. Centers for Disease Control and Prevention, by 2020 more than 70% of Americans with HIV are expected to be 50 and older.
65
Market
|
Number of Competitors for Mytesi's Approved/ Anticipated Labelled Indication |
Market Size/Potential | ||
|---|---|---|---|---|
HIV-D |
0 | We estimate the U.S. market revenue potential for Mytesi to be approximately $100 million in gross annual sales | ||
CTD |
0 |
An estimated 650,000 U.S. cancer patients receive chemotherapy in an outpatient oncology clinic.(1) Comparable supportive care (i.e. CINV) product sales of ~$620 million in 2013, which is projected to reach $1.0 billion by 2020(2) |
||
IBD |
0 |
Estimated 1,171,000 Americans have IBD(3) |
||
IBS-D |
3 |
Most IBS products have estimated revenue potential of greater than $1.0 billion(4) |
||
CDD/SBS |
0 |
Financial benefits of Orphan-drug Designation |
||
Cholera (hydration maintenance) PRV (SB-300) |
0 |
Priority review vouchers have recently sold for $125 million to $350 million(5) |
- (1)
- Centers
for Disease Control and Prevention. Preventing Infections in Cancer Patients: Information for Health Care Providers
(cdc.gov/cancer/preventinfections/providers.htm)
- (2)
- Heron
Therapeutics, Inc. Form 10-K for the fiscal year ended December 31, 2016
- (3)
- Kappelman,
M. et al. Recent Trends in the Prevalence of Crohn's Disease and Ulcerative Colitis in a Commercially Insured US Population. Dig Dis Sci.
2013 Feb; 58(2): 519-525
- (4)
- Merrill
Lynch forecasts peak US sales of roughly $1.5 bn for Ironwood's Linzess
(http://247wallst.com/healthcare-business/2015/04/27/key-analyst-sees-nearly-30-upside-in-ironwood); Rodman & Renshaw estimate peak annual sales of Synergy Pharmaceuticals' Trulance at $2.3 bn
in 2021 (Source: https://www.benzinga.com/analyst-ratings/analyst-color/17/03/9224181/analyst-synergy-pharma-could-achieve-sustainable-profita)
- (5)
- In Aug. 2015, AbbVie Inc. bought a priority review voucher from United Therapeutics Corp for $350 million (http://www.reuters.com/article/us-abbvie-priorityreview/abbvie-buys-special-review-voucher-for-350-million-idUSKCN0QO1LQ20150819). In Feb. 2017 Sarpeta Therapeutics sold a priority review voucher to Gilead Sciences, Inc. for $125 million (http://fortune.com/2017/02/21/sarepta-gilead-review-voucher/).
The following diagram illustrates the mechanism of action of our human and animal gastrointestinal drug products and drug product candidates, which normalize chloride and water flow and transit time of fluids within the intestinal lumen.
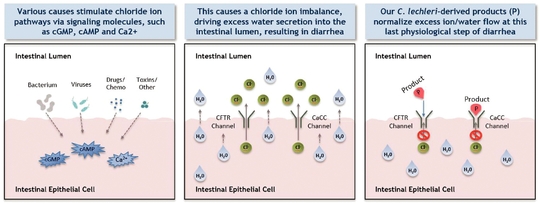
66
Business Strategy
Our goal is to become a leading pharmaceutical company with first-in-class, sustainably derived products that address significant unmet gastrointestinal medical needs globally. To accomplish this goal, we plan to:
Expand Mytesi by leveraging our significant gastrointestinal knowledge, experience and intellectual property portfolio
Mytesi is a novel, first-in-class anti-secretory agent which has a basic normalizing effect locally on the gut, and this mechanism of action has the potential to benefit multiple gastrointestinal disorders. Our Mytesi (crofelemer) product is approved by the U.S. FDA for the symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy. Jaguar, through Napo, controls commercial rights for Mytesi for all indications, territories and patient populations globally. Mytesi is in development for multiple possible follow-on indications, including cancer therapy-related diarrhea; orphan-drug indications for infants and children with congenital diarrheal disorders and short bowel syndrome (SBS); supportive care for inflammatory bowel disease (IBD); irritable bowel syndrome (IBS); and as a second-generation anti-secretory agent for use in cholera patients.
Our management team collectively has more than 100 years of experience in the development of gastrointestinal prescription drug and non-prescription products. This experience covers all aspects of product development, including discovery, preclinical and clinical development, GMP manufacturing, and regulatory strategy. Key members of this team successfully developed Mytesi.
Establish and expand commercial capabilities in Mytesi sales and marketing efforts
As announced on August 7, 2017, we appointed Pete Riojas, a 29-year pharmaceutical industry veteran, to lead Mytesi sales nationally. As of the second quarter of 2018, Napo has a fully deployed direct sales organization consisting of 18 Mytesi field sales representatives strategically positioned to cover U.S. geographies with the highest potential (and two salesforce managers). Additionally, in June 2018, we hired Robert J. Griffing, a seasoned industry veteran with a broad range of experience that includes commercializing supportive care and HIV treatments, as chief commercial officer for Napo. With support provided by concomitant marketing, promotional activities, patient empowerment programs, including an integrated social digital campaign, and medical education initiatives described below, we expect a proportional response in the number of patients treated with Mytesi.
In June 2018, as stated above, Napo entered into an agreement with RedHill Biopharma, a specialty biopharmaceutical company primarily focused on late clinical-stage development and commercialization of proprietary drugs for gastrointestinal diseases and cancer, to establish a U.S. co-promotion program for Mytesi. RedHill's specialized, GI-focused field sales force will promote Mytesi to health care practitioners in 36 U.S. territories that contain significant numbers of HIV patients and health care practitioners that are not currently covered by Napo's field sales force. In these regions, RedHill sales representatives will target gastroenterologists who see large populations of people living with HIV, along with nurse practitioners and physician assistants. RedHill field representatives will also target lower-level ART prescribing infectious disease specialists in regions currently covered by Napo's sales force. Four RedHill inside sales representatives will actively target health care practitioners in other regions not covered by the Napo or RedHill field representatives. We believe this co-promotion program will play a significant role in extending the reach of our commercial efforts into the GI medical community in support of the treatment of people living with HIV (PLWH) with Mytesi. Under the terms of the Agreement, RedHill will be compensated based on performance, and the program can be extended by agreement between the two companies.
With the goal of continuing to drive Mytesi sales and awareness nationwide, our plans for 2018 include the initiation of new and extensive sales and marketing programs, advertising and promotional
67
activities, direct-to-patient hub-service activities, the expected publication of supplemental data, and an increased focus on patient empowerment and educational programs for the important and neglected comorbidity of diarrhea in people living with HIV. Additionally, as stated above, we expect Napo's recently signed agreement with pharmacy services provider TPS to help streamline and expand nationwide patient access to Mytesi. The core benefits of the program, named Mytesi Direct™, include streamlining prescription fulfillment for Mytesi in order to ensure that Mytesi users receive their prescription quickly, coordinating with other Napo programs—such as the Mytesi Copay Savings Card and the NapoCares™ Patient Assistance Program—to help ensure that patient out-of-pocket expenses for Mytesi are as low as possible, and improving Mytesi refill adherence through the transmission of renewal reminders to patients.
Leverage our relationships with key opinion leaders regarding development of follow-on indications
To date, more than 30 key opinion leaders (KOLs) who are recognized specialists in HIV patient care, CTD, IBD, IBS, cholera, SBS, CDD and equine gut health, are participating in our scientific advisory board or KOL advisory program in some manner.
Establish partnerships to support moving pipeline indications to pivotal clinical trials
Jaguar is actively pursuing development of a robust pipeline of potential follow-on indications for crofelemer, and the Company's goal is to establish partnerships to support moving pipeline indications to pivotal clinical trials.
Strategically sequence the development of follow-on indications of Mytesi and seek geographically-focused licensing opportunities
Although it is possible that we may enter into corporate partnering relationships related to Mytesi, our intention would be to retain all commercialization and promotional rights in the U.S., so that we do not become primarily a royalty-collecting organization, and we are opposed to entering into any Mytesi partnering relationship that would require splitting indications. We are seeking to put limited geographically-focused partnerships in place in the near term, while also considering possibilities for a worldwide partnership with a leading global entity (excluding the US exclusive commercial rights) in the field of gastrointestinal care and cancer in the long term.
Reduce risks relating to product development
Risk reduction is a key focus of our product development planning. Mytesi is approved for chronic indication, providing us the ability to leverage this corresponding safety data when seeking approval for planned follow-on indications that are also chronic or chronic episodic indications. Crofelemer manufacturing is being conducted at a new, multimillion-dollar commercial manufacturing facility that has been FDA-inspected and approved. In an effort to reduce risk further, we have implemented the following approach: First, we meet with key opinion leaders, typically at medical conferences—as we did in 2017 at Digestive Disease Week for IBS and IBD, the American Society of Clinical Oncology annual meeting, and the Multinational Association of Supportive Care and Congress. Next, we confirm unmet medical needs with these key opinion leaders, and discuss the practicality of patient enrollment and trial implementation. We then generate protocols to discuss with the FDA, seeking, when possible, special protocol assessments. Our goal, by the time we start devoting significant funds to a clinical trial, is to have de-risked the program as much as we believe we possibly can, in particular the regulatory pathway. We believe this approach will lead to better long-term outcomes for our products in development.
68
We believe that Jaguar is poised to realize a number of synergistic, value adding benefits—and an expanded pipeline of important human follow-on indications and a second-generation anti secretory agent, upon which to build global partnerships.
In May 2016, the New Drug Application ("NDA") and commercial rights for human applications of crofelemer (Mytesi) previously licensed to Salix Pharmaceuticals, Inc. ("Salix") were transferred to Napo. The active pharmaceutical ingredient ("API") in Mytesi is crofelemer, our proprietary, patented gastrointestinal anti-secretory agent sustainably harvested from the rainforest.
Diarrhea is a common adverse event seen with chemotherapy agents typically used in breast and colon cancers, and in particular in the more recently introduced therapeutic classes of epidermal growth factor receptor ("EGFR") monoclonal antibodies and tyrosine kinase inhibitors ("TKI") often used for chronic adjuvant care management of cancer. The increased need for and use of these agents has made diarrhea one of the most disabling issues for cancer patients.
We will seek partnerships outside the United States for the above indications, while focusing on development, and commercial access in the United States directly. We are also focused on investigating SB-300 for various gastrointestinal indications. SB-300 is a proprietary Jaguar pharmaceutical product, a standardized botanical extract distinct from crofelemer, also sustainably derived from the Croton lechleri tree.
We believe SB-300, which has the same mechanism of action as crofelemer and is significantly less costly to produce, may support efforts to receive a priority review voucher from the U.S. FDA for a cholera indication. Priority review vouchers are granted by the FDA to drug developers as an incentive to develop treatments for neglected diseases and rare pediatric diseases. Additionally, we believe SB-300 represents a long-term pipeline opportunity as a second-generation anti-secretory agent, on a global basis, for multiple gastro-intestinal diseases—especially in resource-constrained countries where cost of goods is a factor, in part, because requirements often exist in such regions for drug prices to decrease annually.
The Company has published Phase 2 data on crofelemer for the treatment of devastating dehydration in cholera patients from the renowned International Centre for Diarrhoeal Disease Research (icddr,b) in Bangladesh, and Napo plans to follow the same study design for a trial conducted in association with icddr,b in support of development of SB-300 for the potential cholera-related indication.
Our portfolio development strategy is based on identifying indications that are potentially high-value because they address important medical needs that are significantly or globally unmet, and then strategically sequencing indication development priorities, second-generation product pipeline development, and partnering goals on a global basis.
Our technology for proprietary gastrointestinal disease products is central to the product pipelines of both veterinary and human indications. Crofelemer is also the API in Canalevia, our lead prescription drug product candidate, intended for the treatment of chemotherapy-induced diarrhea in dogs. We expect our first veterinary prescription product launch will be Canalevia for chemotherapy-induced diarrhea, an interesting commercial synergy with the pursuit of follow-on indications for Mytesi.
Mytesi Clinical Data
Mytesi has been clinically demonstrated to have:
- •
- Minimal absorption, with plasma concentrations below the level of detection
- •
- No clinically relevant drug-drug interactions
69
- •
- No effect on viral load or CD4 counts
- •
- Adverse events comparable to those with placebo
The efficacy of Mytesi 125-mg delayed-release tablets twice daily was evaluated in a randomized, double-blind, 24-week, multicenter study (the ADVENT trial) comprised of a placebo-controlled (1 month) treatment period and a placebo-free (5 month) treatment period. The study enrolled HIV-positive patients on stable ART with a history of diarrhea for 1 month or more. In the Mytesi 125mg bid group, more than twice as many patients (18% vs. 8% on placebo, p<0.01) achieved the highly rigorous endpoint defined as reduction to £2 watery stools per week for 2 out of the 4 weeks in the placebo-controlled period (the average baseline in the ADVENT population was 20 watery stools per week).
In a supplemental analysis of the ADVENT study population, 78% of patients in the Mytesi 125mg BID group experienced a decrease in watery stools at week 4. Among these patients that experienced a decrease, 61% had at least a 50% decrease in watery stools. At week 20, 89% of patients in the Mytesi BID group experienced a decrease in watery stools. Among these patients that experienced a decrease, 83% had at least a 50% decrease in watery stools, and over half of patients had no watery stools at all (100% decrease).
Products in Development
Cancer Therapy-Related Diarrhea (CTD)
CTD is a common problem with a relevant mechanism for crofelemer
| National Cancer Institute Criteria for Grading Severity of Diarrhea | ||||||||
|---|---|---|---|---|---|---|---|---|
| |
Grade 1 | Grade 2 | Grade 3 | Grade 4 | ||||
Patients without a colostomy |
Increase of <4 stools per day over pretreatment | Increase of 4 to 6 stools per day or nocturnal stools | Increase of ³7 stools per day or incontinence; need for parenteral support for hydration | Physiologic consequences requiring intensive care; hemodynamic collapse | ||||
Diarrhea is a common adverse event seen with chemotherapy agents in the therapeutic classes of epidermal growth factor receptor ("EGFR") tyrosine kinase inhibitors ("TKI's") and EGFR monoclonal antibodies (for breast, lung, and other malignancies). The increased need for and use of these agents has made diarrhea one of the most disabling issues for cancer patients. Crofelemer offers the potential for an appropriate mechanism of action against this likely secretory diarrhea and has prompted interest among physicians concerned about this diarrheal symptom, stimulating the aforementioned investigator-initiated trials. Diarrhea is also a common adverse event seen with chemotherapy agents used in colorectal and gastric cancers, and chronic maintenance chemotherapy. There are currently no anti-diarrhea agents approved generally for chemotherapy-induced diarrhea.
Clinical Studies
A study titled HALT-D: DiarrHeA Prevention and ProphyLaxis with Crofelemer in HER2 Positive Breast Cancer Patients Receiving Trastuzumab, Pertuzumab, and Docetaxel or Paclitaxel with or without Carboplatin is currently enrolling patients in conjunction with Georgetown University. The primary objective of the study is to characterize the incidence and severity of diarrhea in patients receiving investigational therapy in the setting of prophylactic anti-diarrheal management.
70
A second study, titled An open label study to characterize the incidence and severity of diarrhea in patients with early stage HER2+ breast cancer treated with adjuvant trastuzumab and neratinib followed by neratinib monotherapy, and intensive anti-diarrhea prophylaxis, is currently enrolling patients in conjunction with the University of California at San Francisco. The study is designed to evaluate crofelemer as a salvage anti-diarrheal therapy used with the investigational breast cancer agent neratinib. The primary objective is to characterize the incidence and severity of diarrhea in patients with early stage breast cancer receiving adjuvant trastuzumab and neratinib followed by 1 year of neratinib monotherapy in the setting of prophylactic anti-diarrheal management. The secondary objectives are to evaluate the activity of crofelemer as a rescue anti-diarrheal medication; to assess neratinib adherence, holds, delays, and early discontinuation throughout the course of study therapy, which includes patients receiving neratinib for >1 year; and to assess overall toxicity including constipation and cardiac toxicity with concomitant neratinib and trastuzumab.
Irritable Bowel Syndrome—Diarrhea Predominant (IBS-D)
Diarrhea is a common symptom of irritable bowel syndrome (IBS), a frustrating, underdiagnosed and undertreated condition. IBS-D is a subtype characterized mainly by loose or watery stools at least 25 percent of the time. According to the U.S. FDA, studies estimate that IBS affects 10 to 15 percent of adults in the United States.
Abdominal pain is the key symptom of IBS, and the pain, which is associated with a change in stool frequency or consistency, can be severe. To improve the diagnosis and outcomes for IBS patients and to update clinicians on the latest research, Dr. William Chey, a gastroenterologist and professor of medicine and nutrition sciences at the University of Michigan, along with an international team of collaborators, compiled Rome IV, an updated compendium of diagnostic criteria on functional GI disorders such IBS. Rome IV contains a chapter titled Centrally Mediated Disorders of Gastrointestinal Pain.
Although new agents for IBS-D have come on the market, there is an unmet medical need for long-term, safe management of the abdominal pain associated with IBS-D. We recognize that patients suffering from IBS-D may require a poly-pharmacy approach to lifetime management of their disease. Mytesi, which represents a novel mechanistic approach with the benefit of a long-term safety profile, could possibly be an important addition to the treatment of IBS-D, if approved for this indication.
Mytesi has been demonstrated to be safe for chronic use, and two studies provide statistically significant results of crofelemer use for abdominal pain in women.
The largest group of IBS sufferers are those with the subtype referred to as IBS-M (mixed diarrhea and constipation). IBS-M is also referred to as IBS-A, because the condition often involves frequent alternating between IBS-D and IBS-C (constipation predominant). IBS-M is distressing for patients as well as difficult to diagnose and manage, and is often associated with pain and urgency as well as significant abdominal distension and bloating. No approved drugs currently exist for IBS-M. Leading gastroenterologists have stated that IBS-C drugs may cause diarrhea in an IBS-M patient, and an IBS-D drug may cause significant constipation. Since Mytesi has not caused constipation in clinical trials or real world experience, we therefore believe an opportunity exists for an IBS-M indication for Mytesi. Resultingly, and due to the demonstrated safety of Mytesi for chronic use and its demonstrated benefit for abdominal pain in women, Napo is considering expanding development efforts to evaluate the IBS-M indication.
Clinical Study
Crofelemer has been tested in safety studies and two significant Phase 2 studies for IBS-D as detailed below.
71
Completed Studies—IBS-D
Phase 2a—a randomized double-blind placebo-controlled, dose-ranging (placebo, 125 mg, 250 mg, and 500 mg bid) study over a 12-week treatment period in 246 patients with d-IBS (Rome II criteria), including both males and females, whose average age was 50 years old.
n=245
subjects
61 placebo
62 125 mg crofelemer BID
59 250 mg crofelemer BID
62 500 mg crofelemer BID
IBS symptoms (pain, urgency, stool frequency and consistency, and adequate relief) were self-reported by the patients via an interactive voice response system. Patients needed to exhibit active disease during the two-week baseline period as defined by a mean daily stool frequency greater than or equal to 2/day, pain score greater than or equal to 1 and stool consistency greater than or equal to 3 (5-point Lickert scale for pain and consistency) to be enrolled. Patients received treatment for 12 weeks followed by a two-week treatment free period.
The protocol-specified primary efficacy measure was daily stool consistency. Statistical analysis of the primary endpoint found no significant differences between placebo and any of the crofelemer dose groups (p ³ 0.1434) and no significant dose relationship was seen with regard to change from Baseline to Month 3 in stool consistency scores (p = 0.1165) in the ITT population.
A supplementary analysis of Rome Foundation-defined stool consistency and abdominal pain showed positive results. Responders were subjects who had stool consistency score of ³ 4 for < 25% of days in a given week and ³ 30% improvement in abdominal pain scores a given week (i.e., Rome Foundation-defined stool consistency and abdominal pain responders).
When we look at a supplemental analysis at a reduction in a composite abdominal pain/stool consistency endpoint, the regulatory endpoint in accordance with FDA guidance, we see at the 125 mg dose bid a significant 15% difference with just women patients compared to placebo; and a significant 11% when we include both men and women. The current D-IBS products on the market have a 7-8% reduction (Viberzi and Xifaxan).
In this analysis, Rome Foundation-defined stool consistency and abdominal pain responders were significantly more likely during the entire 3 months in the 125 mg BID group when compared with placebo (24.2% versus 13.1%, p = 0.0399) and there was a statistical trend in favor of crofelemer 125 mg BID during Months 1 through 2 (27.4% versus 16.4%, p = 0.0640). Similar positive effects of crofelemer 125 mg BID were observed in female subjects (n = 183). When the supplementary analysis was applied to the female patients, crofelemer at a dose of 125 mg BID was superior to placebo at Month 3 (26.1% vs 10.9%, p=0.0337).
- •
- Results: The 125mg bid of crofelemer exhibited a consistent response during each month among most efficacy endpoints in women with d-IBS
reaching statistical significance (p<0.05) for pain.
- •
- Crofelemer had little effect on the stool consistency score, though there was a trend toward reduced stool frequency.
- •
- Treatment benefits were not apparent in men, although relatively few men enrolled in the trial (13-16/group).
- •
- As with previous trials of crofelemer, no drug-related serious adverse events were reported. Adverse event rates were similar across all dose groups, although in the two highest doses (250
72
and 500 mg bid) there were a higher percentage of dropouts. There were no drug-related or dose-related differences in constipation. During the two-week treatment-free follow-up period symptoms approached baseline levels.
Safety: Crofelemer at doses of 125, 250 and 500 mg had a safety profile that was generally similar to placebo among men and women with d-IBS.
Phase 2—A Randomized, double-blind, placebo-controlled study to assess the safety and efficacy of crofelemer for the symptomatic treatment of diarrhea predominant irritable bowel syndrome (d-IBS) in 240 female subjects 18 years or older with active d-IBS according to the Rome II criteria for the diagnosis of d-IBS.
The study consisted of a 2-week screening period and a 12-week blinded treatment period followed by a 4-week treatment-free follow-up period. During the 12-week treatment period 240 subjects were given 125 mg of crofelemer BID or placebo BID and recorded daily assessments of their IBS symptoms in the interactive voice response system.
The primary endpoint was the change from baseline for overall percentage of abdominal pain/discomfort free days (PFDs). On a daily basis, respondents recorded the intensity of their abdominal pain/discomfort for that day using the 5-pint Likert scale: 0=none, 1=mild, 2=moderate, 3=intense, 4=severe. Any day that a score of zero (0) was recorded was considered a PFD.
Stool consistency and abdominal pain endpoints were analyzed using definitions of symptom improvement from a recent FDA guidance on IBS endpoints (March 2010) and recommendations of the Rome Foundation (letter dated 28 June 2010) concerning the IBS endpoints described in this guidance.
Results: The overall increase in pain-free days (protocol-specified primary endpoint) for subjects in the crofelemer group was not statistically significant when compared with subjects in the placebo group (p = 0.5107)
A supplementary analysis of abdominal pain showed positive results. Responders were subjects who had ³ 30% improvement in abdominal pain scores a given week (i.e., FDA-defined abdominal pain responders; this definition of abdominal pain responders was presented in the March 2010 guidance on IBS endpoints).
In this analysis, abdominal pain responders were significantly more likely during Months 1 through 2 (58.3% versus 45.0%, p = 0.0303) and during the entire 3 months (54.2% versus 42.5%, p = 0.0371) in the crofelemer group when compared to placebo.
Safety: The overall safety profile for crofelemer 125 mg BID for 12 weeks was comparable to that observed with placebo and was consistent with the IBS population under study.
Rare Pediatric Disease Indications: Congenital Diarrheal Disorders and Short Bowel Syndrome (SBS)
Congenital diarrheal disorders (CDD) are a group of rare, chronic intestinal channel diseases, occurring in early infancy, that are characterized by severe, lifelong diarrhea and a lifelong need for nutritional intake either parenterally or with a feeding tube. CDDs are related to specific genetic defects inherited as autosomal recessive traits, and the incidence of CDDs is much more prevalent in regions where consanguineous marriage is part of the culture. CDDs are directly associated with serious secondary conditions including dehydration, metabolic acidosis, and failure to thrive, prompting the need for immediate therapy to prevent death and limit lifelong disability.
73
Potential Orphan-Drug: Congenital Diarrheal Disorders (CDD) & Short Bowel Syndrome (SBS)
Clinical Study—CDD
We have completed safety studies of crofelemer in children as young as 3 months of age, and Napo has accepted a request for support submitted by Dr. Mohamad Miqdady, Chief of Pediatric Gastroenterology, Hepatology and Nutrition at Sheikh Khalifa Medical City (SKMC) in Abu Dhabi, for an investigator-initiated trial of crofelemer, the active pharmaceutical ingredient in Mytesi, for CDD in children.
A pre-clinical study in mice, conducted by an independent third-party investigator, is underway to support possible orphan-drug designation for crofelemer for Congenital Diarrheal Disorders (CDD). This animal model study is examining the effects of crofelemer on diarrhea caused by microvillous inclusion disease (MVID), a very rare autosomal recessive disorder which belongs to the CDD category.
SBS is a complex condition characterized by malabsorption of fluids and nutrients due to congenital deficiencies or surgical resection of small bowel segments. Consequently, patients suffer from symptoms such as debilitating diarrhea, malnutrition, dehydration and imbalances of fluids and salts. This could be due to either a genetic disorder or premature birth. In countries such as the United Arab Emirates and Saudi Arabia, SBS occurs with much higher incidence. Napo recently visited with medical centers in this region.
We have received orphan-drug status for Mytesi (crofelemer) for the SBS pediatric indication and are pursuing orphan-drug status for CDD. The mission of the FDA Office of Orphan Products Development is to advance the evaluation and development of products (drugs, biologics, devices, or medical foods) that demonstrate promise for the diagnosis and/or treatment of rare diseases or conditions.
IBD—Supportive Care:
Key opinion leaders ("KOLs") identified an unmet need to treat diarrhea in IBD patients, particularly in specific subsets of patients. KOLs felt all IBD patients who undergo ileal pouch-anal anastomosis (IPAA) surgery suffer severe, chronic diarrhea following the procedure. Because this is a highly-motivated patient population with a low placebo-responder risk, we believe a relatively small proof-of-concept trial is the appropriate next step from a development standpoint.
KOLs felt crofelemer's novel mechanism of action may also prove to be an effective treatment for diarrhea that results from bile acid malabsorption, which has been shown to occur in approximately 30% of patients with IBD.
Additionally, KOLs felt crofelemer's novel mechanism of action may prove to be an effective treatment for diarrhea experienced by patients receiving IV infusions of Entyvio, a Takeda Pharmaceuticals prescription medicine used in adults with moderate to severe ulcerative colitis or Crohn's disease. Secretory diarrhea occurs when the intestine does not complete absorption of electrolytes and water from luminal contents. This can happen when a nonabsorbable, osmotically active substance is ingested ("osmotic diarrhea") or when electrolyte absorption is impaired ("secretory diarrhea").
Secretory diarrhea can result from bacterial toxins, luminal secretagogues (such as bile acids or laxatives), reduced absorptive surface area caused by disease or resection, circulating secretagogues (such as various hormones, drugs, and poisons), and medical problems that compromise regulation of intestinal function. These studies in acute diarrhea support the normalizing aspect of the mechanism of action, regardless of the cause of the diarrhea, and are supportive of the supportive care indication under development in IBD patients.
74
Clinical Study
Mytesi has safety studies that support chronic use for the current approved indication, and has demonstrated statistically significant results in multiple supportive care settings, though not specifically in IBD patients. Next steps would include a Phase 2 proof of concept study for supportive care in patients with IBD.
Completed Study—Travelers' Diarrhea (supportive care)
Phase 2—A study of crofelemer in 184 persons in a double-blind, placebo-controlled study for the symptomatic treatment of acute diarrhea among travelers to Jamaica and Mexico.
The study was designed to evaluate the effectiveness of crofelemer in the treatment of travelers' diarrhea.
A total of 184 persons from the United States who acquired diarrhea in Jamaica or Mexico were enrolled in a double-blind, placebo-controlled study examining the effectiveness of three doses of crofelemer in reducing illness. Subjects were treated with 125 mg, 250 mg, or 500 mg crofelemer or a matching placebo four times a day for 2 days. Subjects kept daily diaries of symptoms and were seen each day for 3 days. Of the subjects, 169 (92%) were included in the efficacy analysis.
The most common etiological agent identified was enterotoxigenic Escherichia coli, found in 19% of subjects. The mean time interval from taking the first dose of medication until passage of the last unformed stool during 48-hour therapy (TLUS48) was 38.7 hours for the placebo group.
TLUS48 was shortened by crofelemer:
30.6 h
for the 125-mg dose group (p = 0.005);
30.3 h for the 250-mg group; and
32.6 h for the 500-mg group (p = 0.01).
Treatment failures were seen in 29.3% in the placebo group compared with 7.3% (p = 0.01), 4.3 (p = 0.002), and 9.8 (p = 0.026) in the three treatment groups. Crofelemer was well tolerated at all doses.
The study provided statistically significant results of crofelemer use for shortening the duration of travelers' diarrhea. This antisecretory approach works directly against the pathophysiology of travelers' diarrhea and is not likely to potentiate invasive forms of diarrhea or to produce posttreatment constipation.
Cholera/General Watery Diarrhea
According to the Centers for Disease Control and Prevention of the U.S. Department of Health & Human Services, Cholera is an acute, diarrheal illness caused by infection of the intestine with the bacterium Vibrio cholerae. An estimated 3-5 million cases and over 100,000 deaths occur each year around the world. The infection is often mild or without symptoms, but can sometimes be severe. Approximately one in 10 (5-10%) of infected persons will have severe disease characterized by profuse watery diarrhea, vomiting, and leg cramps. In these people, rapid loss of body fluids leads to dehydration and shock. Without treatment, death can occur within hours. At this time, for example, the largest cholera outbreak in recorded history is occurring in Yemen.
We are investigating SB-300 for the indication of cholera/general watery diarrhea. SB-300 is a distinct and proprietary Napo pharmaceutical formulation of a standardized botanical extract, also sustainably derived from the Croton lechleri tree. We believe SB-300 represents a long-term pipeline opportunity as a second-generation anti-secretory agent, on a global basis, for multiple gastro-intestinal diseases. Additionally, we believe SB-300, which has the same mechanism of action as crofelemer and is
75
significantly less costly to produce, may support efforts to receive a priority review voucher from the U.S. FDA for a cholera indication. Priority review vouchers are granted by the FDA to drug developers as an incentive to develop treatments for neglected diseases and rare pediatric diseases. If approved for this indication, SB-300 could serve as long-term pipeline anti-secretory agent for cholera/general watery diarrhea in geographies where cost of goods is a critical factor, for example, in resource-constrained regions and countries in which a requirement exists for drug prices to decrease annually.
Clinical Study
We have initiated CMC and have multiple animal and human studies in secretory diarrheas with SB-300. We have also completed a successful trial design for cholera with an anti-secretory mechanism of action, published studies with crofelemer in patients with cholera and other acute severe watery diarrhea disease.
Completed Studies—Cholera and Severe Acute Dehydrating Watery Diarrhea
Phase 2 study of crofelemer in the treatment acute, severely dehydrating watery diarrhea with confirmed cholera with the use of an antibiotic (azithromycin) and oral rehydration therapy in 100 adult patients between 18 and 55 in Bangladesh.
A total of 100 adult patients, from Bangladesh, between the ages of 18 and 55, with acute, severely dehydrating watery diarrhea with confirmed cholera were treated with crofelemer on a background of an antibiotic (azithromycin) and oral rehydration therapy. After a four-hour period of rapid rehydration therapy, patients were randomized 1:2:2 to placebo or 125 mg or 250 mg oral dose of crofelemer. Crofelemer or placebo doses were administered about one hour after the oral administration of azithromycin (1 gm dose). The primary objective was to evaluate the safety and effects of crofelemer on reducing the watery stool output normalized to body weight (mL/kg) in the first 24 hours on the background of azithromycin and rehydration therapy. Crofelemer was well tolerated and there were no drug related adverse events in this study. Both doses of crofelemer produced approximately 25-30% reduction in median watery stool volumes in the 0-6 and 0-12 hour period following initiation of therapy. Crofelemer showed a strong trend in the reduction of watery stool output in the 0-6 hour and 0-12 hour intervals (p=0.07). Upon exclusion of three outlier patients, the crofelemer dose of 125 mg produced a statistically significant reduction in the normalized stool output (p=0.028) and the dose of 250 mg crofelemer showed a strong trend for reduction of watery stool output (p=0.07).
In another study, the effects of crofelemer were evaluated in patients with acute dehydrating watery diarrhea caused by various bacterial pathogens, such as enterotoxic strains of Escherichia coli (ETEC) and Vibrio cholerae infection. Crofelemer was evaluated in adult Indian patients with acute watery diarrhea of less than 24-hour duration with suspected bacterial infections, without the use of antibiotics. In this study, patients received oral doses of placebo or crofelemer at a dose of 250 mg every 6 hours for 2 days on the background of oral rehydration therapy only. The use of antibiotics was prohibited in this study. A total of 98 patients were randomized into this study (47 in placebo group and 51 in the crofelemer group). Primary endpoints for this study were changes in stool weight, frequency, consistency, duration of diarrhea. Secondary endpoints included the assessment of clinical symptoms scored as total of 7-item GI index. Clinical success was defined as no diarrhea within 48 hours from study start date and treatment failure was defined as no improvement/worsening of symptoms after 24 hours, fever, bloody stools or dehydration.
Results: 98 patients (51 crofelemer, 47 placebo) were enrolled in the study. 16 patients (4 in the crofelemer group and 12 in the placebo group) used antibiotics and were considered as treatment failures and were excluded from the "per protocol efficacy analysis". Groups were similar in age, weight, vital signs, stool frequency, consistency, dehydration and GI index.
76
The crofelemer group had improvement over baseline and compared to placebo at day 3. More specifically, crofelemer showed superior effects in reducing stool weight (61% vs 11%), stool frequency (65% vs 21%), reversion to soft stool (92% vs 49%) and improved the 7-item GI index (70% C vs 33% P), (all p<0.05).
Crofelemer was well tolerated with no related serious adverse events or concerning changes in lab values. Progression to dehydration and report of fecal incontinence was more common in placebo group (p<0.05).
Conclusions: Clinical success (cessation of diarrhea within 48 hours of 1st dose) was achieved in 79% of crofelemer patients compared to 28% placebo patients (p<0.05).
Other Product Potential Future Indications
Institutional Diarrhea
Patients in medical institutions such as hospitals often experience diarrhea following infection with Clostridium difficile, an anaerobic bacillus shed in feces. According to the Centers for Disease Control and Prevention of the U.S. Department of Health & Human Services, any surface, device, or material (e.g., commodes, bathing tubs, and electronic rectal thermometers) that becomes contaminated with feces may serve as a reservoir for the C. difficile spores, which are transferred to patients mainly via the hands of healthcare personnel who have touched a contaminated surface or item. We believe development of an approved formulation of crofelemer for use in C. difficile has the potential to help patients infected with C. difficile leave the hospital sooner, help keep patients infected with C. difficile out of the hospital, and aid in controlling C. difficile contagion in institutional settings, which would also represent a significant economic benefit.
Competition
There are several significantly larger pharmaceutical companies competing with us in the gastrointestinal segment. These companies include Valeant Pharmaceuticals International, Merck & Co., Inc., and Allergan plc as well as smaller pharmaceutical companies.
Diarrhea in adult patients living with HIV/AIDs. We are not aware of any other FDA-approved drugs for the symptomatic relief of diarrhea in HIV/AIDs patients. HIV/AIDs patients also use loperamide and over the counter anti-diarrheal remedies such as Mylanta or Kaopectate to treat their diarrhea, but these medicines affect motility and can result in rebound diarrhea.
Diarrhea predominant irritable bowel syndrome. Two drugs were approved in 2015 for the treatment of diarrhea predominant irritable bowel syndrome, Allergan plc's Virbezi and Xifaxan which is marketed by Valeant Pharmaceuticals International. Also, Lotronex was approved by the FDA in 2000 but was withdrawn from the market and later reintroduced in 2002 under a Risk Management Program. With the exception of Lotronex, the sponsors of Verbezi and Xifaxan employ extensive media and print promotion for the commercialization of these products. We are seeking a partner to further the clinical development and commercialization of crofelemer for d-IBS. There are currently numerous trials on going for d-IBS.
Pediatric diarrhea. Acute diarrhea in children is commonly treated by a change in diet, oral rehydration therapy and/or antibiotics, assuming the cause of the diarrhea is bacterial in nature. Children aged 12 and younger are advised not to use anti-motility drugs (loperamide for example) unless directed to do so by a physician. There are recent clinical trials for probiotics and zinc sulfate. Other recent anti-diarrheal studies in children include a safety and tolerability study of Fidaxomicin for C difficile associated diarrhea.
77
Chemotherapy induced diarrhea. We are not aware of any FDA-approved drugs specifically indicated for chemotherapy induced diarrhea. A recent Phase IIb trial of elsiglutide for the treatment of chemotherapy induced diarrhea in colorectal cancer patients did not meet statistical significance. Opioids and over the counter drugs are commonly used to treat chemotherapy induced diarrhea, but these drugs affect motility. Certain tyrosine-kinase inhibitor chemotherapy agents have diarrhea as a significant side effect. For example, FDA guidance suggests diarrhea prophylaxis prior to initiating adjuvant therapy with neratinib.
Congenital Diarrheal Disorders and Short Bowel Syndrome. We are not aware of any FDA-approved drugs specifically indicated for Congenital Diarrheal Disorders and Short Bowel Syndrome.
Cholera. We are not aware of any FDA-approved drugs specifically indicated as an anti-secretory agent for use to address the devastating dehydration in cholera patients.
Irritable Bowel Syndrome (IBS). If we receive regulatory approval for Mytesi for IBS, we expect to compete with major pharmaceutical and biotechnology companies that operate in the gastrointestinal space, such as Sucampo AG, Takeda Pharmaceuticals, Allergan, Inc., Ironwood Pharmaceuticals, Inc., Synergy Pharmaceuticals Inc., Sebela Pharmaceuticals, Inc. and Salix Pharmaceuticals. Because Mytesi is approved with chronic safety and several of the other agents have safety concerns, there is likely to be an opportunity for a polypharmacetuical approach to long-term management of these patients, removing a direct competitive scenario from Mytesi's potential entry to the marketplace and disease indication.
To our knowledge, there are currently no FDA-approved anti-secretory products, in particular which act locally in the gut with the chronic safety profile of crofelemer, in development or on the market. Crofelemer represents a new tool in gastro-intestinal disease management.
Distribution and Marketing Agreements
Napo has agreements in place with BexR, a distributor in Texas, and SmartPharma, a marketing and commercialization advisory firm, for the distribution, marketing and sale of Mytesi. The agreements compensate these parties with a percentage of net sales, as defined. Payments by Napo to BexR will be a specified percentage of net sales, ranging in the low double digits but in no instance exceeding 20% of net sales, depending on the period in which the sales occur and the amount of such sales. Payments by Napo to SmartPharma will be a specified percentage of net sales, ranging in the low double digits but in no instance exceeding 20% of net sales, depending on the amount of such sales. In addition, under certain circumstances, Napo will be required to pay SmartPharma a termination fee equal to a certain percentage of net sales generated within a specified period after the termination date.
Manufacturing
The plant material used to manufacture is crude plant latex ("CPL") extracted and purified from Croton lechleri, a widespread and naturally regenerating tree in the rainforest that is managed as part of sustainable harvesting programs. The tree is found in several South American countries and has been the focus of long-term sustainable harvesting research and development work. Napo's collaborating suppliers obtain CPL and arrange for the shipment of CPL to Napo's third-party contract manufacturer.
Napo's third-party contract manufacturer, India-based Glenmark Pharmaceuticals Ltd. (Glenmark), a research-driven, global, integrated pharmaceutical company, is Napo's primary manufacturer of crofelemer, the active pharmaceutical ingredient in Mytesi. Glenmark processes CPL into crofelemer utilizing a proprietary manufacturing process. The processing occurs at two FDA-approved Glenmark
78
facilities. Additionally, Napo plans to establish a third processing site, which will be operated by Indena S.p.A., a Milan, Italy-based contract manufacturer dedicated to the identification, development and production of high-quality active principles derived from plants, for use in the pharmaceutical, health food and personal care industries. Indena has completed the required technology transfer and pilot manufacturing and has the equipment in place for the initiation of commercial scale manufacturing.
Canalevia will be manufactured by the same process used to manufacture the API that was used in the animal safety studies and the human studies in support of the approval of Mytesi. Napo has also licensed this intellectual property to third parties in connection with its agreements related to the manufacture of crofelemer.
In May 2014 and June 2014, and as amended in February 2015, we entered into binding memorandums of understanding with Indena S.p.A. to negotiate a definitive commercial supply agreement for the manufacture of crofelemer and the botanical extract, SB-300.
We have contracts in place with all the manufacturers and third party testing labs required to manufacture Mytesi and SB-300. We are finalizing a master service agreement with Glenmark for the manufacture of crofelemer which addresses cost of goods reductions at increasing scale. We are in the process of evaluating alternative and secondary third parties to reduce costs associated with finished product manufacture and the assays necessary to the release specifications of Mytesi.
Proprietary Library of Medicinal Plants
We possess a proprietary library of more than 2,300 medicinal plants.
Intellectual Property
Trademarks
We plan to market all of our products under a trademark or trademarks we select and we will own all rights, title and interest, including all goodwill, associated with such trademarks. Mytesi is a registered trademark owned by Napo.
License Agreements
Termination, Asset Transfer and Transition Agreement
On September 19, 2017 (the "Transfer Date"), Napo entered into the Termination, Asset Transfer and Transition Agreement (the "Glenmark Transition Agreement") with Glenmark. The Glenmark Transition Agreement supersedes the Glenmark Collaboration Agreement and returns to Napo certain rights which Napo licensed to Glenmark pursuant to the Glenmark Collaboration Agreement related to the development and commercialization of crofelemer for certain specified human indications in India and 140 other countries largely in developing regions (the "Transferred Assets").
As a result of the execution of the Glenmark Transition Agreement, we, through Napo, now control commercial rights for Mytesi for all indications, territories and patient populations globally, and also hold commercial rights to the existing regulatory approvals for crofelemer in Brazil, Ecuador, Zimbabwe and Botswana.
In consideration for Glenmark's assignment and transfer of the Transferred Assets to Napo, Napo agreed to pay Glenmark in cash, within 45 days after receipt by Napo, 25% of any payment that Napo receives from a third party to whom Napo grants a license or sublicense or with whom Napo partners in respect of, or sells or otherwise transfers the certain specified human indications in India and 140 other countries largely in developing regions any of the Transferred Assets, subject to certain limitations, until Glenmark has received a total of $7 million. As additional consideration for the
79
assignment and transfer of the Transferred Assets, Napo agreed (i) to enter into, within 90 days after the Transfer Date, a manufacturing and supply agreement with Glenmark for crofelemer, which will be manufactured at either or both of Glenmark's facilities in India (this master service agreement is in final draft form, though not yet fully executed) and (ii) to transfer and assign to Glenmark all right, title and interest in and to certain required dedicated equipment used to manufacture crofelemer located at Glenmark's Ankleshwar facility, subject to certain limitations.
Patent Portfolio
Napo
Napo owns a portfolio of patents and patent applications covering formulations of and methods of treatment with proanthocyanidin polymers isolated from Croton spp. or Calophyllum spp., including Mytesi (crofelemer). The patent family associated with International Patent publication WO1998/16111 relates to enteric protected formulations of proanthocyanidin polymers isolated from Croton spp. or Calophyllum spp., including crofelemer, and methods of treating watery diarrhea using these enteric protected formulations. There is one U.S. patent in force in this family, US 7,341,744, which has a term until at least June 23, 2019. Napo has applied to extend the term of US 7,341,744 under 35 U.S.C. 156, and the United States Patent and Trademark Office has issued a Notice of Final Determination that the patent term extension for US 7,341,744 is 1,075 days. Based upon the June 23, 2019 expiration date, the patent term would be extended to June 2, 2022 once the patent term extension certificate issues, to account for the regulatory delay in obtaining human marketing approval for crofelemer.
Napo additionally owns a family of patents arising from International Patent Application Publication WO2012/058664 that cover methods of treating HIV-associated diarrhea and HAART-associated diarrhea with proanthocyanidin polymers isolated from Croton spp. or Calophyllum spp., including crofelemer. In the U.S., there are two issued patents, US 8,962,680 and US 9,585,868, both of which expire October 31, 2031. Outside the U.S., patent protection for methods of treating HIV-associated diarrhea has been obtained in Australia, Europe, Hong Kong, Japan, Kenya, Mexico, Russia, Ukraine, South Africa, and Zimbabwe, with expiration dates of October 31, 2031, and applications are pending in Brazil, Hong Kong, Canada, China, India, Mexico, and Malaysia. Napo also has patent families related to methods of treating diarrhea-predominant irritable bowel syndrome, methods of treating constipation-predominant irritable bowel syndrome, and methods of treating inflammatory bowel disease, familial adenomatous polyposis and colon cancer, with proanthocyanidin polymers isolated from Croton spp. or Calophyllum spp., including crofelemer. In particular, for diarrhea-predominant irritable bowel syndrome, Napo has two issued U.S. patents, US 8,846,113 and US 9,980,938, which expire on February 9, 2027, as well as issued patents in Australia, Europe, Hong Kong, Japan, South Korea, Mexico, New Zealand, Singapore, and Taiwan and pending applications in Bangladesh, Bolivia, Canada, Chile, Gulf States, Mexico, Panama, Peru, Paraguay, Thailand, and Venezuela, all of which are estimated to expire April 30, 2027; for constipation-predominant irritable bowel syndrome, Napo has three issued U.S. patents, with terms to at least April 30, 2027, patents in Australia, Canada, Europe, Hong Kong, Mexico, New Zealand, and Singapore, and a pending application in India, all of which are estimated to expire April 30, 2027; and for inflammatory bowel disease, familial adenomatous polyposis and/or colon cancer, Napo has two issued U.S. patents, US 8,852,649 and US 9,987,250 with terms until at least January 4, 2028, as well as issued patents in Australia, Hong Kong, and Europe and an allowed application in Canada, which have estimated expiration dates of April 30, 2027. Napo has a pending U.S. non provisional application for the treatment of chemotherapy induced diarrhea (CID) with crofelemer filed on March 9, 2018 and two International Patent Applications on other human indications including for the treatments of short bowel syndrome and congenital diarrhea disorder filed on May 31, 2018.
For methods of manufacturing proanthocyanidin polymers isolated from Croton spp. or Calophyllum spp., including crofelemer, Napo owns issued patents in India, South Africa, and Eurasia
80
with terms at least until August 26, 2029. Napo also owns issued patents in India, Russia, and South Africa and pending applications in Argentina, Brazil, and Venezuela that also cover methods of manufacturing proanthocyanidin polymers isolated from Croton spp. or Calophyllum spp., including crofelemer, with terms at least until January 17, 2032. Napo holds a patent in South Africa and pending applications in Argentina, Bangladesh, Brazil, Egypt, India, Indonesia, Kenya, Malaysia, Mexico, Nigeria, Philippines, United Arab Emirates, Russia, Thailand, Venezuela, and Vietnam covering non-enteric formulations of crofelemer estimated to expire August 17, 2032. Lastly, Napo owns two U.S. patents covering a formulation of NP 500 (nordihydroguiaretic acid (NDGA)) and its use in treating a metabolic disorder that have terms until April 23, 2031.
Government Regulation
The FDA and comparable regulatory authorities in state and local jurisdictions and in other countries impose substantial and burdensome requirements upon companies involved in the clinical development, manufacture, marketing and distribution of prescription drugs such as those Napo is developing. These agencies and other federal, state and local entities regulate, among other things, the research and development, testing, manufacture, quality control, safety, effectiveness, labeling, storage, record keeping, approval, advertising and promotion, distribution, post-approval monitoring and reporting, sampling and export and import of Napo's drug product candidates. To comply with the regulatory requirements in each of the jurisdictions in which Napo is seeking to market and subsequently sell its prescription products, Napo is establishing processes and resources to provide oversight of the development, approval processes and launch of its products and to position those products in order to gain market share.
81
U.S. Government Regulation
In the United States, the FDA approves and regulates drugs under the Federal Food, Drug, and Cosmetic Act ("FDCA"), and its implementing regulations.
The process of obtaining regulatory approvals and the subsequent compliance with applicable federal, state, local and foreign statutes and regulations requires the expenditure of substantial time and financial resources. Failure to comply with the applicable U.S. requirements at any time during the product development process, approval process or after approval, may subject an applicant to a variety of administrative or judicial sanctions, such as the FDA's refusal to approve pending NDAs, withdrawal of an approval, imposition of a clinical hold, issuance of warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement or civil or criminal penalties.
The process required by the FDA before a drug may be marketed in the United States generally involves the following:
- •
- completion of pre-clinical laboratory tests, animal studies and formulation studies in compliance with the FDA's good laboratory practice, or
GLP, regulations;
- •
- submission to the FDA of an investigational new drug application, or IND, which must become approved before human clinical trials may begin;
- •
- approval by an institutional review board, or IRB, of the study protocol and informed consent forms for the clinical site before each trial may
be initiated. Multiple sites may necessitate the involvement of multiple IRBs and submissions;
- •
- performance of adequate and well-controlled human clinical trials in accordance with good clinical practice, or GCP, requirements to establish
the safety and efficacy of the proposed drug product for each indication;
- •
- submission to the FDA of an NDA which would include the study reports of the clinical trials, chemistry and manufacturing of the active
pharmaceutical ingredient and the final dosage form as well as other required sections to be included in the NDA;
- •
- satisfactory completion of an FDA advisory committee review, if applicable;
- •
- satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced to assess compliance
with current good manufacturing practice, or cGMP, requirements and to assure that the facilities, methods and controls are adequate to preserve the drug's identity, strength, quality and purity; and
- •
- FDA review and approval of the NDA.
Pre-clinical Studies
Pre-clinical studies include laboratory evaluation of the drug product's chemistry, toxicity and formulation, as well as animal studies to assess potential safety and efficacy. An IND sponsor must submit the results of the pre-clinical tests, together with manufacturing information, analytical data and any available clinical data or literature, among other things, to the FDA as part of an IND. Some pre-clinical testing may continue even after the IND is submitted. An IND automatically becomes effective 30 days after receipt by the FDA, unless before that time the FDA raises concerns or questions related to one or more proposed clinical trials and places the clinical trial on a clinical hold. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. As a result, submission of an IND may not result in the FDA allowing clinical trials to commence.
82
Clinical Trials
Clinical trials involve the administration of the investigational new drug to human subjects pursuant to a clinical protocol, under the supervision of qualified investigators in accordance with GCP requirements, which include the requirement that all research subjects provide their informed consent in writing for their participation in any clinical trial. Clinical trials are conducted under protocols detailing, among other things, the objectives or endpoints of the trial, the parameters to be used in monitoring safety, and the effectiveness criteria to be evaluated. A protocol for each clinical trial and any subsequent protocol amendments must be submitted to the FDA under the IND. In addition, an IRB at each institution participating in the clinical trial must review and approve the plan for any clinical trial before it commences at that institution. Information about certain clinical trials must be submitted within specific timeframes to the National Institutes of Health, or NIH, for public dissemination on their www.clinicaltrials.govwebsite.
Human clinical trials are typically conducted in three sequential phases, which may overlap or be combined:
- •
- Phase 1: The drug is initially introduced into healthy human subjects or patients with the target disease or condition and tested for
safety, dosage tolerance, absorption, metabolism, distribution, excretion and, if possible, to gain an early indication of its effectiveness depending on the endpoints set forth in the protocol.
- •
- Phase 2: The drug is administered to a limited patient population to identify possible adverse effects and safety risks, to
preliminarily evaluate the efficacy of the product for specific targeted diseases and to determine dosage tolerance and optimal dosage.
- •
- Phase 3: The drug is administered to an expanded patient population, generally at geographically dispersed clinical trial sites, in well-controlled clinical trials to generate enough data to statistically evaluate the efficacy and safety of the product for approval, to establish the overall risk-benefit profile of the product, and to provide adequate information for the labeling of the product.
Progress reports detailing the results of the clinical trials must be submitted at least annually to the FDA and more frequently if serious adverse events occur. Phase 1, Phase 2 and Phase 3 clinical trials may not be completed successfully within any specified period, or at all. Furthermore, the FDA or the sponsor may suspend or terminate a clinical trial at any time on various grounds, including a finding that the research subjects are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB's requirements or if the drug has been associated with unexpected serious harm to patients.
Special Protocol Assessment
The special protocol assessment, or SPA, process is designed to facilitate the FDA's review and approval of drugs by allowing the FDA to evaluate issues related to the adequacy of certain clinical trials, including Phase 3 clinical trials that can form the primary basis for a drug product's efficacy claim in an NDA. Upon specific request by a clinical trial sponsor, the FDA will evaluate the protocol and respond to a sponsor's questions regarding, among other things, primary efficacy endpoints, trial conduct and data analysis, within 45 days of receipt of the request.
The FDA ultimately assesses whether the protocol design and planned analysis of the trial are acceptable to support regulatory approval of the product candidate with respect to effectiveness of the indication studied. All agreements and disagreements between the FDA and the sponsor regarding an SPA must be clearly documented in an SPA letter or the minutes of a meeting between the sponsor and the FDA.
83
Even if the FDA agrees to the design, execution and analyses proposed in protocols reviewed under the SPA process, the FDA may revoke or alter its agreement under the following circumstances:
- •
- public health concerns emerge that were unrecognized at the time of the protocol assessment;
- •
- the director of the review division determines that a substantial scientific issue essential to determining safety or efficacy has been
identified after testing has begun;
- •
- a sponsor fails to follow a protocol that was agreed upon with the FDA; or
- •
- the relevant data, assumptions, or information provided by the sponsor in a request for SPA change, are found to be false statements or misstatements, or are found to omit relevant facts.
A documented SPA may be modified, and such modification will be deemed binding on the FDA review division, except under the circumstances described above, if FDA and the sponsor agree in writing to modify the protocol and such modification is intended to improve the study.
Marketing Approval
Assuming successful completion of the required clinical testing, the results of the pre-clinical studies and clinical trials, together with detailed information relating to the product's chemistry, manufacture, controls and proposed labeling, among other things, are submitted to the FDA as part of a NDA requesting approval to market the product for one or more indications. In most cases, the submission of a NDA is subject to a substantial application user fee. Under the Prescription Drug User Fee Act ("PDUFA"), guidelines that are currently in effect, the FDA has a goal of ten months from the date of "filing" of a standard NDA for a new molecular entity to review and act on the submission. This review typically takes twelve months from the date the NDA is submitted to FDA because the FDA has approximately two months to make a "filing" decision.
In addition, under the Pediatric Research Equity Act of 2003 ("PREA"), as amended and reauthorized, certain NDAs or supplements to an NDA must contain data that are adequate to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations; this would include information which supports dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults or full or partial waivers from the pediatric data requirements.
The FDA may also require submission of a risk evaluation and mitigation strategy, or REMS, plan to ensure that the benefits of the drug outweigh its risks. The REMS plan could include medication guides, physician communication plans, assessment plans, and/or elements to assure safe use, such as restricted distribution methods, patient registries, or other risk minimization tools.
The FDA may also require other information as part of the NDA filing such as an environmental impact statement. The FDA can waive some or delay compliance with some of these requirements.
The FDA conducts a preliminary review of all NDAs within the first 60 days after submission, before accepting them for filing, to determine whether they are sufficiently complete to permit substantive review. The FDA may request additional information rather than accept a NDA for filing. In this event, the application must be resubmitted with the additional information. The resubmitted application is also subject to review before the FDA accepts it for filing. Once the submission is accepted for filing, the FDA begins an in-depth substantive review. The FDA reviews a NDA to determine, among other things, whether the drug is safe and effective and whether the facility in which it is manufactured, processed, packaged or held meets standards designed to assure the product's continued safety, quality and purity.
84
The FDA may refer an application for a novel drug to an advisory committee. An advisory committee is a panel of independent experts, including clinicians and other scientific experts, that reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
Before approving an NDA, the FDA typically will inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving a NDA, the FDA may inspect one or more clinical trial sites to assure compliance with GCP requirements.
After evaluating the NDA and all related information, including the advisory committee recommendation, if any, and inspection reports regarding the manufacturing facilities and clinical trial sites, the FDA may issue an approval letter, or, in some cases, a complete response letter. A complete response letter must contain a statement of specific items that prevent the FDA from approving the application and will also contain conditions that must be met in order to secure final approval of the NDA and may require additional clinical or pre-clinical testing in order for FDA to reconsider the application. Even with submission of this additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval. If and when those conditions have been met to the FDA's satisfaction, the FDA will typically issue an approval letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications.
Even if the FDA approves a product, it may limit the approved indications for use of the product, require that contraindications, warnings or precautions be included in the product labeling, require that post-approval studies, Phase 4 clinical trials, be conducted to further assess a drug's safety after approval, require testing and surveillance programs to monitor the product after commercialization, or impose other conditions, including distribution and use restrictions or other risk management mechanisms under a REMS, which can materially affect the potential market and profitability of the product. The FDA may prevent or limit further marketing of a product based on the results of post-marketing studies or surveillance programs. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes, and additional labeling claims, are subject to further testing requirements and FDA review and approval.
Post-Approval Requirements
Drugs manufactured or distributed pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to recordkeeping, periodic reporting, product sampling and distribution, advertising and promotion and reporting of adverse experiences with the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims are subject to prior FDA review and approval. New secondary indications must be supported by clinical trials or data. There also are continuing, annual user fee requirements for any marketed products and the establishments at which such products are manufactured, as well as new application fees for supplemental applications with clinical data.
The FDA may impose a number of post-approval requirements as a condition of approval of an NDA. For example, the FDA may require post-marketing testing, including Phase 4 clinical trials, and surveillance to further assess and monitor the product's safety and effectiveness after commercialization.
In addition, drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and state agencies, and are
85
subject to periodic unannounced inspections by the FDA and these state agencies for compliance with cGMP requirements. Changes to the manufacturing process are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP requirements and impose reporting and documentation requirements upon the sponsor and any third-party manufacturers that the sponsor may decide to use. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to maintain cGMP compliance.
Once an approval is granted, the FDA may withdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in mandatory revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical trials to assess new safety risks; or imposition of distribution or other restrictions under a REMS program. Other potential consequences include, include, but are not limited to:
- •
- restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls;
- •
- fines, warning letters or holds on post-approval clinical trials;
- •
- refusal of the FDA to approve pending NDAs or supplements to approved NDAs, or suspension or revocation of product approvals;
- •
- product seizure or detention, or refusal to permit the import or export of products; or
- •
- injunctions or the imposition of civil or criminal penalties.
The FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. Drugs may be promoted only for the approved indications and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability. In addition, the FDA regulates the purity and or consistency of the approved product. Products, if deemed adulterated can lead to serious consequences as set forth above as well as civil and criminal penalties.
Foreign Government Regulation
To the extent that any of Napo's product candidates, once approved, are sold in a foreign country, Napo may be subject to similar foreign laws and regulations, which may include, for instance, applicable post-marketing requirements, including safety surveillance, anti-fraud and abuse laws and implementation of corporate compliance programs and reporting of payments or other transfers of value to healthcare professionals.
In order to market Napo's future products in the EEA (which is comprised of the 28 Member States of the EU plus Norway, Iceland and Liechtenstein) and many other foreign jurisdictions, a sponsor must obtain separate regulatory approvals. More concretely, in the EEA, medicinal products can only be commercialized after obtaining a Marketing Authorization, or MA. There are two types of marketing authorizations:
- •
- the Community MA, which is issued by the European Commission through the Centralized Procedure, based on the opinion of the Committee for Medicinal Products for Human Use of the European Medicines Agency, or EMA, and which is valid throughout the entire territory of the EEA. The Centralized Procedure is mandatory for certain types of products, such as biotechnology medicinal products, orphan medicinal products and medicinal products indicated
86
- •
- National MAs, which are issued by the competent authorities of the Member States of the EEA and only cover their respective territory, are available for products not falling within the mandatory scope of the Centralized Procedure. Where a product has already been authorized for marketing in a Member State of the EEA, this National MA can be recognized in another Member State through the Mutual Recognition Procedure. If the product has not received a National MA in any Member State at the time of application, it can be approved simultaneously in various Member States through the Decentralized Procedure.
for the treatment of AIDS, cancer, neurodegenerative disorders, diabetes, auto-immune and viral diseases. The Centralized Procedure is optional for products containing a new active substance not yet authorized in the EEA, or for products that constitute a significant therapeutic, scientific or technical innovation or which are in the interest of public health in the EU; and
Under the above described procedures, before granting the MA, the EMA or the competent authorities of the Member States of the EEA make an assessment of the risk-benefit balance of the product on the basis of scientific criteria concerning its quality, safety and efficacy.
In the EEA, new products authorized for marketing, or reference products, qualify for eight years of data exclusivity and an additional two years of market exclusivity upon marketing authorization. The data exclusivity period prevents generic or biosimilar applicants from relying on the pre-clinical and clinical trial data contained in the dossier of the reference product when applying for a generic or biosimilar marketing authorization in the EU during a period of eight years from the date on which the reference product was first authorized in the EU. The market exclusivity period prevents a successful generic or biosimilar applicant from commercializing its product in the EU until 10 years have elapsed from the initial authorization of the reference product in the EU. The 10-year market exclusivity period can be extended to a maximum of eleven years if, during the first eight years of those 10 years, the marketing authorization holder obtains an authorization for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are held to bring a significant clinical benefit in comparison with existing therapies.
In the EEA, marketing authorization applications for new medicinal products not authorized have to include the results of studies conducted in the pediatric population, in compliance with a pediatric investigation plan, or PIP, agreed with the EMA's Pediatric Committee ("PDCO"). The PIP sets out the timing and measures proposed to generate data to support a pediatric indication of the drug for which marketing authorization is being sought. The PDCO can grant a deferral of the obligation to implement some or all of the measures of the PIP until there are sufficient data to demonstrate the efficacy and safety of the product in adults. Further, the obligation to provide pediatric clinical trial data can be waived by the PDCO when these data is not needed or appropriate because the product is likely to be ineffective or unsafe in children, the disease or condition for which the product is intended occurs only in adult populations, or when the product does not represent a significant therapeutic benefit over existing treatments for pediatric patients. Once the marketing authorization is obtained in all Member States of the EU and study results are included in the product information, even when negative, the product is eligible for six months' supplementary protection certificate extension. For orphan-drug designated medicinal products, the 10-year period of market exclusivity is extended to 12 years.
Other U.S. Healthcare Laws
In addition to FDA restrictions on marketing of pharmaceutical and biological products, other U.S. federal and state healthcare regulatory laws restrict business practices in the pharmaceutical industry, which include, but are not limited to, state and federal anti-kickback, false claims, data privacy and security and physician payment and drug pricing transparency laws.
87
The U.S. federal Anti-Kickback Statute prohibits, among other things, any person or entity from knowingly and willfully offering, paying, soliciting, receiving or providing any remuneration, directly or indirectly, overtly or covertly, to induce or in return for purchasing, leasing, ordering, or arranging for or recommending the purchase, lease, or order of any good, facility, item or service reimbursable, in whole or in part, under Medicare, Medicaid or other federal healthcare programs. The term "remuneration" has been broadly interpreted to include anything of value. The Anti-Kickback Statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers, formulary managers and beneficiaries on the other. Although there are a number of statutory exceptions and regulatory safe harbors protecting some common activities from prosecution, the exceptions and safe harbors are drawn narrowly. Practices that involve remuneration that may be alleged to be intended to induce prescribing, purchases, or recommendations may be subject to scrutiny if they do not meet the requirements of a statutory or regulatory exception or safe harbor. Failure to meet all of the requirements of a particular applicable statutory exception or regulatory safe harbor does not make the conduct per se illegal under the U.S. federal Anti-Kickback Statute. Instead, the legality of the arrangement will be evaluated on a case-by-case basis based on a cumulative review of all its facts and circumstances. Several courts have interpreted the statute's intent requirement to mean that if any one purpose of an arrangement involving remuneration is to induce referrals of federal healthcare covered business, the statute has been violated.
Additionally, the intent standard under the U.S. federal Anti-Kickback Statute was amended by the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010, or collectively, the ACA, to a stricter standard such that a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. In addition, the ACA codified case law that a claim including items or services resulting from a violation of the U.S. federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal civil False Claims Act. The majority of states also have anti-kickback laws, which establish similar prohibitions and in some cases may apply to items or services reimbursed by any third-party payor, including commercial insurers.
The federal false claims and civil monetary penalties laws, including the civil False Claims Act, prohibit any person or entity from, among other things, knowingly presenting, or causing to be presented, a false, fictitious or fraudulent claim for payment to, or approval by, the federal government, knowingly making, using, or causing to be made or used a false record or statement material to a false or fraudulent claim to the federal government, or from knowingly making a false statement to avoid, decrease or conceal an obligation to pay money to the U.S. federal government. A claim includes "any request or demand" for money or property presented to the U.S. government. Actions under the civil False Claims Act may be brought by the Attorney General or as a qui tam action by a private individual in the name of the government. Violations of the civil False Claims Act can result in very significant monetary penalties and treble damages. Several pharmaceutical and other healthcare companies have been prosecuted under these laws for, among other things, allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product. Other companies have been prosecuted for causing false claims to be submitted because of the companies' marketing of products for unapproved, or off-label, uses. Companies also have been prosecuted for allegedly violating the Anti-Kickback Statute and False Claims Act as a result of impermissible arrangements between companies and healthcare practitioners or as a result of the provision of remuneration by the companies to the healthcare practitioners. In addition, the civil monetary penalties statute imposes penalties against any person who is determined to have presented or caused to be presented a claim to a federal health program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent. Many states also have similar fraud and abuse statutes or regulations that apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor.
88
Violations of fraud and abuse laws, including federal and state anti-kickback and false claims laws, may be punishable by criminal and civil sanctions, including fines and civil monetary penalties, the possibility of exclusion from federal healthcare programs (including Medicare and Medicaid), disgorgement and corporate integrity agreements, which impose, among other things, rigorous operational and monitoring requirements on companies. Similar sanctions and penalties, as well as imprisonment, also can be imposed upon executive officers and employees of such companies. Given the significant size of actual and potential settlements, it is expected that the government authorities will continue to devote substantial resources to investigating healthcare providers' and manufacturers' compliance with applicable fraud and abuse laws.
The federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, created additional federal criminal statutes that prohibit, among other actions, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, including private third-party payors, knowingly and willfully embezzling or stealing from a healthcare benefit program, willfully obstructing a criminal investigation of a healthcare offense and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services. Similar to the U.S. federal Anti-Kickback Statute, the ACA broadened the reach of certain criminal healthcare fraud statutes created under HIPAA by amending the intent requirement such that a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
In addition, there has been a recent trend of increased federal and state regulation of payments made to physicians and certain other healthcare providers. The ACA imposed, among other things, new annual reporting requirements through the Physician Payments Sunshine Act for covered manufacturers for certain payments and "transfers of value" provided to physicians and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members. Failure to submit timely, accurately and completely the required information for all payments, transfers of value and ownership or investment interests may result in civil monetary penalties of up to an aggregate of $150,000 per year and up to an aggregate of $1 million per year for "knowing failures." Covered manufacturers must submit reports by the 90th day of each subsequent calendar year. In addition, certain states require implementation of compliance programs and compliance with the pharmaceutical industry's voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government, impose restrictions on marketing practices and/or tracking and reporting of gifts, compensation and other remuneration or items of value provided to physicians and other healthcare professionals and entities.
Napo may also be subject to data privacy and security regulation by both the federal government and the states in which Napo conducts its business. HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, and their respective implementing regulations, including the Final HIPAA Omnibus Rule published on January 25, 2013, impose specified requirements relating to the privacy, security and transmission of individually identifiable health information held by covered entities and their business associates. Among other things, HITECH made HIPAA's security standards directly applicable to "business associates," defined as independent contractors or agents of covered entities that create, receive, maintain or transmit protected health information in connection with providing a service for or on behalf of a covered entity. HITECH also increased the civil and criminal penalties that may be imposed against covered entities, business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorney's fees and costs associated with pursuing federal civil actions. In addition, state laws govern the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same requirements, thus complicating compliance efforts.
89
Coverage and Reimbursement
Significant uncertainty exists as to the coverage and reimbursement status of any pharmaceutical products for which Napo obtains regulatory approval. In the United States and markets in other countries, patients who are prescribed treatments for their conditions and providers performing the prescribed services generally rely on third-party payors to reimburse all or part of the associated healthcare costs. Patients are unlikely to use Napo's products unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of Napo's products. Sales of any products for which Napo receives regulatory approval for commercial sale will therefore depend, in part, on the availability of coverage and adequate reimbursement from third-party payors. Third-party payors include government authorities, managed care plans, private health insurers and other organizations. In the United States, the process for determining whether a third-party payor will provide coverage for a pharmaceutical or biological product typically is separate from the process for setting the price of such product or for establishing the reimbursement rate that the payor will pay for the product once coverage is approved. Third-party payors may limit coverage to specific products on an approved list, also known as a formulary, which might not include all of the FDA-approved products for a particular indication. A decision by a third-party payor not to cover Napo's product candidates could reduce physician utilization of Napo's products once approved and have a material adverse effect on Napo's sales, results of operations and financial condition. Moreover, a third-party payor's decision to provide coverage for a pharmaceutical or biological product does not imply that an adequate reimbursement rate will be approved. Adequate third-party reimbursement may not be available to enable Napo to maintain price levels sufficient to realize an appropriate return on Napo's investment in product development. Additionally, coverage and reimbursement for products can differ significantly from payor to payor. One third-party payor's decision to cover a particular medical product or service does not ensure that other payors will also provide coverage for the medical product or service, or will provide coverage at an adequate reimbursement rate. As a result, the coverage determination process will require Napo to provide scientific and clinical support for the use of Napo's products to each payor separately and will be a time-consuming process.
In the EEA, governments influence the price of products through their pricing and reimbursement rules and control of national health care systems that fund a large part of the cost of those products to consumers. Some jurisdictions operate positive and negative list systems under which products may only be marketed once a reimbursement price has been agreed to by the government. To obtain reimbursement or pricing approval, some of these countries may require the completion of clinical trials that compare the cost effectiveness of a particular product candidate to currently available therapies. Other member states allow companies to fix their own prices for medicines, but monitor and control company profits. The downward pressure on health care costs in general, particularly prescription products, has become very intense. As a result, increasingly high barriers are being erected to the entry of new products. In addition, in some countries, cross border imports from low-priced markets exert a commercial pressure on pricing within a country.
The containment of healthcare costs has become a priority of federal, state and foreign governments, and the prices of pharmaceutical or biological products have been a focus in this effort. Third-party payors are increasingly challenging the prices charged for medical products and services, examining the medical necessity and reviewing the cost-effectiveness of pharmaceutical or biological products, medical devices and medical services, in addition to questioning safety and efficacy. If these third-party payors do not consider Napo's products to be cost-effective compared to other available therapies, they may not cover Napo's products after FDA approval or, if they do, the level of payment may not be sufficient to allow Napo to sell its products at a profit.
90
Healthcare Reform
A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and other third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medical products. For example, in March 2010, the ACA was enacted, which, among other things, increased the minimum Medicaid rebates owed by most manufacturers under the Medicaid Drug Rebate Program; introduced a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; extended the Medicaid Drug Rebate Program to utilization of prescriptions of individuals enrolled in Medicaid managed care plans; imposed mandatory discounts for certain Medicare Part D beneficiaries as a condition for manufacturers' outpatient drugs coverage under Medicare Part D; subjected drug manufacturers to new annual fees based on pharmaceutical companies' share of sales to federal healthcare programs; created a new Patient Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research; creation of the Independent Payment Advisory Board, once empaneled, will have authority to recommend certain changes to the Medicare program that could result in reduced payments for prescription drugs; and establishment of a Center for Medicare Innovation at the CMS to test innovative payment and service delivery models to lower Medicare and Medicaid spending. Since its enactment, the U.S. federal government has delayed or suspended implementation of certain provisions of the ACA. In addition, there have been judicial and Congressional challenges to certain aspects of the ACA, and Napo expects there will be additional challenges and amendments to the ACA in the future. For example, in January 2017, the U.S. House of Representatives and Senate passed legislation, which, if signed into law, would repeal certain aspects of the ACA. In addition, Congress could consider subsequent legislation to replace those elements of the ACA if so repealed.
Napo expects that the ACA, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and lower reimbursement, and additional downward pressure on the price that Napo receives for any approved product. Any reduction in reimbursement from Medicare or other government-funded programs may result in a similar reduction in payments from private payors. Moreover, recently there has been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products. The implementation of cost containment measures or other healthcare reforms may prevent Napo from being able to generate revenue, attain profitability or commercialize Napo's drugs.
Additionally, on August 2, 2011, the Budget Control Act of 2011 created measures for spending reductions by Congress. A Joint Select Committee on Deficit Reduction, tasked with recommending a targeted deficit reduction of at least $1.2 trillion for the years 2013 through 2021, was unable to reach required goals, thereby triggering the legislation's automatic reduction to several government programs. This included aggregate reductions of Medicare payments to providers of 2% per fiscal year, which went into effect on April 1, 2013 and, due to subsequent legislative amendments to the statute, will stay in effect through 2025 unless additional action is taken by Congress. On January 2, 2013, the American Taxpayer Relief Act of 2012 was signed into law, which, among other things, further reduced Medicare payments to several types of providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. More recently, there has been heightened governmental scrutiny recently over the manner in which manufacturers set prices for their marketed products, which have resulted in several recent Congressional inquiries and proposed bills designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for pharmaceutical products.
91
Napo expects that additional state and federal healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which could result in reduced demand for Napo's products once approved or additional pricing pressures.
Animal Health Business
The development, approval and sale of animal health products are governed by the laws and regulations of each country in which we intend to seek approval, where necessary, to market and subsequently sell our prescription drug and non-drug products. To comply with these regulatory requirements, we are establishing processes and resources to provide oversight of the development, approval processes and launch of its products and to position those products in order to gain market share in each respective market.
Certain U.S. federal regulatory agencies are charged with oversight and regulatory authority of animal health products in the United States. These agencies, depending on the product and its intended use may include the FDA, the USDA and the Environmental Protection Agency. In addition, the Drug Enforcement Administration regulates animal therapeutics that are classified as controlled substances. In addition, the Federal Trade Commission may in the case of non-drug products, regulate the marketing and advertising claims being made.
The approval of prescription drugs intended for animal use is regulated by the FDA's Center for Veterinary Medicine ("CVM"). The CVM consists of six offices that work together to, in part, approve new drugs for commercialization and thereafter monitor those commercialized drugs once in the market. The Office of New Animal Drug Evaluation ("ONADE"), is the lead office for reviewing novel drug candidates. We, as the sponsor of a novel drug candidate, commence the development and approval process by initiating communication with the ONADE and opening an INAD file. As part of this process, we will also schedule a discussion of the novel drug's development plan in order to obtain agreement from the CVM for the number, type and design of studies needed to obtain FDA approval of the novel drug.
As required by the FDA, new animal drug products must obtain marketing approval through the NADA process. Under the Administrative New Animal Drug Application, or Administrative NADA, process, a sponsor can engage in a phased submission of the required technical sections of an NADA, known as a rolling NADA, as opposed to submitting the entire application at once with a standard NADA. The requirements for all NADAs are the same regardless of whether a sponsor chooses the rolling NADA or the standard NADA submission. Under the phased review, once all technical sections have been submitted and reviewed, the sponsor submits an Administrative NADA to reflect that all technical sections of the NADA have been submitted and reviewed, each such technical section meets the requirements for approval and the CVM has issued technical section complete letters for each technical section. The phased review and Administrative NADA allow a drug sponsor to engage with the FDA as to each technical section to ensure that each section meets all requirements prior to submission of the application for approval. Phasing of NADA submissions is a voluntary process.
Once the tasks set forth in the development plan have been completed, including the clinical work as well as the chemistry and manufacturing work (feasibility, validation and stability of the drug inclusive), We, as the novel drug sponsor will need to provide to the FDA through the application process, information as to the safety and efficacy of the drug candidate, and, if needed, human food safety studies. These food safety studies are only required for drugs intended for use in production animals, and we currently have no plans to develop drugs for production animals. Additionally, the application will contain a module on CMC, which describes the plan for manufacturing the drug including the API, the final formulation, where it will be made, how it will be made, how the drug will be packaged, how it can be stored, the conditions required for storage and how long it can be stored
92
before expiry. A major part of the CMC section is the analysis we employ to ensure that the manufactured drug is of a high quality, is consistently manufactured under cGMP and is stable. Other significant components to the application we have to complete before receiving drug approval includes a draft label that will list specific information such as dosing information, intended use, warnings, directions for use, and other information as required by the regulations. The package insert that will contain information on studies, warnings, drug interactions, intended use and dosing is considered part of the label in addition to that which is adhering to the container itself. The CVM ensures that the labeling provides all the necessary information to use the drug safely and effectively, and that it clearly discloses the risks associated with the drug.
MUMS Designation
The Minor Use and Minor Species Animal Health Act ("MUMS Act"), became effective in August 2004. The purpose of the MUMS Act was twofold: first, to encourage the development and availability of more animal drugs that are intended to be used in a major species defined as dogs, cats, cattle, horses, chickens, turkeys and pigs to treat diseases which occur infrequently or in limited geographic areas, therefore having an impact on a smaller number of animals on a yearly basis; and second, to encourage the development and availability of animal drugs for use in minor species (defined as all animals other than humans that are not one of the major species). The drug sponsor may seek conditional approval of the drug product provided the Office of Minor Use Minor Species ("OMUMS") acknowledges that the intended use fits within a small number of animals treated per annum. A drug does not have to be designated to be eligible for conditional approval, however if OMUMS designates a MUMS drug, certain incentives and exclusivities are available to the sponsor. The MUMS designation is modeled on the orphan-drug designation for human drug development and has certain financial incentives available to encourage MUMS drug development such as the availability of grants to help with the cost of the MUMS drug development. Also, drug developers of MUMS drugs are eligible to apply for a waiver of the user fees once the MUMS designation has been given by OMUMS. We believe that we qualify for MUMS designation for Canalevia as a minor use in a major species because the estimated total number of dogs in the United States affected by CID is less than 70,000. We also believe that Canalevia will qualify for MUMS designation for EID because, in our estimate, the total number of dogs in the United States affected by EID on an annual basis is less than 70,000. To obtain conditional approval of a MUMS drug, the company must submit CMC and safety data similar to that required for an NADA, as well as data suggesting a reasonable expectation of effectiveness. After the submission and the review of the application, the FDA through the CVM can then grant a conditional approval (CA-1). This approval allows for a commercialization of the product, while the sponsor continues to collect the substantial evidence of effectiveness required for a full NADA approval. The sponsor has up to five years to demonstrate substantial evidence of effectiveness for a previously conditionally approved drug. Ideally, MUMS designation helps move the product forward in development; however, it may not shorten the time to full commercialization. A sponsor that gains approval or conditional approval for a MUMS designated drug receives seven years of marketing exclusivity.
Protocol Concurrence
As we announced in April 2016, we obtained protocol concurrence from the FDA for our pivotal trial of Canalevia that we initiated in December 2015 for acute diarrhea in dogs. Under this process, a protocol is submitted to the FDA voluntarily by a drug sponsor. The FDA review of the protocol for a pivotal study makes it more likely that the study will generate information the sponsor needs to demonstrate whether the drug is safe and effective for its intended use. It creates an expectation by the sponsor that the FDA will not later alter its perspectives on these issues unless public or animal health concerns appear that were not recognized at the time of protocol assessment. Even if FDA issues a protocol concurrence, ultimate approval of an NADA by the FDA is not guaranteed because a final
93
determination that the agreed-upon protocol satisfies a specific objective, such as the demonstration of efficacy, or supports an approval decision, will be based on a complete review of all the data submitted to the FDA. Even if we were to obtain protocol concurrence, such concurrence does not guarantee that the results of the study will support a particular finding or approval of the new drug.
Marketing Exclusivity
We are currently planning on seeking MUMS designation for some of our prescription drug products and if we receive such a designation, we will be entitled to a seven-year marketing exclusivity, which means that we will face no competition from another sponsor marketing the same drug in the same dosage form for the same intended use. If we were to lose such designation or not receive such designation but our application as a new animal drug is found to be a new chemical entity that meets the criteria described by the FDA, we would be entitled to a five-year marketing exclusivity. In order to receive this five-year exclusivity, the FDA would have to find in its approval of our application that our NADA contains an API not previously approved in another application, that the application itself is an original application, not a supplemental application, and that our application included the following studies: one or more investigations to demonstrate substantial evidence of effectiveness of the drug for which we are seeking approval; animal safety studies and human food safety studies (where applicable). If the NADA is seeking approval of a drug for which we have received conditional approval, we, upon approval would still be entitled to a five-year marketing exclusivity provided we meet the criteria as set forth above. If, however, our NADA is for a drug for which the FDA has determined that the drug contains an API that has previously been approved, regardless of whether the original approval was for use in humans or not, we may only be entitled to a three-year marketing exclusivity provided that the NADA is an original, not supplemental, application and contains both safety and efficacy studies demonstrating the safety and efficacy of the drug which is the subject of the application. We have received MUMS designation for Canalevia for the indication of chemotherapy-induced diarrhea, or CID, in dogs. Additionally, the FDA has indicated that the use of Canalevia for the treatment of EID in dogs qualifies as a "minor use", which means that Canalevia is eligible for conditional approval for the indication of EID in dogs.
As we announced on March 8, 2018, the U.S. Food & Drug Administration's Center for Veterinary Medicine (CVM) has indicated that the Company's Reasonable Expectation of Effectiveness (RxE) Technical Section is complete towards conditional approval of Canalevia (crofelemer delayed-release tablets) for treatment of CID in dogs, based on CVM's review of the results of Jaguar's completed pilot study (CANA-001) of Canalevia for this indication. If Canalevia is approved for CID and EID in dogs, we expect to conduct the commercial launch of Canalevia for these indications in 2019.
Labeling
The FDA plays a significant role in regulating the labeling, advertising and promotion of animal drugs. This is also true of regulatory agencies in the EU and other territories. In addition, advertising and promotion of animal health products is controlled by regulations in many countries. These rules generally restrict advertising and promotion to those claims and uses that have been reviewed and approved by the applicable agency. We will conduct a review of advertising and promotional material for compliance with the local and regional requirements in the markets where it eventually may sell its product candidates.
Other Regulatory Considerations
We believe regulatory rules relating to human food safety, food additives, or drug residues in food will not apply to the products we currently are developing because our animal prescription drug product candidates are not intended for use in production animals, with the exception of horses, which qualify as food animals in Europe and Canada; and our non-prescription products are not regulated by
94
section 201(g) of the Federal Food, Drug, and Cosmetic Act, which the FDA is authorized to administer.
Our animal prescription drug product candidates currently in development, if approved, may eventually face generic competition in the United States and in the EU after the period of exclusivity has expired. In the United States, a generic animal drug may be approved pursuant to an abbreviated new animal drug application ("ANADA"). With an ANADA, a generic applicant is not subject to the submission of new clinical and safety data but instead must only show that the proposed generic product is a copy of the novel drug product, and bioequivalent to the approved novel product. However, if our product candidates are the first approved by the FDA or the EMA as applicable for use in animals, they will be eligible for a five-year marketing exclusivity in the United States and 10 years in the EU thereby prohibiting generic entry into the market. If the product has MUMS designation it has a seven-year marketing exclusivity.
We do not believe that our animal non-prescription products are currently subject to regulation in the United States. The CVM only regulates those animal supplements that fall within the FDA's definition of an animal drug, food or feed additive. The Federal Food Drug and Cosmetic Act defines food as "articles used for food or drink for man or other animals and articles used as components of any such article." Animal foods are not subject to pre-market approval and are designed to provide a nutritive purpose to the animals that receive them. Feed additives are defined as those articles that are added to an animal's feed or water as illustrated by the guidance documents. Our non-prescription products are not added to food, are not ingredients in food nor are they added to any animal's drinking water. Therefore, our non-prescription products do not fall within the definition of a food or feed additive. The FDA seeks to regulate such supplements as food or food additives depending on the intended use of the product. The intended use is demonstrated by how the article is included in a food, or added to the animals' intake (i.e., through its drinking water). If the intended use of the product does not fall within the proscribed use making the product a food, it cannot be regulated as a food. There is no intent to make our non-prescription products a component of an animal food, either directly or indirectly. A feed additive is a product that is added to a feed for any reason including the top dressing of an already prepared feed. Some additives, such as certain forage, are deemed to be Generally Recognized as Safe ("GRAS"), and therefore, not subject to a feed Additive Petition approval prior to use. However, the substances deemed GRAS are generally those that are recognized as providing nutrients as a food does. We do not believe that our non-prescription products fit within this framework either. Finally, a new animal drug refers to drugs intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease in animals. Our non-prescription products are not intended to diagnose, cure, mitigate, treat or prevent disease and therefore, do not fit within the definition of an animal drug. Our non-prescription products are intended to support a healthy gut, support fluid retention, and normalize stool formation in animals suffering from scours. Additionally, because a previously marketed human formulation of the botanical extract in our non-prescription products was considered a dietary supplement subject to the Dietary Supplement Health and Education Act of 1994 (and not regulated as a drug by the FDA), we do not believe that the FDA would regulate the animal formulation used in our non-prescription products in a different manner. We do not believe that our non-prescription products fit the definition of an animal drug, food or food additive and therefore are not regulated by the FDA at this time.
In addition to the foregoing, we may be subject to state, federal and foreign healthcare and/or veterinary medicine laws, including but not limited to anti-kickback laws, as we may from time to time enter consulting and other financial arrangements with veterinarians, who may prescribe or recommend our products. If our financial relationships with veterinarians are found to be in violation of such laws that apply to us, we may be subject to penalties.
95
Legal Proceedings
From time to time, we may become involved in litigation relating to claims arising from the ordinary course of business. Other than as set forth below, there are currently no claims or actions pending against us, the ultimate disposition of which could have a material adverse effect on our results of operations, financial condition or cash flows.
On March 27, 2018, the Company received a demand letter from a law firm representing a purported stockholder, relating to certain approvals obtained at a special meeting of stockholders on March 12, 2018 (the "2018 Special Meeting"). The demand letter alleges that the Company miscalculated the votes with respect to (i) the proposal to amend the Company's Third Amended and Restated Certificate of Incorporation as filed with Secretary of State of the State of Delaware on March 15, 2018 (the "COI"), which increased the authorized shares of Common Stock from 250,000,000 to 500,000,000 (the "Share Increase Proposal") and (ii) the proposal to amend the COI to effect a reverse stock split at a ratio of not less than 1-for-1.2 and not greater than 1-for-10 (the "Former Reverse Stock Split Proposal"). The Company did not implement the Former Reverse Stock Split Proposal. In addition, at the 2018 annual meeting of stockholders held on May 18, 2018, stockholders approved amendments to the COI to (i) effect a reverse stock split at a ratio of not less than 1-for-11 and not greater than 1-for-15 and (ii) decrease the number of authorized shares of Common Stock to 150,000,000.
On September 5, 2018, the Company responded to the law firm, indicating that the Board unanimously rejected the demands set forth in the demand letter. While no proceedings with respect to the demand letter have been initiated by the purported stockholder to date, the Company believes that the allegations set forth in the demand letter are without merit and would intend to vigorously defend against any such proceeding.
On July 20, 2017, a putative class action complaint was filed in the United States District Court, Northern District of California, Civil Action No. 3:17-cv-04102, by Tony Plant (the "Plaintiff") on behalf of shareholders of the Company who held shares on June 30, 2017 and were entitled to vote at the 2017 Special Shareholders Meeting, against the Company and certain individuals who were directors as of the date of the vote (collectively, the "Defendants"), in a matter captioned Tony Plant v. Jaguar Animal Health, Inc., et al., making claims arising under Section 14(a) and Section 20(a) of the Exchange Act and Rule 14a-9, 17 C.F.R. § 240.14a-9, promulgated thereunder by the SEC. The claims allege false and misleading information provided to investors in the Joint Proxy Statement/Prospectus on Form S-4 (File No. 333-217364) declared effective by the Commission on July 6, 2017 related to the solicitation of votes from shareholders to approve the merger and certain transactions related thereto. The Company accepted service of the complaint and summons on behalf of itself and the United States-based director Defendants on November 1, 2017. The Company has not accepted service on behalf of, and Plaintiff has not yet served, the non-U.S.-based director Defendants. On October 3, 2017, Plaintiff filed a motion seeking appointment as lead plaintiff and appointment of Monteverde & Associates PC as lead counsel. That motion has been granted. Plaintiff filed an amended complaint against the Company and the United States-based director Defendants on January 10, 2018. If the Plaintiff were able to prove its allegations in this matter and to establish the damages it asserts, then an adverse ruling could have a material impact on the Company. However, the Company disputes the claims asserted in this putative class action case and is vigorously contesting the matter. The Defendants filed a motion to dismiss on March 12, 2018, for which oral arguments were held on June 14, 2018. The court has not yet ruled on the motion. The Company believes that it is not probable that an asset has been impaired or a liability has been incurred as of the date of the financial statements and the amount of any potential loss is not reasonably estimable.
96
Other than as described above, there are currently no claims or actions pending against us, the ultimate disposition of which could have a material adverse effect on our results of operations, financial condition or cash flows.
Corporate Information
We were incorporated in the State of Delaware on June 6, 2013. Our principal executive offices are located at 201 Mission Street, Suite 2375, San Francisco, CA 94015 and our telephone number is (415) 371-8300. Our website address is www.jaguar.health. The information contained on, or that can be accessed through, our website is not part of this prospectus. Our voting common stock is listed on the NASDAQ Capital Market and trades under the symbol "JAGX." On July 31, 2017, we completed the acquisition of Napo (the "Merger") pursuant to the Agreement and Plan of Merger, dated March 31, 2017, by and among the Company, Napo, Napo Acquisition Corporation, and Napo's representative (the "Merger Agreement").
Jaguar Health, our logo, Napo Pharmaceuticals, Mytesi, Canalevia, Equilevia and Neonorm are our trademarks that are used in this prospectus. This prospectus also includes trademarks, tradenames and service marks that are the property of other organizations. Solely for convenience, trademarks and tradenames referred to in this prospectus appear without the ©, ® or ™ symbols, but those references are not intended to indicate that we will not assert, to the fullest extent under applicable law, our rights or that the applicable owner will not assert its rights, to these trademarks and tradenames.
Employees
As of September 4, 2018, we had 45 employees. Six employees hold D.V.M. or Ph.D. degrees. 14 of our employees are engaged in research and development activities and 23 employees are engaged in sales and marketing. None of our employees are represented by labor unions or covered by collective bargaining agreements.
Description of Properties
Our corporate headquarters are located in San Francisco, California, where we lease 6,311 rentable square feet of office space from CA-Mission Street Limited Partnership. Our lease agreement expires on September 30, 2020. We believe that our existing facilities are adequate for our near term needs.
DIRECTORS AND CORPORATE GOVERNANCE
The following table lists our directors and their respective ages and positions as of September 5, 2018:
Name
|
Age | Position | |||
|---|---|---|---|---|---|
| James J. Bochnowski(1)(2)(3) | 75 | Chairman of the Board (Class I) | |||
| Lisa A. Conte | 59 | Chief Executive Officer, President and Director (Class I) | |||
| Jeffery C. Johnson(2)(3) | 46 | Director (Class III) | |||
| Greg J. Divis | 51 | Director (Class III) | |||
| John Micek III(1)(3) | 65 | Director (Class II) | |||
| Jiahao Qui | 32 | Director (Class II) | |||
| Jonathan B. Siegel(1)(2) | 45 | Director (Class I) | |||
| Murray David MacNaughtan | 51 | Director (Class III) | |||
- (1)
- Member
of the audit committee.
- (2)
- Member of the compensation committee.
97
- (3)
- Member of the nominating committee.
Executive Officers
Lisa A. Conte. Ms. Conte has served as our President, Chief Executive Officer and a member of our board of directors since she founded the company in June 2013. From 2001 to 2014, Ms. Conte served as the Chief Executive Officer of Napo Pharmaceuticals, Inc., a biopharmaceutical company she founded in November 2001. In 1989, Ms. Conte founded Shaman Pharmaceuticals, Inc., a natural product pharmaceutical company. Ms. Conte is also currently a member of the board of directors of Healing Forest Conservatory, a California not-for-profit public benefit corporation. Ms. Conte holds an M.S. in Physiology and Pharmacology from the University of California, San Diego, and an M.B.A. and A.B. in Biochemistry from Dartmouth College.
We believe Ms. Conte is qualified to serve on our board of directors due to her extensive knowledge of our company and experience with our product and product candidates, as well as her experience managing and raising capital for public and private companies.
Steven R. King, Ph.D. Dr. King has served as our Executive Vice President of Sustainable Supply, Ethnobotanical Research and Intellectual Property since March 2014 and as our Secretary since September 2014. From 2002 to 2014, Dr. King served as the Senior Vice President of Sustainable Supply, Ethnobotanical Research and Intellectual Property at Napo Pharmaceuticals, Inc. Prior to that, Dr. King served as the Vice President of Ethnobotany and Conservation at Shaman Pharmaceuticals, Inc. Dr. King has been recognized by the International Natural Products and Conservation Community for the creation and dissemination of research on the long-term sustainable harvest and management of Croton lechleri, the widespread source of crofelemer. Dr. King is currently a member of the board of directors of Healing Forest Conservatory, a California not-for-profit public benefit corporation. Dr. King holds a Ph.D. in Biology from the Institute of Economic Botany of the New York Botanical Garden and an M.S. in Biology from the City University of New York.
Karen S. Wright. Ms. Wright has served as our Chief Financial Officer since December 15, 2015. Prior to joining us, Ms. Wright served as head of finance for Clene Nanomedicine, Inc., beginning in August 2014. From June 2011 to May 2014, Ms. Wright served as vice president of finance and corporate controller at Veracyte, Inc., and from 2006 to 2011, she served as vice president of finance, corporate controller and principal accounting officer of VIA Pharmaceuticals, Inc. Ms. Wright holds a B.S. in Accounting and Marketing from the University of California Walter A. Haas School of Business.
Non-Employee Directors
James J. Bochnowski. Mr. Bochnowski has served as a member of our board of directors since February 2014 and as Chairman of our board since June 2014. Since 1988, Mr. Bochnowski has served as the founder and Managing Member of Delphi Ventures, a venture capital firm. In 1980, Mr. Bochnowski co-founded Technology Venture Investors. Mr. Bochnowski holds an M.B.A. from Harvard University Graduate School of Business and a B.S. in Aeronautics and Astronautics from Massachusetts Institute of Technology.
We believe Mr. Bochnowski is qualified to serve on our board of directors due to his significant experience with venture capital backed healthcare companies and experience as both an executive officer and member of the board of directors of numerous companies.
Jeffery C. Johnson. Mr. Johnson has served as a member of our board of directors since March 2018. Mr. Johnson is a partner at Sagard Holdings, ULC and is an investment manager at Sagard Capital Partners Management Corp. Mr. Johnson has been employed by entities affiliated with Sagard Holdings, ULC since 2012. From 2006 to 2011, he served as portfolio manager and senior analyst at Evercore Asset Management. Before that, Mr. Johnson was a portfolio manager and analyst
98
at Citigroup Asset Management where he was responsible for consumer and healthcare investments. He began his career at strategy consulting firm Braxton Associates. He also serves on the board of directors of Peak Achievement Athletics and previously served on the board of directors of Vein Clinics of America. Mr. Johnson received his M.B.A. in Finance and Accounting from the Kellogg School of Management in 1999. Mr. Johnson was elected to our board of directors pursuant to the terms of the Series A Preferred Stock Purchase Agreement, dated as of March 23, 2018, by and between the Company and Sagard Capital Partners, L.P., and the Certificate of Designation, which gives the Preferred Stock holders the right to elect two Class III directors so long as they are entitled to vote in the aggregate 5% or more of all of the votes entitled to be cast by holders of all voting securities of the Company at any meeting of stockholders.
We believe Mr. Johnson is qualified to serve on our board of directors due to his experience evaluating, investing in and managing companies in the health care sector for Sagard Holdings, ULC, and for other investment firms he was previously employed by.
Jiahao Qiu. Mr. Qiu has served as a member of our board of directors since February 2014. Mr. Qiu has been employed at BioVeda Management, Ltd., a life science investment firm, as associate (2010-2012), senior associate (2012-2014) and Principal since April 2014. From 2009 to 2010, he served as an interpreter for the Delegation of the European Union to China. Mr. Qiu holds a B.S. in Biotechnology from the Jiao Tong University in Shanghai, China.
We believe Mr. Qiu is qualified to serve on our board of directors due to his experience with evaluating, managing and investing in life science portfolio companies for BioVeda Management, Ltd.
Greg J. Divis. Mr. Divis has served as a member of our board of directors since June 2018. Mr. Divis currently serves as the Chief Operating Officer of Avadel Pharmaceuticals, an emerging branded specialty pharmaceutical company he joined in 2017. Prior to Avadel he served as an Executive-in-Residence and Operating Partner for Linden Capital, a healthcare-focused middle market private equity firm. Previous roles also include President and Chief Executive Officer of Lumara Health, a specialty-branded pharmaceutical company focused on women's health, where Mr. Divis led the successful turnaround and transformation of the business resulting in a series of transactions culminating in the successful sale to AMAG Pharmaceuticals. Mr. Divis has also held such notable roles as Vice President, Business Development & Lifecycle Management at Sanofi-Aventis, and Vice-President and General Manager, UK and Ireland, for Schering-Plough Corporation. He currently serves on the Board of Directors of Mobius Therapeutics and previously served on the Board of Tolero Pharmaceuticals. Mr. Divis is a graduate of the University of Iowa.
We believe Mr. Divis is qualified to serve on our board of directors due to his extensive experience in the pharmaceutical industry and experience as both an executive officer and member of the board of directors of other companies.
Murray David MacNaughtan. Mr. MacNaughtan has served as a member of our board of directors since June 2018. Most recently, Mr. MacNaughtan led the intellectual property investing strategy at CPPIB Credit Investments Inc., a wholly-owned subsidiary of the Canada Pension Plan Investment Board, for eight years, focused on the acquisition and securitization of pharmaceutical royalty streams. Prior to this, he was co-head of the royalty monetization fund at DRI Capital (formerly Drug Royalty Corp.). From 1999 to 2002, he was Vice President of Business Development at Paladin Labs, a specialty pharmaceutical company, where he led a team responsible for acquiring and licensing products for the Canadian market. He joined Paladin from Royal Bank Capital Corp., the venture capital subsidiary of RBC, where he was an investment manager. Mr. MacNaughtan began his career in the biopharmaceutical industry at Hemosol Inc. as a process development engineer. Mr. MacNaughtan earned a B.Sc. and M.Sc. in Applied Science from Queen's University in Ontario, and an M.B.A. from the University of Toronto.
99
We believe Mr. MacNaughtan is qualified to serve on our board of directors due to his extensive experience in specialty finance and the pharmaceutical industry.
John Micek III. Mr. Micek has served as a member of our board of directors since April 2016. From 2000 to 2010, Mr. Micek was managing director of Silicon Prairie Partners, LP, a Palo Alto, California based family-owned venture fund. Since 2010, Mr. Micek has been managing partner of Verdant Ventures, a merchant bank dedicated to sourcing and funding university and corporate laboratory spinouts in areas including pharmaceuticals and cleantech. Mr. Micek serves on the board of directors of Armanino Foods of Distinction, Innovare Corporation and JAL/Universal Assurors. He is also a board member and the Chief Executive Officer and Chief Financial Officer of Enovo Systems and from March 2014 to August 2015 he served as interim Chief Financial Officer for Smith Electric Vehicles, Inc. Mr. Micek is a cum laude graduate of Santa Clara University and the University of San Francisco School of Law, and is a practicing California attorney specializing in financial services.
We believe Mr. Micek is qualified to serve on our board of directors due to his many years of executive experience in management and on boards of director.
Jonathan B. Siegel. Mr. Siegel has served as a member of our board of director since March 2018. Mr. Siegel is founder of JBS Healthcare Ventures, which pursues investments in public and private healthcare entities. In 2017 he left Kingdon Capital ("Kingdon"), where he was principal of the firm, a member of the executive committee and the sector head for healthcare. He joined Kingdon in 2011 and has more than 18 years of investment experience. Prior to joining Kingdon, Mr. Siegel was with SAC Capital Advisors from 2005 to 2011, serving as a portfolio manager for healthcare starting in 2007. Before joining SAC, he was an associate director of pharmaceutical and specialty pharmaceutical research with Bear, Stearns & Co., a research associate with Dresdner Kleinwort Wasserstein, specializing in pharmaceuticals, a consultant to the Life Sciences Division of Computer Sciences Corporation, a research associate at the Novartis Center for Immunobiology, Harvard Medical School, Beth Israel Deaconess Medical Center, and a research assistant at Tufts University School of Medicine. Additionally, he previously served on the board of KV Pharmaceutical Company. Mr. Siegel received a B.S. in Psychology from Tufts University in 1995 and an M.B.A. from Columbia Business School in 1999.
We believe Mr. Siegel is qualified to serve on our board of directors due to his extensive experience in the pharmaceutical investment sector.
Family Relationships
There are no family relationships among any of our directors or executive officers.
Board Composition and Risk Oversight
Our business and affairs are managed under the direction of our board of directors, which consists of eight members. Certain members of our board of directors were elected pursuant to the provisions of a voting agreement among certain of our major stockholders, as amended. See "Certain Relationships and Related Persons Transactions—Voting Agreement" for more information regarding the voting agreement.
Six of the eight directors that comprise our board are independent within the meaning of the independent director rules of the NASDAQ Stock Market, LLC, or NASDAQ.
Our board of directors are divided into three classes of directors. At each annual meeting of stockholders, a class of directors will be elected for a three-year term to succeed the class whose term is then expiring. The terms of the directors will expire upon the election and qualification of successor
100
directors at the annual meeting of stockholders to be held during the years 2019 for the Class I directors, 2020 for the Class II directors and 2021 for the Class III directors.
The Class I directors are Ms. Conte, Mr. Bochnowski, and Mr. Siegel.
The Class II directors are Mr. Qiu and Mr. Micek.
The Class III directors are Mr. Johnson, Mr. Divis and Mr. MacNaughtan.
We expect that any additional directorships resulting from an increase in the number of directors will be distributed among the three classes so that, as nearly as possible, each class will consist of one-third of the directors. The division of our board of directors into three classes with staggered three-year terms may delay or prevent a change of our management or a change in control.
Our board of directors has an active role, as a whole and at the committee level, in overseeing the management of our risks. Our board of directors is responsible for general oversight of risks and regular review of information regarding our risks, including credit risks, liquidity risks and operational risks. Our compensation and nominating committees are responsible for overseeing the management of risks relating to our executive compensation plans and arrangements and the risks associated with the independence of our board of directors and potential conflicts of interest. Our audit committee is responsible for overseeing the management of our risks relating to accounting matters and financial reporting. While each committee is responsible for evaluating certain risks and overseeing the management of such risks, our entire board of directors is regularly informed through discussions from committee members about such risks. Our board of directors believes its administration of risk oversight function has not affected our board of directors' leadership structure.
Director Independence
Our common stock is listed on The Nasdaq Capital Market. Under Nasdaq rules, independent directors must comprise a majority of a listed company's board of directors. In addition, Nasdaq rules require that, subject to specified exceptions, each member of a listed company's Audit, Compensation and Nominating Committee must be independent. Audit Committee members must also satisfy the independence criteria set forth in Rule 10A-3 under the Exchange Act. Under Nasdaq rules, a director will only qualify as an "independent director" if, in the opinion of the company's board of directors, such person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director.
To be considered independent for purposes of Rule 10A-3, a member of an audit committee of a listed company may not, other than in his or her capacity as a member of the audit committee, our board of directors, or any other board committee (1) accept, directly or indirectly, any consulting, advisory, or other compensatory fee from the listed company or any of its subsidiaries or (2) be an affiliated person of the listed company or any of its subsidiaries.
Our board of directors periodically undertakes a review of its composition, the composition of its committees and the independence of our directors and considered whether any director has a material relationship with us that could compromise his or her ability to exercise independent judgment in carrying out his or her responsibilities. Based upon information requested from and provided by each director concerning his or her background, employment and affiliations, including family relationships, our board of directors has determined that six of our eight directors (i.e., Mr. Bochnowski, Mr. Siegel, Mr. Micek, Mr. Qiu, Mr. Divis and Mr. MacNaughtan) do not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director and that each of these directors is "independent" as that term is defined under the Nasdaq rules. Our board of directors also determined that Mr. Micek (chairperson), Mr. Siegel and Mr. Bochnowski, who comprise our Audit Committee, Mr. Bochnowski (chairperson), Mr. Siegel and Mr. Johnson, who comprise our Compensation Committee, and Mr. Johnson (chairperson), Mr. Bochnowski and Mr. Micek, who
101
comprise our Nominating Committee, satisfy the independence standards for those committees established by applicable SEC rules and the Nasdaq rules and listing standards.
In making this determination, our board of directors considered the relationships that each non-employee director has with us and all other facts and circumstances our board of directors deemed relevant in determining independence, including the beneficial ownership of our capital stock by each non-employee director.
Board Committees
Audit Committee
The members of our Audit Committee are Mr. Micek, Mr. Siegel and Mr. Bochnowski. Mr. Micek is the chairperson of the Audit Committee. Our Audit Committee's responsibilities include:
- •
- appointing, approving the compensation of, and assessing the independence of our registered public accounting firm;
- •
- overseeing the work of our independent registered public accounting firm, including through the receipt and consideration of reports from that
firm;
- •
- reviewing and discussing with management and our independent registered public accounting firm our annual and quarterly financial statements
and related disclosures;
- •
- monitoring our internal control over financial reporting, disclosure controls and procedures and code of conduct;
- •
- discussing our risk management policies;
- •
- establishing policies regarding hiring employees from our independent registered public accounting firm and procedures for the receipt and
retention of accounting related complaints and concerns;
- •
- reviewing and approving or ratifying any related person transactions; and
- •
- preparing the Audit Committee report required by SEC rules.
All audit and non-audit services, other than de minimis non-audit services, to be provided to us by our independent registered public accounting firm must be approved in advance by our Audit Committee.
Our board of directors has determined that each of Mr. Micek, Mr. Siegel and Mr. Bochnowski is an independent director under Nasdaq rules and under Rule 10A-3. All members of our Audit Committee meet the requirements for financial literacy under the applicable rules and regulations of the SEC and Nasdaq. Our board of directors has determined that Mr. Micek is an "audit committee financial expert," as defined by applicable SEC rules, and has the requisite financial sophistication as defined under the applicable Nasdaq rules and regulations.
The Audit Committee held five meetings in 2017 and three meetings in 2018 as of August 31, 2018. The Audit Committee has adopted a written charter approved by the board of directors, which is available on our website at https://jaguarhealth.gcs-web.com/corporate-governance.
102
Compensation Committee
The members of our Compensation Committee are Mr. Bochnowski, Mr. Siegel and Mr. Johnson. Mr. Bochnowski is the chairperson of the Compensation Committee. Our Compensation Committee's responsibilities include:
- •
- determining, or making recommendations to our board of directors with respect to, the compensation of our Chief Executive Officer;
- •
- determining, or making recommendations to our board of directors with respect to, the compensation of our other executive officers;
- •
- overseeing and administering our cash and equity incentive plans;
- •
- reviewing and making recommendations to our board of directors with respect to director compensation;
- •
- reviewing and discussing at least annually with management our "Compensation Discussion and Analysis" disclosure if and to the extent then
required by SEC rules; and
- •
- preparing the Compensation Committee report and necessary disclosure in our annual proxy statement in accordance with applicable SEC rules.
Our board has determined that each of Mr. Bochnowski, Mr. Siegel and Mr. Johnson is independent under the applicable Nasdaq rules and regulations, is a "non-employee director" as defined in Rule 16b-3 promulgated under the Exchange Act, and is an "outside director" as that term is defined in Section 162(m) of the Internal Revenue Code of 1986, as amended.
The Compensation Committee held three meetings in 2017 and one meeting in 2018 as of August 31, 2018. All compensation-related matters were approved at the board of directors' level. The Compensation Committee has adopted a written charter approved by the board of directors, which is available on our website at https://jaguarhealth.gcs-web.com/corporate-governance.
Nominating Committee
The members of our Nominating Committee are Mr. Johnson, Mr. Bochnowski and Mr. Micek. Mr. Johnson is the chairperson of the Nominating Committee. Our Nominating Committee's responsibilities include:
- •
- identifying individuals qualified to become members of our board of directors;
- •
- evaluating qualifications of directors;
- •
- recommending to our board of directors the persons to be nominated for election as directors and to each of the committees of our board of
directors; and
- •
- overseeing an annual evaluation of our board of directors.
The Nominating Committee held one meeting in 2017 and one meeting in 2018 as of August 31, 2018. All nomination-related matters were approved at the board of directors level. The Nominating Committee has adopted a written charter approved by the board of directors, which is available on our website at https://jaguarhealth.gcs-web.com/corporate-governance.
Code of Ethics and Conduct
We have adopted a Code of Business Conduct and Ethics that applies to our directors, officers and employees, including our President and Chief Executive Officer, our Chief Financial Officer and other employees who perform financial or accounting functions. The Code of Business Conduct and Ethics sets forth the basic principles that guide the business conduct of our employees. A current copy of the
103
code is available on our website at https://jaguarhealth.gcs-web.com/corporate-governance. We intend to disclose future amendments to certain provisions of our code of business conduct and ethics, or waivers of such provisions on our website to the extent required by applicable rules and exchange requirements. The inclusion of our website address in this prospectus does not incorporate by reference the information on or accessible through our website into this prospectus.
Compensation Committee Interlocks and Insider Participation
None of the members of our Compensation Committee has ever been an officer or employee of our company. None of our executive officers currently serves, or in the past year has served, as a member of the board of directors or Compensation Committee or other board committee performing equivalent functions of any entity that has one or more of its executive officers serving on our board of directors or Compensation Committee.
Limitation of Liability and Indemnification
Our third amended and restated certificate of incorporation, as amended, and amended and restated bylaws contain provisions that limit the personal liability of our directors for monetary damages to the fullest extent permitted by Delaware law. Delaware law provides that directors of a corporation will not be personally liable to us or our stockholders for monetary damages for any breach of fiduciary duties as directors, except liability for:
- •
- any breach of the director's duty of loyalty to us or our stockholders;
- •
- any act or omission not in good faith or that involves intentional misconduct or a knowing violation of law;
- •
- unlawful payments of dividends or unlawful stock repurchases or redemptions as provided in Section 174 of the DGCL; or
- •
- any transaction from which the director derived an improper personal benefit.
Such limitation of liability does not apply to liabilities arising under federal securities laws and does not affect the availability of equitable remedies, such as injunctive relief or rescission.
Our third amended and restated certificate of incorporation provides that we indemnify our directors to the fullest extent permitted by Delaware law. In addition, our amended and restated bylaws provide that we indemnify our directors and officers to the fullest extent permitted by Delaware law. Our amended and restated bylaws also provide that we shall advance expenses incurred by a director or officer in advance of the final disposition of any action or proceeding, and permit us to secure insurance on behalf of any officer, director, employee or other agent for any liability arising out of his or her actions in that capacity, regardless of whether we would otherwise be permitted to indemnify him or her under the provisions of Delaware law. We have entered and expect to continue to enter into agreements to indemnify our directors, executive officers and other employees as determined by our board of directors. With certain exceptions, these agreements provide for indemnification for related expenses including, among others, attorneys' fees, judgments, fines and settlement amounts incurred by any of these individuals in any action or proceeding. We believe that these bylaw provisions and indemnification agreements are necessary to attract and retain qualified persons as directors and officers. We also maintain directors' and officers' liability insurance.
The limitation of liability and indemnification provisions in our third amended and restated certificate of incorporation and amended and restated bylaws and our indemnification agreements, may discourage stockholders from bringing a lawsuit against our directors for breach of their fiduciary duty of care. They may also reduce the likelihood of derivative litigation against our directors and officers, even though an action, if successful, might benefit us and other stockholders. Furthermore, a
104
stockholder's investment may be adversely affected to the extent that we pay the costs of settlement and damage awards against directors and officers. There is no pending litigation or proceeding involving any of our directors, officers or employees for which indemnification is sought, and we are not aware of any threatened litigation that may result in claims for indemnification.
Board Leadership Structure
Our Bylaws and corporate governance guidelines provide our board of directors with flexibility in its discretion to combine or separate the positions of Chairperson of the board of directors and chief executive officer. As a general policy, our board of directors believes that separation of the positions of Chairperson and chief executive officer reinforces the independence of the board of directors from management, creates an environment that encourages objective oversight of management's performance and enhances the effectiveness of the board of directors as a whole. We expect and intend the positions of Chairperson of the board and chief executive officer to be held by two individuals in the future.
105
CERTAIN RELATIONSHIPS AND RELATED PERSON TRANSACTIONS
The following includes a summary of transactions since January 1, 2018, to which we have been a party in which the amount involved exceeded or will exceed the lesser of (i) $120,000 and (ii) one percent (1%) of the average of our total assets at year-end for the prior two fiscal years, and in which any of our directors, executive officers or beneficial owners of more than 5% of our capital stock or any member of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest. Compensation arrangements for our directors and executive officers are described in our annual proxy statement on Schedule 14A.
Transactions with Sagard
Preferred Stock Offering
Preferred Stock Purchase Agreement
On March 23, 2018, we entered into the Preferred Stock Purchase Agreement with Sagard, pursuant to which we, in a private placement, agreed to issue and sell to Sagard 5,524,926 shares of Preferred Stock (the "Preferred Shares"), for an aggregate purchase price of $9,199,001. The Preferred Stock Purchase Agreement also provides for customary representations, warranties and covenants among the parties. Among other things, the Preferred Stock Purchase Agreement requires that we (i) file prior to the initial closing the Certificate of Designation and (ii) enter into a registration rights agreement with Sagard providing for the registration of shares of our Common Stock, issuable upon conversion of the Preferred Shares (the "Conversion Shares"). In addition, so long as Sagard or its affiliates own, in the aggregate, no less than 50% or more of the cumulative amount of the Preferred Shares and Conversion Shares issued in the Preferred Stock Offering, Sagard and its affiliates have the right to purchase (x) 100% of the first $10 million of any new securities issued by us and thereafter (y) a pro rata portion of any new securities that we may issue from time to time, subject to certain exceptions specified in the Preferred Stock Purchase Agreement. The Preferred Shares are subject to a 12-month lock-up period, which period may be shortened in limited circumstances specified in the Preferred Stock Purchase Agreement.
The Preferred Stock Purchase Agreement also provides that Sagard has the right to designate at least one non-voting observer (subject to increase to two if at any time two designees of the Preferred Shares and the Conversion Shares are not represented on the board of directors) to attend meetings of the Board, the board of directors of any of our subsidiaries and each committee of any of the foregoing (a "Board Observer"). In addition, at such time as no shares of Preferred Stock are outstanding, and so long as Sagard holds (i) at least 35% of the total number of the Conversion Shares that have been issued upon conversion of all shares of Preferred Stock issued in the Preferred Stock Offering, Sagard shall be entitled thereunder to nominate two directors of the Company (each, a "Series A Director") and (ii) less than 35% but at least 20% of the total number of the Conversion Shares that have been issued upon conversion of all shares of Preferred Stock issued in the Preferred Stock Offering, Sagard shall be entitled thereunder to nominate one director of the Company.
Notwithstanding the foregoing, the number of Series A Directors shall be reduced to the extent necessary to comply with our obligations, if any, under the rules or regulations of the Nasdaq Stock Market (including Nasdaq Listing Rule 5640). The Preferred Stock Purchase Agreement provides that, if one Series A Director may not be appointed due to compliance with Nasdaq Listing Rule 5640, then Sagard shall be entitled to designate one additional Board Observer to attend meetings of the board of directors, the board of directors of any of our subsidiaries and each committee of any of the foregoing as an observer.
106
Terms of Series A Preferred Stock
The Certificate of Designation authorizes 5,524,926 shares of Preferred Stock and provides for the rights, preferences and privileges of such Preferred Stock. Any reference to share prices in the below description of the Preferred Stock, including but not limited to the conversion price for the Preferred Shares and the amount of the liquidation preference per share, is subject to adjustment in the event of any stock dividend, stock split, reverse stock split, combination or other similar recapitalization, as further described in the Certificate of Designation.
Dividends
Holders of shares of Preferred Stock are entitled to participate equally and ratably with the holders of shares of Common Stock in all dividends paid and distributions made to the holders of Common Stock on the shares of Common Stock on an as converted basis.
Election of Directors and Voting Rights
The holders of a majority of the outstanding shares of Preferred Stock are entitled to elect two (2) members of the Company's Board of Directors. Notwithstanding the foregoing, the number of Series A Directors shall be reduced to the extent necessary to comply with the Company's obligations, if any, under the rules or regulations of the Nasdaq Stock Market (including Nasdaq Listing Rule 5640).
The holders of shares of Preferred Stock have the right to vote with holders of shares of the Common Stock, voting together as one class on all other matters, with each share of Preferred Stock entitling the holder thereof to cast that number of votes per share as is equal to the aggregate number of shares of Common Stock into which it is then convertible, using the market value of the Common Stock on the date of the Preferred Stock Agreement (i.e., $0.1935) as the conversion price; provided that, at any time prior to the time the Company obtains stockholder approval, as required pursuant to Nasdaq Rule 5635(b), no holder (together with such holder's attribution parties) is permitted to have a number of votes in excess of such aggregate number of votes granted to the holders of 19.99% of the shares of Common Stock then outstanding (including any votes with respect to any shares of Common Stock and Preferred Stock beneficially owned by the holder or such holder's attribution parties).
Voluntary Conversion
Each share of Preferred Stock is initially convertible into nine shares of Common Stock at an effective conversion price of $0.185 per share (based on an original price per Preferred Share of $1.665), provided that, at any time prior to the time the Company obtains stockholder approval, as required pursuant to Nasdaq Rule 5635(b) any conversion of Preferred Stock by a holder into shares of the Common Stock would be prohibited if, as a result of such conversion, the holder, together with such holder's attribution parties, would beneficially own more than 19.99% of the total number of shares of the Common Stock issued and outstanding after giving effect to such conversion. Subject to certain limited exceptions, the shares of Preferred Stock cannot be offered, pledged or sold by Sagard for one year from the date of issuance. The conversion price is subject to certain adjustments in the event of any stock dividend, stock split, reverse stock split, combination or other similar recapitalization.
Mandatory Conversion
The shares of Preferred Stock will be mandatorily converted upon the date and time, or the occurrence of an event, specified by vote or written consent of the holders of a majority of the then outstanding shares of Preferred Stock at a conversion price of $0.185 per share. In each case, the number of shares of Common Stock issuable upon such conversion will be limited to the extent
107
necessary to satisfy limitations on beneficial ownership as described under "Voluntary Conversion" above.
Optional Redemption
At any time after the first anniversary of the issuance of the Preferred Shares, so long as certain call conditions specified in the Certificate of Designation have been satisfied, the Company shall have the right to offer to redeem shares of Preferred Stock at a share price equal to two times the original share issue price of the Preferred Shares. The Company is only permitted to exercise this right to redeem two times, the first of which must be for an aggregate redemption price of $9,199,001 and the second of which must be for all remaining shares of Preferred Stock remaining. If a holder of Preferred Shares fails to accept the Company's redemption offer, such holder's shares of Preferred Stock shall be automatically converted into shares of Common Stock pursuant to the terms of "Mandatory Conversion" as described above.
Mandatory Redemption
If (i) the Company's consolidated net revenues attributable to the Mytesi products ("Mytesi Revenues") for the six-month period ended March 31, 2021 are less than $22 million, (ii) the average volume-weighted average price of the Common Stock for the thirty days immediately prior to the Measurement Date (as defined below) is less than $1.00 or (iii) the Company fails to file with the SEC on or before June 30, 2021 its quarterly report on Form 10-Q for the three months ended March 31, 2021, then the holders of at least a majority of shares of Preferred Stock then outstanding may require the Company to redeem all shares of Preferred Stock then outstanding at a per share purchase price equal to $2.3057. For purposes of the foregoing sentence, "Measurement Date" means the later of (x) April 30, 2021 and (y) the date on which the Company files its quarterly report on Form 10-Q for the three months ended March 31, 2021 (but in no event later than June 30, 2021).
The mandatory redemption right described above shall terminate if, prior to the Measurement Date, both (i) the Mytesi Revenues for any six-month period ending at the end of a calendar quarter are equal to or exceed $22 million and (ii) the average volume-weighted average price of the Common Stock for the thirty days immediately preceding the end of such calendar quarter is equal to or greater than $1.00.
Fundamental Change
The Certificate of Designation provides the holders of Preferred Stock with a right to require the Company to repurchase shares of Preferred Stock at a price to be calculated pursuant to the terms of the Certificate of Designation upon the occurrence of any of the following events (each a "Fundamental Change"):
(X) (a) any person or "group" (within the meaning of Rules 13d-3 and 13d-5 under the Exchange Act), other than certain "Permitted Holders" (and/or any direct transferee of shares of Preferred Stock from any Permitted Holder) (as defined in the Certificate of Designation), (i) shall have acquired beneficial ownership of more than fifty percent (50%) or more on a fully diluted basis of the voting and/or economic interest in the capital stock of the Company (or surviving entity in a merger or consolidation, if applicable)) or (ii) shall have obtained the power (whether or not exercised) to elect a majority of the members of the Company's Board of Directors (or similar governing body); or (b) the occurrence of any "change of control" or similar event under any agreements relating to any indebtedness of the Company or its subsidiaries; or
(Y) except in the case of a Deemed Liquidation Event (as defined in the Certificate of Designation and described below) in which holders of Preferred Stock receive, concurrently with the consummation of such Deemed Liquidation Event, a cash payment pursuant to Sections 2.1 and 2.4 of
108
the Certificate of Designation in full, in an amount equal to the "Fundamental Change Price" as defined in the Certificate of Designation that would otherwise be payable, the Company or any of its subsidiaries enters into any transaction of merger or consolidation (except that a person may be merged with or into the Company or another wholly-owned subsidiary thereof so long as the Company or another wholly-owned subsidiary is the continuing or surviving person), or conveys, sells, leases, subleases (as lessor or sublessor), exchanges, transfers or otherwise disposes of, in one transaction or a series of transactions, all or substantially all of the consolidated business, assets or property of the Company and its subsidiaries.
The "Fundamental Change Price" for each share of Preferred Stock, as of any date, shall be calculated as the sum of (i) the amount payable in respect of such share under Section 2.1 of the Certificate of Designation in the event of a "Liquidation Event" as of such date, plus (ii) any and all accrued and unpaid dividends upon the Preferred Stock, whether or not declared, as of the date of the Fundamental Change, plus (iii) the "Participation Amount" as defined in the Certificate of Designation.
Merger or Liquidation
Subject to the Fundamental Change provision described above, under the terms of the Certificate of Designation, upon merger or consolidation resulting in a change of control, sale, lease, transfer, exclusive license or other disposition, in a single transaction or series of related transactions, by the Company or any subsidiary of the Company, of substantially all of the assets of the Company and its subsidiaries taken as a whole, or the sale or disposition (whether by merger, consolidation or otherwise) of Napo (as defined below) (or any successor in interest) or one or more other subsidiaries of the Company if substantially all of the assets of the Company and its subsidiaries taken as a whole are held by such subsidiary or subsidiaries (collectively, a "Deemed Liquidation"), liquidation, dissolution or winding up of the Company as determined under the Certificate (collectively, a "Liquidation Event"), each share of Preferred Stock will be entitled to a preference of $1.665 per share (or the equivalent of $0.185 per share on an as-converted to Common Stock basis) plus a participation right described below. Thereafter, the holders of Common Stock then outstanding shall be entitled to receive an amount per share of Common Stock (in stock or cash as determined under the Certificate of Designation) equal to $0.185 (as adjusted for stock splits, reverse splits, stock dividends, reclassifications, recapitalizations and/or other similar events). Thereafter, all of the remaining assets of the Company and/or proceeds from a Deemed Liquidation or Liquidation Event, as applicable, will in general be divided pro rata among the holders of the shares of Preferred Stock and the shares of Common Stock, on an as converted basis (all as more fully specified and calculated under the Certificate of Designation).
Covenants
Pursuant to the terms of the Preferred Stock as provided in the Certificate of Designation, so long as any shares of Preferred Stock are outstanding, the Company may not agree, among other things, without the prior written consent or vote of the holders of at least a majority of the then outstanding shares of Preferred Stock, to: (a) amend, alter, repeal or waive any provision of the Certificate of Designation, (b) amend, alter or repeal any provision of the Company's charter documents in a manner that would adversely affect the powers, privileges, preferences or rights of the Preferred Stock, (c) create additional classes or series of capital stock having rights, preferences or privileges senior to or pari passu with the Preferred Stock or (d) increase or decrease the authorized number of shares of Preferred Stock.
So long as Sagard or its affiliates own at least 35% of the shares of Preferred Stock that were originally issued, the Certificate of Designation provides that the Company may not, among other things, without the prior written consent or vote of the holders of at least a majority of the then outstanding shares of Preferred Stock, (a) authorize or issue any capital stock of any subsidiary which is
109
not wholly-owned by the Company, (b) declare or pay dividends on the Company's equity securities or redeem any of the Company's equity securities, (c) incur, guarantee or assume any indebtedness, subject to certain limited exceptions, (d) grant or incur any lien, subject to certain limited exceptions, (e) enter into any transaction for the acquisition of all or substantially of the equity interests or assets of another person, subject to certain limited exceptions (f) make any investments, subject to certain limited exceptions or (g) enter into any transactions with the Company's affiliates, subject to certain limited exceptions.
Services Agreement
On March 23, 2018, the Company entered into a management services agreement with Sagard Capital Partners Management Corp. ("SCPM"), an affiliate of Sagard, pursuant to which SCPM will provide certain consulting and management advisory services to the Company over a three-year period (the "Initial Term") for an annual fee of $450,000, which fees will be paid in equal installments over the Initial Term beginning in the second year of the Initial Term (the "Services Agreement"). The Services Agreement may be terminated earlier than the initial three-year term (i) upon mutual consent of the parties, (ii) by either party following a breach of the Services Agreement by the other party that remains uncured following 30 days' written notice thereof, (iii) in SCPM's sole discretion with 10 day's prior written notice, or (iv) upon the consummation of a Deemed Liquidation (so long as all accrued and unpaid fees payable thereunder as of such termination have been paid in full) or a Fundamental Change in which all of the Company's shares of Preferred Stock are repurchased by the Company.
As described above, Jeffery C. Johnson, a member of the Company's board of directors, is an investment manager at SCPM.
Transactions with Lisa A. Conte
Lisa A. Conte has served as our President, Chief Executive Officer and a member of our board of directors since she founded the company in June 2013. On July 18, 2018, Ms. Conte purchased 1,500 shares of common stock for an aggregate purchase price of $1,668.60.
Transactions with Jonathan B. Siegel
On March 29, 2018, our board of directors appointed Mr. Jonathan B. Siegel to fill the vacancy created by Dr. Azhir's resignation and serve as Class I director of the Company until the 2019 annual meeting of stockholders or until his successor is elected and qualified.
Mr. Siegel was formerly a principal and member of the executive committee of Kingdon Capital ("Kingdon") and the head for Kingdon's healthcare sector. As described further above, on March 31, 2017, Napo entered into the Kingdon NPA with the Kingdon Purchasers, which are affiliates of Kingdon, under which remains outstanding approximately $10 million in aggregate principal amount of the Kingdon Notes. Napo's obligations under the Kingdon Notes are secured by a security interest in substantially all of Napo's assets, including Napo intellectual property.
On July 16, 17, and 18, 2018, JBS Healthcare Ventures LLC purchased an aggregate of 15,000 shares of common stock in the open market for an aggregate purchase price of $18,653. Mr. Siegel is the sole member of JBS Healthcare Ventures LLC.
Transactions with Charles C. Conte
Charles C. Conte is the brother of Lisa A. Conte, who Conte has served as our President, Chief Executive Officer and a member of our board of directors since she founded the company in June 2013.
110
On September 11, 2018, Charles C. Conte purchased a convertible promissory note in the aggregate principal amount of $111,250 with an exercise price of $0.85 per share of common stock and a warrant exercisable for 33,918 shares of common stock with an exercise price of $1.23 per share.
Financings
In January 2018, the Company issued 50,000 shares of common stock to an existing investor in exchange for $6,425 in services rendered.
In the first quarter of 2018, the Company issued 12,314,291 shares of its common stock in exchange for redemption of certain convertible debt.
On February 26, 2018, the Company entered into a securities purchase agreement with CVP, pursuant to which the Company issued to CVP a promissory note in the aggregate principal amount of $2,240,909 for an aggregate purchase price of $1,560,000. The Note carries an original issue discount of $655,909, and the initial principal balance also includes $25,000 to cover CVP's transaction expenses. The Note bears interest at the rate of 8% per annum and matures on (i) August 26, 2019 if the Company has raised at least $12 million in equity after the issuance date of the Note (the "Redemption Start Condition") and on or before April 1, 2018 or (ii) November 26, 2018 if the Redemption Start Condition is not satisfied on or before April 1, 2018.
On March 21, 2018, the Company entered into a securities purchase agreement with CVP, pursuant to which the Company issued to CVP a promissory note in the aggregate principal amount of $1,090,341 for an aggregate purchase price of $750,000. The Note carries an original issue discount of $315,341, and the initial principal balance also includes $25,000 to cover CVP's transaction expenses. The Note bears interest at the rate of 8% per annum and matures on September 21, 2019.
In March of 2018, the Company issued 4,285,423 shares of its common stock in exchange for payment of interest expense on certain long-term convertible debt.
In March 2018, the Company entered into a stock purchase agreement with Sagard Capital Partners, L.P. pursuant to which the Company, in a private placement, agreed to issue and sell to Sagard 5,524,926 shares of the Company's series A convertible participating preferred stock, $0.0001 par value per share, for an aggregate purchase price of $9,199,001. Each share of preferred stock is initially convertible into nine shares of common stock at an effective conversion price of $0.185 per share (based on an original price per Preferred Share of $1.665), provided that, at any time prior to the time the Company obtains stockholder approval, as required pursuant to Nasdaq Rule 5635(b) any conversion of Preferred Stock by a holder into shares of the Common Stock would be prohibited if, as a result of such conversion, the holder, together with such holder's attribution parties, would beneficially own more than 19.99% of the total number of shares of the Common Stock issued and outstanding after giving effect to such conversion. Subject to certain limited exceptions, the shares of Preferred Stock cannot be offered, pledged or sold by Sagard for one year from the date of issuance. The conversion price is subject to certain adjustments in the event of any stock dividend, stock split, reverse stock split, combination or other similar recapitalization. Concurrently with the consummation of the preferred stock offering, the Company entered into share purchase agreements with certain institutional investors pursuant to which the Company issued 29,411,766 shares of the Company's common stock in exchange for $5.0 million in cash.
Pursuant to the November 24, 2017 share purchase agreement with an investor, on July 12, 2018 the Company received $624,897 in exchange for 470,781 shares of its voting common stock.
Effective August 13, 2018, the Company entered into an acknowledgement agreement with CVP extending the maturity date of the $2,155,000 secured convertible promissory note dated July 29, 2017 from August 2, 2018 to August 26, 2019 and also extending the maturity date of the $1,587,500 secured promissory note dated December 8, 2017 from September 8, 2018 to August 26, 2019.
111
On September 11, 2018, Charles C. Conte purchased a convertible promissory note in the aggregate principal amount of $111,250 with an exercise price of $0.85 per share of common stock and a warrant exercisable for 33,918 shares of common stock with an exercise price of $1.23 per share.
Indemnification Agreements
We have entered into indemnification agreements with each of our directors and officers. These agreements, among other things, require us or will require us to indemnify each director to the fullest extent permitted by Delaware law, including indemnification of expenses such as expenses, judgments, penalties, fines and amounts paid in settlement to the extent legally permitted incurred by the director or officer in any action or proceeding, including any action or proceeding by or in right of us, arising out of the person's services as a director or officer.
112
The following table sets forth certain information with respect to the beneficial ownership of our common stock as of August 31, 2018 by:
- •
- each person, or group of affiliated persons, who is known by us to be the beneficial owner of more than 5% of our outstanding common stock;
- •
- each of our directors;
- •
- each of our executive officers; and
- •
- all of our executive officers and directors as a group.
Beneficial ownership is determined in accordance with the rules and regulations of the SEC and includes voting or investment power with respect to our common stock. Shares of our common stock subject to options, warrants or RSUs that are currently exercisable or vested, or exercisable or subject to vesting within 60 days of the date of this prospectus are considered outstanding and beneficially owned by the person holding the options, warrants, or RSUs for the purpose of calculating the percentage ownership of that person but not for the purpose of calculating the percentage ownership of any other person. Except as otherwise noted, the persons and entities in this table have sole voting and investing power with respect to all of the shares of our common stock beneficially owned by them, subject to community property laws, where applicable. The information is not necessarily indicative of beneficial ownership for any other purpose, including for purposes of Section 13(d) and Section 13(g) of the Securities Act.
The column titled "Percentage of Shares Beneficially Owned—Before Offering" is based on a total of 9,528,103 shares of common stock outstanding as of August 31, 2018. The total shares of common stock outstanding may be adjusted for the purpose of calculating the percentage ownership of a person that has options, warrants or RSUs that are currently exercisable or vested, or exercisable or subject to vesting within 60 days of the date of this prospectus but not for the purpose of recalculating the percentage ownership of any other person. The column titled "Percentage of Shares Beneficially Owned—After Offering" is based on shares of common stock to be outstanding after this offering, and includes the shares of common stock that we are selling in this offering.
113
Except as otherwise set forth below, the address of each beneficial owner listed in the table below is c/o Jaguar Health, Inc., 201 Mission Street, Suite 2375, San Francisco, California 94105.
| |
|
Percentage of Shares Beneficially Owned |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
Name and Address of Beneficial Owner
|
Number of Shares Beneficially Owned |
Before Offering(16) |
After Offering |
|||||||
5% Stockholders |
||||||||||
Sagard Capital Partners, L.P.(1) |
3,314,956 | 20.52 | % | |||||||
Invesco Ltd.(2) |
26,885,838 | 18.81 | % | |||||||
Nantucket Investments Limited(3) |
1,575,621 | 12.90 | % | |||||||
Entities affiliated with Kingdon Capital Management L.L.C.(4) |
1,364,932 | 14.33 | % | |||||||
Executive Officers and Directors |
||||||||||
Lisa A. Conte(5) |
223,392 | 2.29 | % | |||||||
Steven R. King, Ph.D.(6) |
78,485 | —* | ||||||||
Karen S. Wright(7) |
50,245 | —* | ||||||||
James J. Bochnowski(8) |
150,658 | 1.57 | % | |||||||
Jeffery C. Johnson(9) |
17,476 | —* | ||||||||
John Micek III(10) |
41,237 | —* | ||||||||
Jiahao Qiu(11) |
5,812 | —* | ||||||||
Jonathan B. Siegel(12) |
32,208 | —* | ||||||||
Greg Divis(13) |
8,738 | —* | ||||||||
Murray David MacNaughtan(14) |
8,738 | —* | ||||||||
All executive officers and directors as a group (10 persons)(15) |
616,989 | 6.70 | % | |||||||
- *
- Less
than 1%.
- (1)
- Represents
as if converted 5,524,926 shares of Series A Convertible Participating Preferred Stock.
- (2)
- Represents
13G filed on April 10, 2018 pre 15-1 reverse split; if converted 1,792,396 shares post-split and as calculated, of common stock owned by
Invesco Ltd.
- (3)
- Represents
325,615 common shares plus 1,250,006 non-voting common shares owned by Nantucket Investment Ltd.
- (4)
- Represents
625,324 shares of common stock, 18,887 warrants that are exercisable within 60 days of August 31, 2018 and 720,721 convertible note
conversion shares, owned by Kingdon Capital Management, L.L.C.
- (5)
- Represents
753 shares of common stock and 221,139 shares of stock issuable under stock options that are exercisable or will become exercisable within 60 days
of August 31, 2018 and shares of restricted stock that have vested or will vest within 60 days of August 31, 2018.
- (6)
- Represents
442 shares of common stock and 78,043 shares of stock issuable under stock options that are exercisable or will become exercisable within 60 days
of August 31, 2018 and shares of restricted stock that have vested or will vest within 60 days of August 31, 2018.
- (7)
- Represents
50,245 shares of stock issuable under stock options that are exercisable or will become exercisable within 60 days of August 31, 2018 and
shares of restricted stock that have vested or will vest within 60 days of August 31, 2018.
- (8)
- Includes (i) 72,503 shares of common stock, (ii) 71,563 shares of common stock issuable under stock options that are exercisable or will become exercisable within 60 days of August 31, 2018 and (iii) 6,592 shares of common stock issuable under warrants that are exercisable or will become exercisable within 60 days of August 31, 2018. All securities other than stock options are held by
114
the Bochnowski Family Trust. Mr. Bochnowski is a co-trustee and beneficiary of such trust and shares voting and investment control over such shares with his spouse.
- (9)
- Represents
17,476 shares of stock issuable under stock options that are exercisable or will become exercisable within 60 days of August 31, 2018 and
shares of restricted stock that have vested or will vest within 60 days of August 31, 2018. Mr. Johnson holds the Sagard board seat and is part of the Sagard executive management
team.
- (10)
- Represents
41,247 shares of stock issuable under stock options that are exercisable or will become exercisable within 60 days of August 31, 2018 and
shares of restricted stock that have vested or will vest within 60 days of August 31, 2018.
- (11)
- Represents
5,812 shares of stock issuable under stock options that are exercisable or will become exercisable within 60 days of August 31, 2018 and
shares of restricted stock that have vested or will vest within 60 days of August 31, 2018.
- (12)
- Represents
17,476 shares of stock issuable under stock options that are exercisable or will become exercisable within 60 days of August 31, 2018 and
shares of restricted stock that have vested or will vest within 60 days of August 31, 2018.
- (13)
- Represents
8,738 shares of stock issuable under stock options that are exercisable or will become exercisable within 60 days of August 31, 2018 and
shares of restricted stock that have vested or will vest within 60 days of August 31, 2018.
- (14)
- Represents
8,738 shares of stock issuable under stock options that are exercisable or will become exercisable within 60 days of August 31, 2018 and
shares of restricted stock that have vested or will vest within 60 days of August 31, 2018.
- (15)
- See
notes (5) - (10)
- (16)
- Excludes the 670,586 shares of common stock, issuable upon exercise of a warrant issued on August 28, 2018 to Pacific Capital Management, LLC with an exercise price that is the lower of (i) $0.85 per share and (ii) the average of the closing sales price of the common stock for the 30 consecutive trading days commencing on September 4, 2018; the 185,417 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to L2 Capital, LLC with an exercise price of $0.90 per share; 33,918 shares of common stock issuable upon exercise of warrants issued on September 11, 2018 to Charles C. Conte with an exercise price of $1.23 per share; up to 706,765 shares of common stock issuable upon conversion of a convertible promissory note delivered to L2 Capital, LLC on September 11, 2018 in the aggregate principal amount of $445,000 with an exercise price of $0.85 per share; and up to 176,691 shares of common stock issuable upon conversion of a convertible promissory note delivered to Charles C. Conte on September 11, 2018 in the aggregate principal amount of $111,250 with an exercise price of $0.85 per share.
115
MATERIAL U.S. FEDERAL INCOME TAX CONSEQUENCES TO NON-U.S. HOLDERS OF COMMON STOCK AND/OR PRE-FUNDED WARRANTS
The following is a general discussion of the material U.S. federal income tax consequences applicable to a non-U.S. holder (as defined below) with respect to the acquisition, ownership and disposition of our common stock and/or pre-funded warrants. This discussion is limited to non-U.S. holders who purchase our common stock and/or pre-funded warrants issued pursuant to this offering for cash and who hold our common stock and/or pre-funded warrants as a "capital asset" within the meaning of Section 1221 of the Internal Revenue Code of 1986, as amended, or the Code (generally, property held for investment). This discussion is based upon the applicable provisions of the Code, applicable U.S. Treasury regulations promulgated thereunder, or the Treasury Regulations, and administrative and judicial interpretations thereof, promulgated thereunder, all as in effect on the date hereof, and all of which are subject to change, possibly on a retroactive basis. Any such changes could alter the tax consequences to non-U.S. holders described herein. This discussion is not a complete analysis of all of the potential U.S. federal income tax consequences applicable to a non-U.S. holder, and does not address all of the U.S. federal income tax consequences that may be relevant to a particular non-U.S. holder in light of such non-U.S. holder's particular circumstances or the U.S. federal income tax consequences applicable to non-U.S. holders that are subject to special rules, such as United States expatriates, banks, financial institutions, insurance companies, regulated investment companies, real estate investment trusts, controlled foreign corporations, passive foreign investment companies, corporations that accumulate earnings to avoid U.S. federal income tax, brokers, dealers or traders in securities, commodities or currencies, partnerships or other pass-through entities (or investors in such entities), tax-exempt organizations, tax-qualified retirement plans, persons subject to the alternative minimum tax, and non-U.S. holders that hold our common stock and/or pre-funded warrants as part of a straddle, hedge, conversion transaction or other integrated investment. In addition, this discussion does not describe any state or local income, estate or other tax consequences of holding and disposing of our common stock and/or pre-funded warrants.
As used in this discussion, the term "non-U.S. holder" means any beneficial owner of our common stock and/or pre-funded warrants that is, for U.S. federal income tax purposes, neither a partnership nor any of the following:
- •
- an individual citizen or resident of the United States;
- •
- a corporation or other entity taxable as a corporation created or organized under the laws of the United States or any political subdivision
thereof;
- •
- an estate, the income of which is subject to U.S. federal income tax regardless of its source; or
- •
- a trust if (i) a United States court is able to exercise primary supervision over the administration of the trust and one or more United States persons have authority to control all substantial decisions of the trust or (ii) the trust has a valid election in effect under applicable Treasury Regulations to be treated as a United States person.
If any entity classified as a partnership for U.S. federal income tax purposes holds our common stock and/or pre-funded warrants, the tax treatment of a partner in such partnership generally will depend on the status of the partner and the activities of the partnership. Partnerships and their partners should consult their tax advisors as to the tax consequences to them of the acquisition, ownership and disposition of our common stock and/or pre-funded warrants.
THE FOLLOWING DISCUSSION IS FOR GENERAL INFORMATION ONLY AND IS NOT TAX ADVICE. PROSPECTIVE INVESTORS ARE URGED TO CONSULT THEIR TAX ADVISORS REGARDING THE PARTICULAR U.S. FEDERAL, STATE, LOCAL AND FOREIGN TAX CONSEQUENCES TO THEM OF THE ACQUISITION, OWNERSHIP AND DISPOSITION OF OUR COMMON STOCK AND/OR PRE-FUNDED WARRANTS.
116
Distributions on Common Stock
Distributions on our common stock generally will constitute dividends for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. If a distribution exceeds our current and accumulated earnings and profits, the excess will be treated first as a tax-free return of a capital to the extent of the non-U.S. holder's adjusted tax basis in the common stock below zero, and thereafter as capital gain, subject to the tax treatment described under "—Sale, Exchange or Other Disposition of Our Common Stock," below.
Subject to the discussions below regarding backup withholding and FATCA, the gross amount of dividends paid to a non-U.S. holder of our common stock that are not effectively connected with a U.S. trade or business conducted by such non-U.S. holder generally will be subject to U.S. federal withholding tax at a rate of 30%, or such lower rate specified by an applicable income tax treaty if we have received proper certification as to the application of such treaty. If a non-U.S. holder holds our common stock in connection with the conduct of a trade or business within the United States, and dividends paid on our common stock are effectively connected with such non-U.S. holder's U.S. trade or business (and, if under an applicable income tax treaty, such dividends are attributable to a permanent establishment or fixed base maintained by the non-U.S. holder within the United States), such non-U.S. holder generally will be subject to U.S. federal income tax at ordinary U.S. federal income tax rates (on a net income basis), and such dividends will not be subject to the U.S. federal withholding tax described above. In the case of a non-U.S. holder that is a corporation, such non-U.S. holder may also be subject to a 30% "branch profits tax" unless such corporate non-U.S. holder qualifies for a lower rate under an applicable income tax treaty.
In general, to claim the benefit of any applicable income tax treaty or an exemption from U.S. federal withholding because the income is effectively connected with the conduct of a trade or business within the United States, a non-U.S. holder must provide a properly executed Internal Revenue Service, or IRS, Form W-8BEN-E for treaty benefits or IRS Form W-8ECI for effectively connected income (or such successor form as the IRS designates), before the distributions are made. These forms must be updated periodically. If you are a non-U.S. holder, you may obtain a refund of any excess amounts withheld by timely filing an appropriate claim for refund with the IRS. Non-U.S. holders should consult their tax advisers regarding their entitlement to benefits under an applicable income tax treaty and the specific manner of claiming the benefits of such treaty.
Sale, Exchange or Other Disposition of Common Stock and/or Pre-Funded Warrants
Although it is not entirely free from doubt, a pre-funded warrant should be treated as a share of our common stock for U.S. federal income tax purposes and a holder of pre-funded warrants should generally be taxed in the same manner as a holder of common stock as described below. Accordingly, upon exercise, the holding period of a pre-funded warrant should carry over to the share of common stock received. Similarly, the tax basis of the pre-funded warrant should carry over to the share of common stock received upon exercise increased by the exercise price of $0.01. The balance of this discussion generally assumes that the characterization described above is respected for U.S. federal income tax purposes.
Subject to the discussions below regarding backup withholding and FATCA, a non-U.S. holder generally will not be subject to U.S. federal income tax on any gain realized upon the sale, exchange or other disposition (collectively, a "disposition") of our common stock and/or pre-funded warrants, unless:
- •
- the gain is effectively connected with the non-U.S. holder's conduct of a trade or business within the United States, and if an income tax treaty applies, is attributable to a permanent establishment maintained by the non-U.S. holder within the United States;
117
- •
- the non-U.S. holder is an individual who is present in the United States for 183 days or more during the taxable year of
the disposition and certain other requirements are met; or
- •
- we are or have been a U.S. real property holding corporation, or USRPHC, for U.S. federal income tax purposes at any time within the shorter of (i) the five-year period ending on the date of the disposition of our common stock and/or pre-funded warrants or (ii) the non-U.S. holder's holding period for our common stock and/or pre-funded warrants.
If the gain is described in the first bullet point above, the non-U.S. holder generally will be subject to U.S. federal income tax on a net income basis with respect to such gain in the same manner as if such non-U.S. holder were a United States person. In addition, if the non-U.S. holder is a corporation for U.S. federal income tax purposes, such gain may be subject to a 30% branch profits tax unless such corporate non-U.S. holder qualifies for a lower rate under an applicable income tax treaty.
A non-U.S. holder described in the second bullet point above generally will be subject to U.S. federal income tax with respect to such gain at a flat 30% rate (or such lower rate specified by an applicable income tax treaty), which may be offset by U.S. source capital losses of the non-U.S. holder during the taxable year of disposition (even though the individual is not considered a resident of the United States), provided that the non-U.S. holder has timely filed U.S. federal income tax returns with respect to such losses.
With respect to the third bullet point above, we believe that we are not currently, and we do not anticipate becoming, a USRPHC. However, because the determination of whether we are a USRPHC depends on the fair market value of our U.S. real property interests relative to the fair market value of our other business assets and our non-U.S. real property interests, there can be no assurance that we will not become a USRPHC in the future. In general, a corporation is a USRPHC if the fair market value of its "United States real property interests" (as defined in the Code) equals or exceeds 50% of the sum of the fair market value of its worldwide real property interests and its other assets used or held for use in a trade or business. Even if we are or become a USRPHC, a non-U.S. holder would not be subject to U.S. federal income tax on a sale, exchange or other taxable disposition of shares of our common stock and/or pre-funded warrants by reason of our status as a USRPHC so long as (i) shares of our common stock and/or pre-funded warrants continue to be regularly traded on an established securities market (within the meaning of Section 897(c)(3) of the Code) during the calendar year in which such disposition occurs and (ii) such non-U.S. holder does not own and is not deemed to own (directly, indirectly or constructively) more than 5% of the shares of our common stock at any time during the shorter of the five-year period ending on the date of the disposition of our common stock and/or pre-funded warrants or the non-U.S. holder's holding period for our common stock and/or pre-funded warrants. If gain on the disposition of our common stock and/or pre-funded warrants were subject to taxation under the third bullet point above, the non-U.S. holder generally would be subject to U.S. federal income tax with respect to such gain in the same manner as gain that is effectively connected with the conduct of a U.S. trade or business (as described above), except that the branch profits tax generally would not apply.
Information Reporting and Backup Withholding
In general, a non-U.S. holder will be required to comply with certain certification procedures to establish that such holder is not a United States person in order to avoid backup withholding with respect to dividends or the proceeds from disposition of common stock and/or pre-funded warrants. In addition, we are required to report annually to the IRS the amount of any dividends paid to a non-U.S. holder, regardless of whether we actually withheld any tax. Copies of the information returns reporting such dividends and the amount withheld may also be made available to the tax authorities in the country in which the non-U.S. holder resides under the provisions of an applicable income tax treaty.
118
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules may be allowed as a refund or a credit against a non-U.S. holder's U.S. federal income tax liability, provided the required information is timely furnished to the IRS.
Foreign Accounts Tax Compliance Act
Under the Foreign Account Tax Compliance Act, as modified by Treasury Regulations and subject to any official interpretations thereof, any applicable intergovernmental agreement between the United States and a non-U.S. government to implement these rules and improve international tax compliance, or any fiscal or regulatory legislation or rules adopted pursuant to any such agreement (collectively, "FATCA"), after June 30, 2014, withholding at a rate of 30% will be required on dividends in respect of, and, after December 31, 2016, gross proceeds from the disposition of, our common stock and/or pre-funded warrants held by or through certain foreign financial institutions (including investment funds), unless such institution enters into an agreement with the Secretary of the Treasury to report, on an annual basis, information with respect to interests in, and accounts maintained by, the institution to the extent such interests or accounts are held by certain United States persons and by certain non-U.S. entities that are wholly or partially owned by United States persons and to withhold on certain payments. An intergovernmental agreement between the United States and an applicable foreign country, or future Treasury Regulations or other guidance, may modify these requirements. Accordingly, the entity through which our common stock and/or pre-funded warrants is held will affect the determination of whether such withholding is required. Similarly, dividends in respect of, and gross proceeds from the sale of, our common stock and/or pre-funded warrants held by an investor that is a non-financial non-U.S. entity that does not qualify under certain exemptions will be subject to withholding at a rate of 30%, unless such entity either (i) certifies to us that such entity does not have any "substantial United States owners" or (ii) provides certain information regarding the entity's "substantial United States owners," which we will provide to Secretary of the Treasury. We will not pay any additional amounts to holders in respect of any amounts withheld. Prospective investors are urged to consult their tax advisors regarding the possible implications of FATCA on their investment in our common stock and/or pre-funded warrants.
119
We have entered into an underwriting agreement with H.C. Wainwright & Co., LLC (the "underwriter"), dated , 2018, as the sole book-running manager of this offering. Our common stock trades on the NASDAQ Capital Market under the symbol "JAGX."
Pursuant to the terms and subject to the conditions contained in the underwriting agreement, we have agreed to sell to the underwriter named below, and the underwriter has agreed to purchase from us, the respective number of shares of common stock and pre-funded warrants set forth opposite its name below:
Underwriter
|
Number of Shares of Common Stock |
Number of Pre-funded Warrants |
|||||
|---|---|---|---|---|---|---|---|
H.C. Wainwright & Co., LLC |
|||||||
The underwriting agreement provides that the obligation of the underwriter to purchase the shares of common stock and/or pre-funded warrants offered by this prospectus is subject to certain conditions. The underwriter is obligated to purchase all of the shares of common stock and/or pre-funded warrants if any of the securities are purchased, other than those shares covered by the option to purchase additional securities described below.
Option to Purchase Additional Securities
We have granted the underwriter an over-allotment option. This option, which is exercisable for up to 30 days after the date of this prospectus, permits the underwriter to purchase up to shares of common stock at the public offering price per share of common stock from us to cover over-allotments, if any, less the underwriting discount and commissions. The underwriter may exercise its option at any time, and from time to time, within 30 days from the date of this prospectus.
Discounts, Commissions and Expenses
The underwriter proposes to offer the shares of common stock and/or pre-funded warrants purchased pursuant to the underwriting agreement to the public at the public offering price set forth on the cover page of this prospectus and to certain dealers at that price less a concession not in excess of $ per share or $ per pre-funded warrant. After this offering, the public offering price and concession may be changed by the underwriter. No such change shall change the amount of proceeds to be received by us as set forth on the cover page of this prospectus.
In connection with the sale of the common stock and/or pre-funded warrants to be purchased by the underwriter, the underwriter will be deemed to have received compensation in the form of underwriting commissions and discounts. The underwriter's commissions and discounts will be 8% of the gross proceeds of this offering, or $ per share of common stock or per pre-funded warrant.
The following table shows the underwriting discounts and commissions payable to the underwriter by us in connection with this offering:
| |
Per Share | Per Pre-Funded Warrant |
Total Per Share |
Total Per Pre-Funded Warrant |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Public offering price |
$ | $ | $ | $ | |||||||||
Underwriting discounts and commissions payable by us |
$ | $ | $ | $ | |||||||||
We have also agreed to pay the underwriter a management fee equal to 1.0% of the gross proceeds raised in this offering, to reimburse the underwriter for reasonable out-of-pocket expenses,
120
including legal fees and expenses, of up to $100,000 and to provide the underwriter with a $35,000 non-accountable expense allowance. We estimate the total expenses payable by us for this offering will be approximately $ , which amount excludes the underwriting discount and commissions.
Indemnification
Pursuant to the underwriting agreement, we have agreed to indemnify the underwriter against certain liabilities, including liabilities under the Securities Act, or to contribute to payments that the underwriter or such other indemnified parties may be required to make in respect of those liabilities.
Determination of Offering Price
The actual offering price of the securities we are offering will be negotiated between us and the underwriter based on the trading of our shares of common stock prior to the offering, among other things, and may be at a discount to the current market price.
Lock-Up Agreements
We have agreed not to (i) offer, pledge, issue, sell, contract to sell, purchase, contract to purchase, lend or otherwise transfer or dispose of, directly or indirectly, any shares of our common stock or any securities convertible into or exercisable or exchangeable for our common stock; (ii) enter into any swap or other arrangement that transfers, in whole or in part, any of the economic consequences of ownership of shares of common stock; or (iii) file any registration statement with the SEC relating to the offering of any shares of our common stock or any securities convertible into or exercisable or exchangeable for shares of our common stock, without the prior written consent of H.C. Wainwright & Co., LLC for a period of 90 days following the date of this prospectus. This consent may be given at any time without public notice. These restrictions on future issuances are subject to exceptions, including for (i) the issuance of shares of our common stock, warrants and shares of our common stock underlying the pre-funded warrants sold in this offering, (ii) the issuance of shares of our common stock upon the exercise of outstanding options or warrants or pursuant to certain other rights, (iii) the issuance of shares of our common stock or options to acquire shares of our common stock pursuant to our existing equity incentive plans and (iv) the filing of one or more registration statements on Form S-8 with respect to shares of our common stock underlying our equity incentive plans from time to time.
In addition, subject to certain limited circumstances, each of our directors and executive officers, and certain of our principal stockholders, will enter into a lock-up agreement with the underwriter. Under the lock-up agreements, the directors, executive officers and applicable stockholders may not, directly or indirectly, sell, offer to sell, contract to sell, or grant any option for the sale (including any short sale), grant any security interest in, pledge, hypothecate, hedge, establish an open "put equivalent position" (within the meaning of Rule 16a-1(h) under the Securities Exchange Act of 1934, as amended, or the Exchange Act), or otherwise dispose of, or enter into any transaction which is designed to or could be expected to result in the disposition of, any shares of our common stock or securities convertible into or exchangeable for shares of our common stock, or publicly announce any intention to do any of the foregoing, without the prior written consent of H.C. Wainwright & Co., LLC, for a period of 90 days from the date of this prospectus. This consent may be given at any time without public notice.
Underwriter Warrants
We have agreed to issue to the underwriter warrants to purchase shares of our common stock which represent 8% of the aggregate number of shares of common stock and pre-funded warrants sold in this offering. The underwriter warrants will have a term of five years from the effective date of this
121
prospectus and an exercise price per share equal to $ per share, which represents 125% of the public offering price of the shares sold in this offering. Pursuant to FINRA Rule 5110(g), the underwriter warrants and any shares issued upon exercise of the underwriter warrants shall not be sold, transferred, assigned, pledged, or hypothecated, or be the subject of any hedging, short sale, derivative, put or call transaction that would result in the effective economic disposition of the securities by any person for a period of 180 days immediately following the date of effectiveness or commencement of sales of this offering, except the transfer of any security: (i) by operation of law or by reason of our reorganization; (ii) to any FINRA member firm participating in the offering and the officers or partners thereof, if all securities so transferred remain subject to the lock-up restriction set forth above for the remainder of the time period; (iii) if the aggregate amount of our securities held by the underwriter or related persons do not exceed 1% of the securities being offered; (iv) that is beneficially owned on a pro rata basis by all equity owners of an investment fund, provided that no participating member manages or otherwise directs investments by the fund and the participating members in the aggregate do not own more than 10% of the equity in the fund; or (v) the exercise or conversion of any security, if all securities remain subject to the lock-up restriction set forth above for the remainder of the time period.
Electronic Distribution
This prospectus may be made available in electronic format on websites or through other online services maintained by the underwriter or by its affiliates. In those cases, prospective investors may view offering terms online and prospective investors may be allowed to place orders online. Other than this prospectus in electronic format, the information on the underwriter's websites or our website and any information contained in any other websites maintained by the underwriter or by us is not part of this prospectus or the registration statement of which this prospectus forms a part, has not been approved and/or endorsed by us or the underwriter in its capacity as underwriter, and should not be relied upon by investors.
Price Stabilization, Short Positions and Penalty Bids
The underwriter may engage in syndicate covering transactions, stabilizing transactions and penalty bids or purchases for the purpose of pegging, fixing or maintaining the price of our shares of common stock:
- •
- Syndicate covering transactions involve purchases of securities in the open market after the distribution has been completed in order to cover
syndicate short positions. Such a naked short position would be closed out by buying securities in the open market. A naked short position is more likely to be created if the underwriter is concerned
that there could be downward pressure on the price of the securities in the open market after pricing that could adversely affect investors who purchase in the offering.
- •
- Stabilizing transactions permit bids to purchase the underlying security so long as the stabilizing bids do not exceed a specific maximum.
- •
- Penalty bids permit the underwriter to reclaim a selling concession from a syndicate member when the securities originally sold by the syndicate member are purchased in a stabilizing or syndicate covering transaction to cover syndicate short positions.
These syndicate covering transactions, stabilizing transactions and penalty bids may have the effect of raising or maintaining the market prices of our securities or preventing or retarding a decline in the market prices of our securities. As a result, the price of our shares of common stock may be higher than the price that might otherwise exist in the open market. Neither we nor the underwriter make any representation or prediction as to the effect that the transactions described above may have on the price of our shares of common stock. These transactions may be effected on the NYSE American, in
122
the over-the-counter market or on any other trading market and, if commenced, may be discontinued at any time.
In connection with this offering, the underwriter also may engage in passive market making transactions in our shares of common stock in accordance with Regulation M during a period before the commencement of offers or sales of our shares of common stock in this offering and extending through the completion of the distribution. In general, a passive market maker must display its bid at a price not in excess of the highest independent bid for that security. However, if all independent bids are lowered below the passive market maker's bid that bid must then be lowered when specific purchase limits are exceeded. Passive market making may stabilize the market price of the securities at a level above that which might otherwise prevail in the open market and, if commenced, may be discontinued at any time.
Neither we nor the underwriter make any representation or prediction as to the direction or magnitude of any effect that the transactions described above may have on the prices of our securities. In addition, neither we nor the underwriter make any representation that the underwriter will engage in these transactions or that any transactions, once commenced, will not be discontinued without notice.
Nasdaq Capital Market Listing
Our common stock is currently traded on The Nasdaq Capital Market under the symbol "JAGX." On September 10, 2018, the last reported sale price of our common stock was $0.66 per share.
Other Relationships
From time to time, certain of the underwriter and its affiliates have provided, and may provide in the future, various advisory, investment and commercial banking and other services to us in the ordinary course of business, for which they have received and may continue to receive customary fees and commissions.
The validity of the securities being offered by this prospectus will be passed upon for us by Reed Smith LLP, San Francisco, California. Ellenoff Grossman & Schole LLP, New York, New York is representing the underwriter in connection with this offering.
The consolidated financial statements for the years ended December 31, 2017 and 2016 included in this Prospectus and the Registration Statement, have been so included in reliance on the report of BDO USA, LLP, an independent registered public accounting firm (the report on the consolidated financial statements contains an explanatory paragraph regarding the Company's ability to continue as a going concern), appearing elsewhere herein and in the Registration Statement, given on the authority of said firm as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We are subject to the reporting requirements of the Securities Exchange Act of 1934, as amended, and file annual, quarterly and current reports, proxy statements and other information with the SEC. You may read and copy these reports, proxy statements and other information at the SEC's public reference facilities at 100 F Street, N.E., Room 1580, Washington, D.C. 20549. You can request copies of these documents by writing to the SEC and paying a fee for the copying cost. Please call the SEC at 1-800-SEC-0330 for more information about the operation of the public reference facilities. SEC filings are also available at the SEC's web site at http://www.sec.gov.
123
This prospectus is only part of a registration statement on Form S-1 that we have filed with the SEC under the Securities Act and therefore omits certain information contained in the registration statement. We have also filed exhibits and schedules with the registration statement that are excluded from this prospectus, and you should refer to the applicable exhibit or schedule for a complete description of any statement referring to any contract or other document. You may inspect a copy of the registration statement, including the exhibits and schedules, without charge, at the public reference room or obtain a copy from the SEC upon payment of the fees prescribed by the SEC.
We also maintain a website at www.jaguaranimalhealth.com. You may access our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act with the SEC free of charge at our website as soon as reasonably practicable after such material is electronically filed with, or furnished to, the SEC. The information contained in, or that can be accessed through, our website is not incorporated by reference into this prospectus.
INCORPORATION OF INFORMATION BY REFERENCE
The SEC allows us to "incorporate by reference" information that we file with them. Incorporation by reference allows us to disclose important information to you by referring you to those other documents. The information incorporated by reference is an important part of this prospectus, and information that we file later with the SEC will automatically update and supersede this information. This prospectus omits certain information contained in the registration statement, as permitted by the SEC. You should refer to the registration statement and any prospectus supplement filed hereafter, including the exhibits, for further information about us and the securities we may offer pursuant to this prospectus. Statements in this prospectus regarding the provisions of certain documents filed with, or incorporated by reference in, the registration statement are not necessarily complete and each statement is qualified in all respects by that reference. Copies of all or any part of the registration statement, including the documents incorporated by reference or the exhibits, may be obtained upon payment of the prescribed rates at the offices of the SEC listed above in "Where You Can Find More Information." The documents we are incorporating by reference are:
- •
- our Annual Report on Form 10-K for the fiscal year ended December 31, 2017 filed on April 9, 2018;
- •
- our definitive proxy statement and definitive additional materials, on Schedule 14A, relating to our Annual Meeting of Stockholders held
on May 18, 2018, filed on April 24, 2018;
- •
- our Quarterly Reports on Form 10-Q for the fiscal quarter ended March 31, 2018 filed on May 15, 2018, and for the fiscal
quarter ended June 30, 2018 filed on August 13, 2018;
- •
- our Current Reports on Form 8-K filed on January 2, 2018, February 16, 2018, February 28, 2018, March 2,
2018, March 12, 2018, March 27, 2018, April 2, 2018, April 18, 2018, May 18, 2018, May 29, 2018, June 1, 2018, June 11, 2018, June 18,
2018, June 20, 2018, July 5, 2018, July 12, 2018, September 4, 2018 and September 12, 2018;
- •
- the description of our common stock contained in our registration statement on Form 8-A filed on October 30, 2014 (Registration
No. 001-36714) with the SEC, including any amendment or report filed for the purpose of updating such description; and
- •
- all reports and other documents subsequently filed by us pursuant to Sections 13(a), 13(c), 14 and 15(d) of the Exchange Act after the date of this prospectus and prior to the termination or completion of the offering of securities under this prospectus shall be deemed to be incorporated by reference in this prospectus and to be a part hereof from the date of filing such reports and other documents.
124
Unless otherwise noted, the SEC file number for each of the documents listed above is 001-36714.
In addition, all reports and other documents filed by us pursuant to the Exchange Act after the date of the initial registration statement and prior to effectiveness of the registration statement shall be deemed to be incorporated by reference into this prospectus.
Any statement contained in this prospectus or in a document incorporated or deemed to be incorporated by reference into this prospectus will be deemed to be modified or superseded for purposes of this prospectus to the extent that a statement contained in this prospectus or any other subsequently filed document that is deemed to be incorporated by reference into this prospectus modifies or supersedes the statement. Any statement so modified or superseded will not be deemed, except as so modified or superseded, to constitute a part of this prospectus.
You may request, orally or in writing, a copy of any or all of the documents incorporated herein by reference. These documents will be provided to you at no cost, by contacting: Investor Relations, Jaguar Health, Inc., 201 Mission Street, Suite 2375, San Francisco, CA, 94105 or call (415) 371-8300.
125
Shares of Common Stock
Pre-Funded Warrants to Purchase Common Stock

PROSPECTUS
Sole Book-Running Manager
H.C. Wainwright & Co.
, 2018
Part II—INFORMATION NOT REQUIRED IN PROSPECTUS
ITEM 13. OTHER EXPENSES OF ISSUANCE AND DISTRIBUTION.
The following table sets forth an itemized statement of the expenses (excluding underwriting discounts) that are payable by us in connection with the registration, offer and sale of the common stock described in this registration statement. With the exception of the SEC registration fee, the FINRA filing fee and The NASDAQ Capital Market listing fee, the amounts set forth below are estimates.
| |
Amount to be Paid |
|||
|---|---|---|---|---|
SEC registration fee |
$ | 1,575 | ||
FINRA filing fee |
$ | 2,398 | ||
NASDAQ Capital Market listing fee |
$ | |||
Blue-sky qualification fees and expenses |
* | |||
Legal fees and expenses |
* | |||
Accounting fees and expenses |
* | |||
Transfer agent and registrar fees and expenses |
* | |||
Miscellaneous expenses |
* | |||
| | | | | |
Total |
$ | |||
| | | | | |
| | | | | |
| | | | | |
- *
- To be filed by amendment
ITEM 14. INDEMNIFICATION OF DIRECTORS AND OFFICERS.
Section 102(b)(7) of the DGCL authorizes a corporation in its certificate of incorporation to eliminate or limit personal liability of directors of the corporation for violations of the directors' fiduciary duty of care. However, directors remain liable for breaches of duties of loyalty, failing to act in good faith, engaging in intentional misconduct, knowingly violating a law, paying a dividend or approving a stock repurchase which was illegal under DGCL Section 174 or obtaining an improper personal benefit. In addition, equitable remedies for breach of fiduciary duty of care, such as injunction or recession, are available.
Our current certificate of incorporation eliminates the personal liability of the members of our board of directors to the fullest extent permitted by the DGCL. Any repeal or modification of that provision by the stockholders of the corporation will not adversely affect any right or protection of a director of the corporation existing at the time of such repeal or modification.
Section 145 of the DGCL provides that a corporation has the power to indemnify a director, officer, employee or agent of the corporation, or a person serving at the request of the corporation for another corporation, partnership, joint venture, trust or other enterprise in related capacities against expenses (including attorneys' fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by the person in connection with an action, suit or proceeding to which he was or is a party or is threatened to be made a party to any threatened, ending or completed action, suit or proceeding by reason of such position, if such person acted in good faith and in a manner he reasonably believed to be in or not opposed to the best interests of the corporation, and, in any criminal action or proceeding, had no reasonable cause to believe his conduct was unlawful, except that, in the case of actions brought by or in the right of the corporation, no indemnification shall be made with respect to any claim, issue or matter as to which such person shall have been adjudged to be liable to the corporation unless and only to the extent that the Court of Chancery or other adjudicating court determines that, despite the adjudication of liability but in view of all of the circumstances of the
II-1
case, such person is fairly and reasonably entitled to indemnity for such expenses which the Court of Chancery or such other court shall deem proper.
Our current bylaws provide for indemnification of our officers and directors to the fullest extent permitted by the DGCL.
We have entered into indemnification agreements with each of our directors, and intend to enter into such agreements with each of our officers prior to this offering, pursuant to which we agreed, to the maximum extent permitted by applicable law and subject to the specified terms and conditions set forth in each agreement, to indemnify a director or officer who acts on our behalf and is made or threatened to be made a party to any action or proceeding against expenses, judgments, fines and amounts paid in settlement that are incurred by such officer or director in connection with the action or proceeding. The indemnification provisions apply whether the action was instituted by a third party or by us.
We have purchased and maintain insurance on behalf of our officers and directors that provides coverage for expenses and liabilities incurred by them in their capacities as officers and directors.
In any underwriting agreement we enter into in connection with the sale of securities being registered hereby, the underwriter will agree to indemnify, under certain conditions, us, our directors, our officers and persons who control us within the meaning of the Securities Act against certain liabilities.
ITEM 15. RECENT SALES OF UNREGISTERED SECURITIES.
Common Stock Purchase Agreement
Shares of our common stock were sold pursuant to the common stock purchase agreement with Aspire Capital Fund, LLC, as disclosed on our Form 8-K filed with the SEC on June 9, 2016.
Common Stock Purchase Agreement
In October 2016, pursuant to a common stock purchase agreement dated October 18, 2016, we issued 170,455 shares of common stock to an accredited investor for gross proceeds of $150,000.
Amendment to Convertible Note
On November 8, 2016, we entered into an amendment to extend the maturity date of the $150,000 convertible note, issued pursuant to the convertible note purchase agreement dated December 23, 2014, from October 31, 2016 to January 1, 2017. In exchange for the extension of the maturity date, on November 8, 2016, we issued the convertible noteholder a warrant to purchase 120,000 shares of common stock at an exercise price of $0.01 per share, which expires July 28, 2022. On January 31, 2017, we entered into another amendment to further extend the maturity date of the $150,000 convertible note to January 1, 2018. In exchange for the extension, we issued the convertible note holder a warrant to purchase 370,916 shares of our common stock at an exercise price of $0.51 per share, which expires on January 31, 2019.
2016 Securities Purchase Agreement
On November 22, 2016, we entered into a Securities Purchase Agreement with certain institutional investors, pursuant to which we sold securities to such investors in a private placement transaction (the "2016 Private Placement"). In the 2016 Private Placement, we sold an aggregate of 1,666,668 shares of our common stock at a price of $0.60 per share for gross proceeds of approximately $1.0 million, as disclosed on our Form 8-K filed with the SEC on November 29, 2016.
II-2
Invesco Asset Management Limited
On February 21, 2017, Invesco Asset Management Limited ("Invesco") delivered a signed commitment letter to us, pursuant to which Invesco agreed, subject to the terms and conditions of such letter, to purchase, simultaneously with the consummation of the merger, $3.0 million of our common stock at a price equal to $0.925 per share, as disclosed on our Form 10-Q filed with the SEC on May 15, 2017.
Settlement of Outstanding Napo Debt
Pursuant to the Agreement and Plan of Merger, dated March 31, 2017 (the "Merger Agreement"), by and among the Company, Napo, Napo Acquisition Corporation ("Merger Sub"), and Napo's representative (the "Merger"), Napo entered into a Settlement and Discounted Payoff Agreement with Nantucket Investments Limited ("Nantucket") and the lenders named therein (the "Settlement Agreement") pursuant to which Napo agreed, simultaneously with the consummation of the merger, (a) to make a cash payment to Nantucket of no less than $8 million, which will reduce the outstanding principal obligations under the Litigation Financing Agreement, dated October 10, 2014, by and between Napo and Nantucket (the "Financing Agreement"), and (b) in satisfaction as a compromise for the outstanding obligations under the Financing Agreement and the release of any lien or security interest in respect of such outstanding obligations, (x) to transfer to Nantucket 2,666,666 shares of Jaguar common stock owned by Napo and (y) pursuant to the merger agreement, to cause Jaguar to issue to Nantucket 1,940,382 newly issued shares of Jaguar voting common stock and 38,380,028 newly issued shares of Jaguar non-voting common stock, as disclosed on our Form 8-K filed with the SEC on March 30, 2017.
Sales Pursuant to Share Purchase Agreement
On June 28, 2017, pursuant to a share purchase agreement, we issued 100,000 shares of our common stock to an existing investor for gross proceeds of $50,000, as disclosed on our Form 10-Q filed with the SEC on August 9, 2017.
Completion of Acquisition or Disposition of Assets
On July 31, 2017, the Company completed its merger with Napo pursuant to the Merger Agreement, as disclosed on our Form 8-K filed with the SEC on August 1, 2018. In connection with the Merger, (i) each issued and outstanding share of Napo common stock (other than dissenting shares and shares held by the Company or Napo) was converted into a contingent right to receive (x) up to a whole number of shares of Company common stock comprising in the aggregate up to approximately 20.2% of the fully diluted shares of Company common stock immediately following the consummation of the merger, which contingent right will vest only if the resale of certain shares of Company common stock (the "Tranche A Shares") issued by the Company to Nantucket pursuant to the Napo debt settlement provides Nantucket with specified cash returns over a specified period of time (the "Hurdle Amounts"), and (y) if the applicable Hurdle Amount is achieved before all of the Tranche A Shares are sold, additional shares of Company common stock (equal to 50% of the unsold Tranche A Shares), which will be distributed pro rata among holders of contingent rights and holders of Napo restricted stock units, (ii) existing creditors of Napo (inclusive of Nantucket) were issued in the aggregate approximately 42,903,018 shares of Company non-voting common stock and 2,282,445 shares of Company voting common stock in full satisfaction of all existing indebtedness then owed by Napo to such creditors, and (iii) Invesco, an existing Napo stockholder, was issued an aggregate of approximately 3,243,243 shares of Company common stock in return for $3 million of new funds invested into the Company by such investor, which were immediately loaned to Napo to partially facilitate the extinguishment of the debt that Napo owed to Nantucket. The minimum Hurdle Amount needed for the vesting of the contingent rights will vary depending on a number of factors (including,
II-3
among other things, the time period over which Nantucket receives specified cash returns in connection with the resale of the Tranche A Shares), and Napo stockholders may not receive any shares of Jaguar common stock in certain circumstances (including if the minimum Hurdle Amount is not satisfied).
Exercise of Warrants
On July 31, 2017, the Company entered into Warrant Exercise Agreements (the "Exercise Agreements") with certain investors who own the right to purchase up to an aggregate of 1,666,668 shares of common stock at an exercise price of $1.00 per share (the "Series C Warrants"). Such holders (the "Existing Holders") own, in the aggregate, Series C Warrants exercisable for 908,334 shares of Common Stock. Pursuant to the Exercise Agreements, the Exercising Holders and the Company agreed that the Exercising Holders would exercise their Series C Warrants with respect to 908,334 shares of Common Stock underlying such Series C Warrants for a reduced exercise price equal to $0.40 per share, as disclosed on our Form 8-K filed with the SEC on July 31, 2017.
KCSA Strategic Communications
On July 31, 2017, we issued 64,866 shares of our voting common stock to KCSA Strategic Communications ("KCSA") pursuant to the Merger Agreement and an agreement between Napo and KCSA, as a complete settlement and satisfaction of Napo's outstanding obligations to KCSA, as disclosed on our Form 10-Q filed with the SEC on November 20, 2017.
On October 30, 2017, pursuant to a debt settlement agreement dated October 30, 2017, we issued 235,134 shares of common stock to KCSA, which together with the 64,866 shares of common stock previously issued on July 31, 2017, fully satisfied $60,000 in debt incurred by us for services rendered by KCSA, as disclosed on our Form 10-K filed with the SEC on April 9, 2018.
L2 Capital, LLC
Shares of our common stock were sold pursuant to the common stock purchase agreement with L2 Capital, LLC, as disclosed on our Form 8-K filed with the SEC on November 24, 2017.
Chicago Venture Partners, L.P.
In November and December 2017, through a series of partial redemptions pursuant to the terms of the Secured Convertible Promissory Note issued to Chicago Venture Partners, L.P. (the "CVP Note") as disclosed in our Form 8-K filed with the SEC on July 3, 2017, we issued 4,000,000 shares of common stock to redeem $601,311.80 of the CVP Note, as disclosed on our Form 10-K filed with the SEC on April 9, 2018.
Sales Pursuant to Share Purchase Agreement
In December 2017, pursuant to a share purchase agreement dated December 27, 2017, we issued 4,010,000 shares of our common stock to certain investors for gross proceeds of $401,000, as disclosed on our Form 10-K filed with the SEC on April 9, 2018.
First Amendment to Note Purchase Agreement and Notes
On December 29, 2017, Napo, our wholly-owned subsidiary, entered into an amendment (the "First Amendment") to the Note Purchase Agreement and Notes with each of the purchasers (the "Purchasers") party to the Note Purchase Agreement, dated March 1, 2017, by and among the Napo and the Purchasers (as amended, the "Note Purchase Agreement"). In connection with the First Amendment, Napo amended the original issue discount exchangeable promissory notes previously issued to the Purchasers on March 1, 2017 (the "First Tranche Notes") and April 27, 2017 (the
II-4
"Second Tranche Notes" and together with the First Tranche Notes, the "Notes") pursuant to the Note Purchase Agreement to, among other things, (a) increase the principal amount outstanding under the First Tranche Notes and the Second Tranche Notes by twelve percent (12%), (b) lower the price at which the Notes are exchangeable for shares (the "Exchange Shares") of the Company's common stock (the "Common Stock") from $0.56 per share to $0.20 per share, and (c) extend the maturity date of the First Tranche Notes from December 1, 2017 to February 15, 2018 and the Second Tranche Notes from January 27, 2018 to April 1, 2018.
In connection with the First Amendment, the Company also issued 2,492,084 shares of common stock to the Purchasers as repayment of $299,050.08 principal amount of the First Tranche Notes, as disclosed on our Form 8-K filed with the SEC on January 2, 2018. Following such repayment and the 12% increase to the outstanding balance of the Notes described above, $435,949.92 and $735,000.00 principal amount remain outstanding under the First Tranche Notes and Second Tranche Notes, respectively.
Investor Awareness, Inc.
In January 2018, pursuant to a consulting agreement dated August 14, 2017, we issued 50,000 shares of our common stock to Investor Awareness, Inc. as partial consideration for financial public relations services rendered, as disclosed on our Form 10-Q filed with the SEC on May 5, 2018.
Issuance Pursuant to Share Purchase Agreements
In January 2018, pursuant to a share purchase agreement dated January 18, 2018, we issued 9,215,900 shares of our common stock to certain investors for gross proceeds of approximately $954,000, as disclosed on our Form 10-Q filed with the SEC on May 5, 2018.
Second Amendment to Note Purchase Agreement and Notes
On February 16, 2018, Napo, our wholly-owned subsidiary, entered into the Second Amendment to Note Purchase Agreement and Notes and Payoff Agreement (the "Second Amendment") with each of the Purchasers party to the Note Purchase Agreement, to extend the maturity date of the Second Tranche Notes from April 1, 2018 to May 1, 2018.
In connection with the Second Amendment, the Company also issued 3,783,444 shares of common stock of the Company to the Purchasers as repayment of the remaining $435,949.92 aggregate principal amount of the original issue discount exchangeable promissory notes previously issued by Napo to the Purchasers on March 1, 2017 pursuant to the Note Purchase Agreement and $18,063.24 in accrued and unpaid interest thereon, ad disclosed on our Form 8-K filed with the SEC on February 16, 2018.
Chicago Venture Partners, L.P.
In January through March 2018, through a series of partial redemptions pursuant to the terms of the CVP Note, as disclosed in our Form 8-K filed with the SEC on July 3, 2017, we issued 8,542,637 shares of common stock to redeem $950,000 of the CVP Note, including accrued and unpaid interest thereon, as disclosed on our Form 10-Q filed with the SEC on May 5, 2018.
Sagard Capital Partners, L.P.
On March 23, 2018, pursuant to a stock purchase agreement, we issued 5,524,926 shares of our Series A Convertible Participating Preferred Stock, $0.0001 par value per share, to Sagard Capital Partners, L.P. for gross proceeds of $9,199,001, as disclosed on our Form 10-Q filed with the SEC on May 5, 2018.
II-5
Sales Pursuant to Share Purchase Agreements
On March 23, 2018, pursuant to share purchase agreements, we issued 29,411,766 shares of our common stock to certain investors for gross proceeds of approximately $5 million. We used net proceeds from the offering to repay certain aged payables relating to our acquisition of Napo in July 2017, as disclosed on our Form 10-Q filed with the SEC on May 5, 2018.
Letter of Credit and Warrant
To satisfy the letter of credit requirement in the Office Lease Agreement with CA-Mission Street Limited Partnership ("Landlord"), effective as of August 30, 2018, Pacific Capital Management, LLC (the "LC Facilitator"), one of the Company's existing shareholders, caused its financial institution to issue a letter of credit in the amount of $475,000 (the "Letter of Credit") on behalf of the Company in favor of Landlord pursuant to the terms of the Landlord Letter of Credit & Warrant Issuance Agreement, dated August 28, 2018, by and between the Company and the LC Facilitator. In consideration of the LC Facilitator causing a Letter of Credit from LC Facilitator's financial institution to be issued to the Landlord, the Company issued to the LC Facilitator a five-year warrant (the "Facilitator Warrant") to purchase 670,586 shares of common stock, subject to adjustment for reclassification or change of the common stock, stock splits, dividends, distributions or changes to the exercise price of the Facilitator Warrant in accordance with the terms of the Facilitator Warrant. The Facilitator Warrant is exercisable commencing after the 7-month anniversary date of issuance, and the exercise price of the Facilitator Warrant is the lower of (i) $0.85 per share and (ii) the average of the closing sales price of the common stock for the 30 consecutive trading days commencing on September 4, 2018.
Charles C. Conte
On September 11, 2018, Charles C. Conte purchased a convertible promissory note in the aggregate principal amount of $111,250 with an exercise price of $0.85 per share of common stock and a warrant exercisable for 33,918 shares of common stock with an exercise price of $1.23 per share.
L2 Capital, LLC
On September 11, 2018, L2 Capital, LLC purchased a convertible promissory note in the aggregate principal amount of $445,000 with an exercise price of $0.85 per share of common stock and a warrant exercisable for 185,417 shares of common stock with an exercise price of $0.90 per share.
The offers, sales, and issuances of the securities described above were deemed to be exempt from registration under the Securities Act in reliance on Section 4(a)(2) of the Securities Act, Regulation D or Regulation S promulgated thereunder as transactions by an issuer not involving a public offering. The recipients of securities in each of these transactions acquired the securities for investment only and not with a view to or for sale in connection with any distribution thereof and appropriate legends were affixed to the securities issued in these transactions. Each of the recipients of securities in these transactions was an accredited or sophisticated person and had adequate access, through employment, business or other relationships, to information about us.
ITEM 16. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES.
- (a)
- Exhibits. The exhibits are incorporated by reference from the Exhibit Index attached hereto.
II-6
The undersigned registrant hereby undertakes to provide to the underwriters at the closing specified in the underwriting agreement certificates in such denominations and registered in such names as required by the underwriters to permit prompt delivery to each purchaser.
The undersigned registrant hereby undertakes that:
- (a)
- For
purposes of determining any liability under the Securities Act, the information omitted from the form of prospectus filed as part of this registration statement
in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be
part of this registration statement as of the time it was declared effective.
- (b)
- For the purpose of determining any liability under the Securities Act, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer, or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question of whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
II-7
- *
- Filed
herewith.
- **
- To be filed by amendment.
II-8
Pursuant to the requirements of the Securities Act of 1933, the registrant has duly caused this Registration Statement to be signed on its behalf by the undersigned, thereunto duly authorized in the City of San Francisco, State of California, on September 11, 2018.
| JAGUAR HEALTH, INC. | ||||||
By: |
/s/ LISA A. CONTE |
|||||
| Name: | Lisa A. Conte | |||||
| Title: | Chief Executive Officer and President | |||||
We, the undersigned officers and Directors of Jaguar Health, Inc., a Delaware corporation, hereby severally constitute and appoint Lisa A. Conte and/or Karen Wright, our true and lawful attorney-in-fact and agents, with full power of substitution and resubstitution for her or him and in her or his name, place and stead, and in any and all capacities, to sign for us and in our names in the capacities indicated below any and all amendments (including post-effective amendments) to this registration statement (or any other registration statement for the same offering that is to be effective upon filing pursuant to Rule 462(b) under the Securities Act of 1933, as amended), and to file the same, with all exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorney-in-fact and agent, full power and authority to do and perform each and every act and thing requisite or necessary to be done in and about the premises, as full to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorney-in-fact and agent, or her or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Act of 1933, this Registration Statement has been signed by the following persons in the capacities and on the date indicated.
Signature
|
Title
|
Date
|
||
|---|---|---|---|---|
| /s/ LISA A. CONTE Lisa A. Conte |
Chief Executive Officer, President and Director (Principal Executive Officer) | September 11, 2018 | ||
/s/ KAREN WRIGHT Karen Wright |
Chief Financial Officer and Treasurer (Principal Financial and Accounting Officer) |
September 11, 2018 |
||
/s/ JAMES J. BOCHNOWSKI James J. Bochnowski |
Chairman of the Board |
September 11, 2018 |
||
Jiahao Qiu |
Director |
II-9
Signature
|
Title
|
Date
|
||
|---|---|---|---|---|
Greg J. Divis |
Director | |||
/s/ JEFFERY C. JOHNSON Jeffery C. Johnson |
Director |
September 11, 2018 |
||
/s/ JOHN MICEK III John Micek III |
Director |
September 11, 2018 |
||
/s/ JONATHAN B. SIEGEL Jonathan B. Siegel |
Director |
September 11, 2018 |
||
/s/ MURRAY DAVID MACNAUGHTAN Murray David MacNaughtan |
Director |
September 11, 2018 |
II-10
QuickLinks -- Click here to rapidly navigate through this document
Consent of Independent Registered Public Accounting Firm
Jaguar
Health, Inc.
San Francisco, California
We hereby consent to the incorporation by reference in the Prospectus constituting a part of this Registration Statement of our report dated April 9, 2018, relating to the consolidated financial statements of Jaguar Health, Inc., which is incorporated by reference in that Prospectus. Our report contains an explanatory paragraph regarding the Company's ability to continue as a going concern.
We also consent to the reference to us under the caption "Experts" in the Prospectus.
/s/
BDO USA, LLP
San Jose, California
September 11, 2018
BDO USA, LLP, a Delaware limited liability partnership, is the U.S. member of BDO International Limited, a UK company limited by guarantee, and forms part of the international BDO network of independent member firms.
BDO is the brand name for the BDO network and for each of the BDO Member Firms.
Consent of Independent Registered Public Accounting Firm
Serious News for Serious Traders! Try StreetInsider.com Premium Free!
You May Also Be Interested In
- Jaguar Health (JAGX) Files $75M Mixed Shelf
- Massimo Group (MAMO) Expands Partnership with Global Omnichannel Retailer for Youth Series Vehicles
- Jaguar Health Appoints Biopharmaceutical Industry Veteran to Execute Company's In-licensing Growth Strategy in the Areas of Cancer and GI Supportive Care
Create E-mail Alert Related Categories
SEC FilingsRelated Entities
S1Sign up for StreetInsider Free!
Receive full access to all new and archived articles, unlimited portfolio tracking, e-mail alerts, custom newswires and RSS feeds - and more!



 Tweet
Tweet Share
Share