Form 8-K ALIMERA SCIENCES INC For: May 02
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): May 2, 2018
ALIMERA SCIENCES, INC.
(Exact name of registrant as specified in its charter)
Delaware | 001-34703 | 20-0028718 | ||||
(State or other Jurisdiction of Incorporation) | (Commission File Number) | (IRS Employer Identification No.) | ||||
6120 Windward Parkway Suite 290 Alpharetta, Georgia | 30005 | |||||
(Address of Principal Executive Offices) | (Zip Code) | |||||
Registrant’s telephone number, including area code: (678) 990-5740
Not Applicable |
(Former name or former address if changed since last report.) |
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
o Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
o Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
o Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
o Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company o
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. o
Item 2.02. Results of Operations and Financial Condition.
On May 2, 2018, Alimera Sciences, Inc. (“Alimera”) issued a press release regarding its results of operations and financial condition for the first quarter ended March 31, 2018. Alimera will be hosting a conference call, as well as a live webcast on the Investor Relations section of its corporate website at www.alimerasciences.com, on May 3, 2018 at 9:00 A.M. ET to discuss its first quarter ended March 31, 2018 financial results and to provide regulatory and commercial updates. The full text of the press release, which includes information regarding Alimera’s use of non-GAAP financial measures, is furnished as Exhibit 99.1 to this Current Report on Form 8-K.
Various statements to be made during the conference call and webcast are “forward-looking statements,” within the meaning of the Private Securities Litigation Reform Act of 1995, regarding, among other things, the positive effects of Alimera’s new loan agreement, including removal of a stringent liquidity requirement present in the former loan agreement, which Alimera believes will allow it to focus on initiatives and opportunities that will grow its business successfully. Words such as “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “plan,” “contemplate,” “predict,” “project,” “target,” “likely,” “potential,” “continue,” “ongoing,” “will,” “would,” “should,” “could,” or the negative of these terms and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Such forward-looking statements are based on current expectations and involve inherent risks and uncertainties, including factors that could delay, divert or change any of them, and could cause actual results to differ materially from those projected in its forward-looking statements. Meaningful factors which could cause actual results to differ include, but are not limited to, (a) whether the new loan agreement will allow the Company to focus on initiatives and opportunities that will grow its business successfully and (b) other factors discussed in the “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” sections of Alimera’s Annual Report on Form 10-K for the year ended December 31, 2017, which is on file with the Securities and Exchange Commission (SEC) and available on the SEC’s website at http://www.sec.gov. Additional factors may also be set forth in those sections of Alimera’s Annual Report on Form 10-Q for the three months ended March 31, 2018, to be filed with the SEC in the near future. In addition to the risks described above and in Alimera’s Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and other filings with the SEC, other unknown or unpredictable factors could affect Alimera’s results. There can be no assurance that the actual results or developments anticipated by Alimera will be realized or, even if substantially realized, that they will have the expected consequences to, or effects on, Alimera. Therefore, no assurance can be given that the outcomes stated in such forward-looking statements and estimates will be achieved.
All forward-looking statements made during the conference call and webcast are expressly qualified by the cautionary statements contained or referred to herein. Alimera cautions investors not to rely too heavily on the forward-looking statements Alimera makes or that are made on its behalf. These forward-looking statements speak only as of the date of this press release (unless another date is indicated). Alimera undertakes no obligation, and specifically declines any obligation, to publicly update or revise any such forward-looking statements, whether as a result of new information, future events or otherwise.
The information in Item 2.02 of this Current Report on Form 8-K and the press release furnished as Exhibit 99.1 hereto shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended (the “Securities Act”), or the Exchange Act, except as expressly set forth by specific reference in such a filing.
Item 7.01. Regulation FD.
The earnings webcast will include certain non-GAAP financial measures. Reconciliations of these non-GAAP financial measures to the comparable measures calculated and presented in accordance with GAAP are included in Alimera’s press release issued May 2, 2018 and attached hereto as Exhibit 99.1.
The information in this Current Report on Form 8-K under Item 7.01 is being “furnished” and not “filed” with the SEC for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities under such section. Furthermore, such information shall not be deemed incorporated by reference in any filing under the Securities Act, or the Exchange Act, unless specifically identified as being incorporated therein by reference.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
ALIMERA SCIENCES, INC. | ||
Dated: May 3, 2018 | By: | /s/ RICHARD S. EISWIRTH, JR. |
Name: | Richard S. Eiswirth, Jr. | |
Title: | President and Chief Financial Officer | |
Exhibit 99.1

Alimera Sciences Reports First Quarter 2018 Results
• | Net Revenue in Q1 of 2018 increased 48% over Q1 of 2017 | |
• | U.S. Net Revenue increased 59% in Q1 of 2018 over Q1 of 2017 | |
• | International Revenue in Q1 of 2018 increased 27% over Q1 of 2017 | |
• | End User Demand in the U.S. was up 21% over Q1 of 2017 | |
ATLANTA, May 2, 2018 (GLOBE NEWSWIRE) -- Alimera Sciences, Inc. (Nasdaq: ALIM) (Alimera), a global pharmaceutical company that specializes in the commercialization and development of prescription ophthalmic pharmaceuticals, today announced financial results for the three months ended March 31, 2018. Alimera will host a conference call on May 3, 2018, at 9:00 a.m. ET to discuss these results.
“During the first quarter of 2018, our expanded U.S. sales presence and the promotion of our strong Phase 4 USER post-market clinical study data drove a 21% increase in U.S. end-user demand, contributing to a 48% increase in consolidated net revenues,” said Dan Myers, Alimera’s Chief Executive Officer. “Our international segment also grew in the first quarter of 2018, with increased revenue in both the markets where we sell direct and in markets where we sell through our distributors.”
First Quarter 2018 Financial Results
Consolidated Net Revenue
Consolidated net revenue increased by approximately $3.2 million, or 48%, to approximately $9.8 million for the three months ended March 31, 2018, compared to consolidated net revenue of approximately $6.6 million for the three months ended March 31, 2017.
Consolidated net revenue increased by approximately $3.2 million, or 48%, to approximately $9.8 million for the three months ended March 31, 2018, compared to consolidated net revenue of approximately $6.6 million for the three months ended March 31, 2017.
U.S. Net Revenue
U.S. net revenue increased by approximately $2.6 million, or 59% to approximately $7.0 million for three months ended March 31, 2018, compared to U.S. net revenue of approximately $4.4 million for the three months ended March 31, 2017. The increase was driven by both increases in end user demand and increases in distributor orders compared to the first quarter of 2017.
U.S. net revenue increased by approximately $2.6 million, or 59% to approximately $7.0 million for three months ended March 31, 2018, compared to U.S. net revenue of approximately $4.4 million for the three months ended March 31, 2017. The increase was driven by both increases in end user demand and increases in distributor orders compared to the first quarter of 2017.
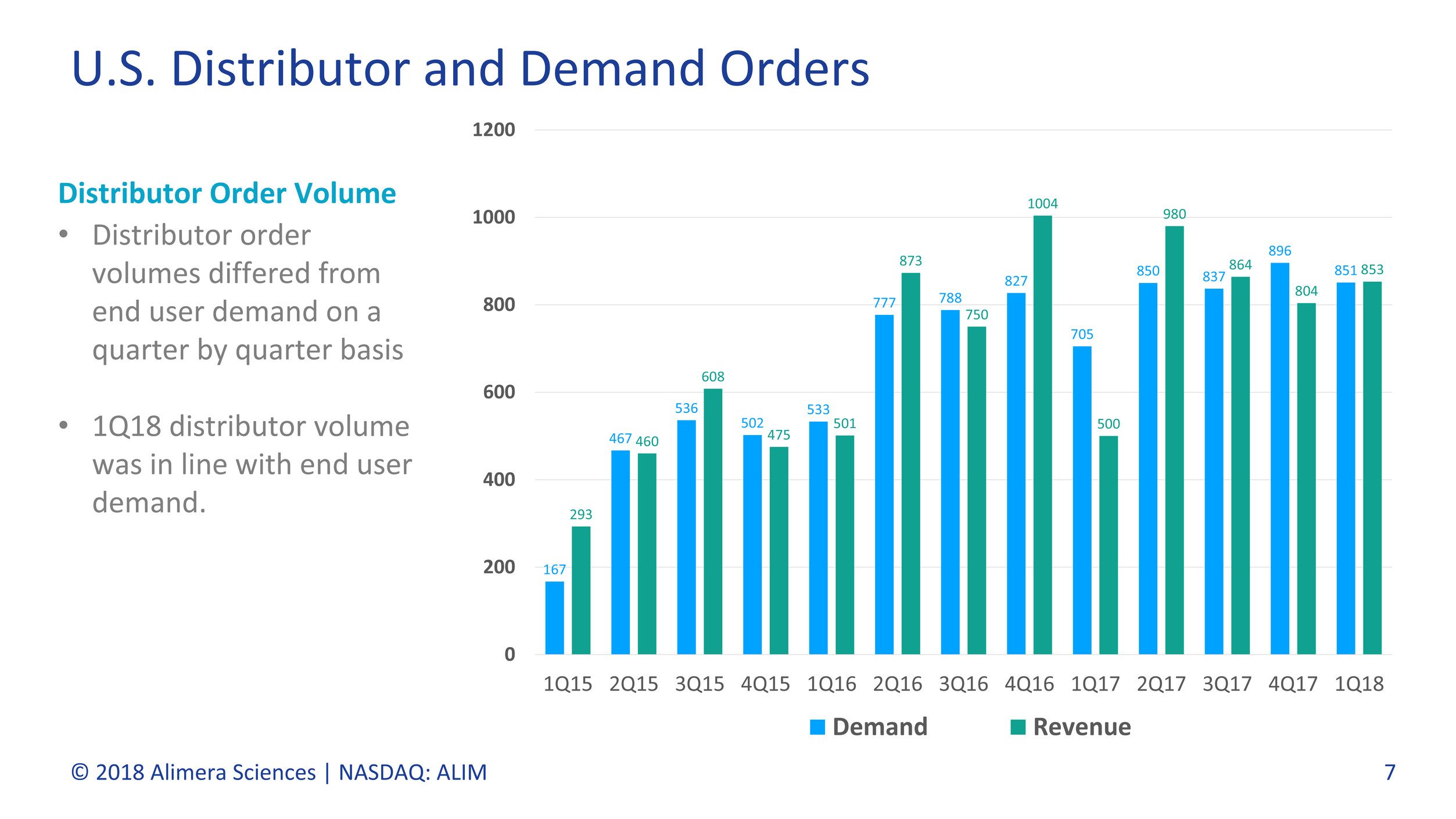
U.S. end user demand, representing units purchased by physicians and pharmacies from the Company’s distributors, increased 21% in the first quarter of 2018. U.S. distributor purchases reported as GAAP revenue increased by 59% over the first quarter of 2017. The difference in growth rates is due to the timing of distributor purchases from quarter to quarter. In the first quarter of 2017, the Company’s distributors purchased approximately 41% units fewer than they sold to end users, using previously acquired inventory to meet demand. In the first quarter of 2018, there was no material difference between the number of units acquired by the Company’s distributors and end user unit sales by those distributors to physicians and pharmacies.
International Net Revenue
Net revenues from the Company’s international segment increased by approximately $600,000, or 27%, to approximately $2.8 million for the three months ended March 31, 2018, compared to approximately $2.2 million for the three months ended March 31, 2017. The growth was driven by an increase of approximately 9% in the countries in which the Company sells direct and approximately $400,000 in sales to international distributors in markets where the Company sells through distributors. Revenues from the Company’s international distributors are comprised of,
Net revenues from the Company’s international segment increased by approximately $600,000, or 27%, to approximately $2.8 million for the three months ended March 31, 2018, compared to approximately $2.2 million for the three months ended March 31, 2017. The growth was driven by an increase of approximately 9% in the countries in which the Company sells direct and approximately $400,000 in sales to international distributors in markets where the Company sells through distributors. Revenues from the Company’s international distributors are comprised of,
depending upon the distribution arrangement, both upfront payments for inventory and a revenue share of the distributors’ sales of product to end users. Revenues from the Company’s international distributors will fluctuate from quarter to quarter based upon the timing of the distributors’ stocking practices.
Operating Expenses
Total operating expenses were approximately $13.3 million for the three months ended March 31, 2018, compared to approximately $11.5 million for the three months ended March 31, 2017, an increase of 16%. “Adjusted Operating Expenses,” a non-GAAP financial measure defined below, were approximately $11.4 million for the three months ended March 31, 2018, compared to Adjusted Operating Expenses of approximately $9.7 million for the three months ended March 31, 2017, an increase of 18%.
Total operating expenses were approximately $13.3 million for the three months ended March 31, 2018, compared to approximately $11.5 million for the three months ended March 31, 2017, an increase of 16%. “Adjusted Operating Expenses,” a non-GAAP financial measure defined below, were approximately $11.4 million for the three months ended March 31, 2018, compared to Adjusted Operating Expenses of approximately $9.7 million for the three months ended March 31, 2017, an increase of 18%.
Research, development and medical affairs expenses for the three months ended March 31, 2018 increased by approximately $700,000, or 33%, to approximately $2.8 million, compared to $2.1 million for the three months ended March 31, 2017. The increase was primarily attributable to the Company receiving a refund from the FDA that offset expenses incurred in the first quarter of 2017, which was not repeated in the 2018 period, and costs incurred related to seeking the approval to treat uveitis in the EU for ILUVIEN.
General and administrative expenses for the three months ended March 31, 2018 increased by approximately $600,000, or 18%, to $3.9 million, compared to $3.3 million for the three months ended March 31, 2017. The increase was primarily attributable to increases in professional fees and personnel costs.
Sales and marketing expenses increased $500,000, or 9%, to $6.0 million for the three months ended March 31, 2018, compared to $5.5 million reported for the three months ended March 31, 2017. The increase was primarily attributable to an increase in personnel costs related to the Company’s expanded sales force in the U.S.
Net loss
Net loss for the three months ended March 31, 2018 was approximately $7.7 million, compared to a net loss of approximately $6.7 million for the three months ended March 31, 2017. The net loss in the 2018 period reflected a loss on early extinguishment of debt of approximately $1.8 million due to the Company paying off its prior loan facility with funds borrowed under its new debt facility with Solar Capital Ltd. (announced in January 2018 and discussed below).
Net loss for the three months ended March 31, 2018 was approximately $7.7 million, compared to a net loss of approximately $6.7 million for the three months ended March 31, 2017. The net loss in the 2018 period reflected a loss on early extinguishment of debt of approximately $1.8 million due to the Company paying off its prior loan facility with funds borrowed under its new debt facility with Solar Capital Ltd. (announced in January 2018 and discussed below).
Basic and diluted net loss per share for the three months ended March 31, 2018 was $(0.11) per share on 69,883,012 weighted average shares outstanding, compared to basic and diluted net loss per share of $(0.10) per share on 64,862,904 weighted average shares outstanding during the three months ended March 31, 2017.
Adjusted EBITDA
“Adjusted EBITDA,” a non-GAAP financial measure defined below, improved by 22% to approximately a $2.9 million loss for the three months ended March 31, 2018, compared to Adjusted EBITDA of approximately a $3.7 million loss for the three months ended March 31, 2017.
“Adjusted EBITDA,” a non-GAAP financial measure defined below, improved by 22% to approximately a $2.9 million loss for the three months ended March 31, 2018, compared to Adjusted EBITDA of approximately a $3.7 million loss for the three months ended March 31, 2017.
Cash and cash equivalents; Solar Loan Agreement
As of March 31, 2018, Alimera had cash and cash equivalents of approximately $20.3 million. In early January 2018 Alimera entered into a $40 million term loan agreement with Solar Capital Ltd. Alimera used the proceeds to pay off its then existing $35 million term loan, along with related fees and expenses, and to provide additional working capital for general corporate purposes. Unlike the previous loan facility, the new loan agreement does not contain a stringent liquidity requirement. The Company believes the elimination of this liquidity requirement will
As of March 31, 2018, Alimera had cash and cash equivalents of approximately $20.3 million. In early January 2018 Alimera entered into a $40 million term loan agreement with Solar Capital Ltd. Alimera used the proceeds to pay off its then existing $35 million term loan, along with related fees and expenses, and to provide additional working capital for general corporate purposes. Unlike the previous loan facility, the new loan agreement does not contain a stringent liquidity requirement. The Company believes the elimination of this liquidity requirement will
allow the Company to focus on operating initiatives and opportunities that will grow the business successfully.
Definitions of Non-GAAP Financial Measures
For purposes of this press release, “Adjusted Operating Expenses” is operating expenses minus depreciation, amortization and stock-based compensation expenses. “Adjusted EBITDA” is earnings before interest, taxes, depreciation, amortization, stock-based compensation expenses, net unrealized gains and losses from foreign currency exchange transactions, gains and losses from the change in the fair value of derivative warrant liability and losses on extinguishment of debt. The Company provides non-GAAP financial information because it believes this information can enhance an overall understanding of its financial performance when considered together with GAAP figures. Refer to the sections of this press release entitled “Non-GAAP Financial Measures” and “Reconciliation of GAAP Measures to Non-GAAP Adjusted Measures,” which includes Adjusted Operating Expenses and Adjusted EBITDA.
Conference Call to be Held May 3, 2018
A conference call will be hosted by Dan Myers, Chief Executive Officer, and Rick Eiswirth, President and Chief Financial Officer, to discuss the results. The call will be held at 9:00 a.m. ET, on May 3, 2018. Please refer to the information below for conference call dial-in information and webcast registration.
Conference date: May 3, 2018, 9:00 AM ET
Conference dial-in: 877-269-7756
International dial-in: 201-689-7817
Conference Call Name: Alimera Sciences First Quarter 2018 Results Call
Webcast Registration: Click Here
Following the live call, a replay will be available on Alimera’s website, www.alimerasciences.com, under “Investor Relations.”
About Alimera Sciences
Alimera, founded in June 2003, is a pharmaceutical company that specializes in the commercialization and development of prescription ophthalmic pharmaceuticals. Alimera’s focus is on diseases affecting the back of the eye, or retina, because these diseases are not well treated with current therapies and will affect millions of people in aging populations. Alimera’s commitment to retina specialists and their patients is manifest in its product portfolio. For more information, please visit www.alimerasciences.com.
Non-GAAP Financial Measures
This press release contains a discussion of non-GAAP financial measures, as defined in Regulation G promulgated under the Securities Exchange Act of 1934, as amended. Alimera reports its financial results in compliance with GAAP but believes that the non-GAAP measures of Adjusted Operating Expenses and Adjusted EBITDA are useful measures in evaluating Alimera’s operating performance. Alimera uses Adjusted Operating Expenses and Adjusted EBITDA in the management of its business. Accordingly, Adjusted Operating Expenses and Adjusted EBITDA for the three months ended March 31, 2018 have been presented in certain instances excluding items identified in the reconciliations provided. For a reconciliation of these non-GAAP financial measures to their most directly comparable GAAP financial measures, see the tables below.
These non-GAAP financial measures, as presented, may not be comparable to similarly titled measures reported by other companies because not all companies may calculate these measures in an identical manner. Therefore, they are not necessarily an accurate measure of comparison between companies.
The presentation of these non-GAAP financial measures is not intended to be considered in isolation or as a substitute for guidance prepared in accordance with GAAP. The principal limitation of these non-GAAP financial measures is that they exclude significant elements required by GAAP to be recorded in Alimera’s financial statements. In addition, they are subject to inherent limitations as they reflect the exercise of judgments by management in determining these non-GAAP financial measures. To compensate for these limitations, Alimera presents its non-GAAP financial results in connection with its GAAP results. Investors are encouraged to review the reconciliation of each of our non-GAAP financial measures to its most directly comparable GAAP financial measure.
Forward Looking Statements
This press release contains “forward-looking statements,” within the meaning of the Private Securities Litigation Reform Act of 1995, regarding the positive effects of Alimera’s new loan agreement, including removal of a stringent liquidity requirement present in the former loan agreement, which Alimera believes will allow it to focus on initiatives and opportunities that will grow its business successfully.
Such forward-looking statements are based on current expectations and involve inherent risks and uncertainties, including factors that could delay, divert or change any of them, and could cause actual results to differ materially from those projected in its forward-looking statements. Meaningful factors which could cause actual results to differ include, but are not limited to, (a) whether the new loan agreement will allow the Company to focus on initiatives and opportunities that will grow its business successfully and (b) other factors discussed in the “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” sections of Alimera’s Annual Report on Form 10-K for the year ended December 31, 2017, which are on file with the Securities and Exchange Commission (SEC) and available on the SEC’s website at http://www.sec.gov. Additional factors may also be set forth in those sections of Alimera’s Quarterly Report on Form 10-Q for the three months ended March 31, 2018, to be filed with the SEC in the near future.
In addition to the risks described above and in Alimera’s Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and other filings with the SEC, other unknown or unpredictable factors could affect Alimera’s results. There can be no assurance that the actual results or developments anticipated by Alimera will be realized or, even if substantially realized, that they will have the expected consequences to, or effects on, Alimera. Therefore, no assurance can be given that the outcomes stated in such forward-looking statements and estimates will be achieved. All forward-looking statements contained in this press release are expressly qualified by the cautionary statements contained or referred to herein. Alimera cautions investors not to rely too heavily on the forward-looking statements Alimera makes or that are made on its behalf. These forward-looking statements speak only as of the date of this press release (unless another date is indicated). Alimera undertakes no obligation, and specifically declines any obligation, to publicly update or revise any such forward-looking statements, whether as a result of new information, future events or otherwise.
ALIMERA SCIENCES, INC.
CONSOLIDATED BALANCE SHEETS
(in thousands)
March 31, 2018 | December 31, 2017 | ||||||
(unaudited) | |||||||
CURRENT ASSETS: | |||||||
Cash and cash equivalents | $ | 20,253 | $ | 24,067 | |||
Restricted cash | 35 | 34 | |||||
Accounts receivable, net | 12,354 | 11,435 | |||||
Prepaid expenses and other current assets | 1,992 | 2,278 | |||||
Inventory | 1,224 | 1508 | |||||
Total current assets | 35,858 | 39,322 | |||||
NON-CURRENT ASSETS: | |||||||
Property and equipment, net | 1,531 | 1,410 | |||||
Intangible asset, net | 18,185 | 18,664 | |||||
Deferred tax asset | 543 | 528 | |||||
TOTAL ASSETS | $ | 56,117 | $ | 59,924 | |||
CURRENT LIABILITIES: | |||||||
Accounts payable | $ | 4,969 | $ | 5,905 | |||
Accrued expenses | 2,280 | 3,582 | |||||
Capital lease obligations | 216 | 184 | |||||
Total current liabilities | 7,465 | 9,671 | |||||
NON-CURRENT LIABILITIES: | |||||||
Note payable | 37,254 | 34,365 | |||||
Capital lease obligations — less current portion | 283 | 203 | |||||
Other non-current liabilities | 2,564 | 766 | |||||
COMMITMENTS AND CONTINGENCIES | |||||||
STOCKHOLDERS’ EQUITY: | |||||||
Preferred stock: | |||||||
Series A Convertible Preferred Stock | 19,227 | 19,227 | |||||
Series B Convertible Preferred Stock | 49,568 | 49,568 | |||||
Common stock | 700 | 691 | |||||
Additional paid-in capital | 342,822 | 341,622 | |||||
Common stock warrants | 3,707 | 3,707 | |||||
Accumulated deficit | (406,758 | ) | (399,075 | ) | |||
Accumulated other comprehensive loss | (715 | ) | (821 | ) | |||
TOTAL STOCKHOLDERS’ EQUITY | 8,551 | 14,919 | |||||
TOTAL LIABILITIES AND STOCKHOLDERS’ EQUITY | $ | 56,117 | $ | 59,924 | |||
ALIMERA SCIENCES, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
FOR THE THREE MONTHS ENDED MARCH 31, 2018 AND 2017
(in thousands, except share and per share data)
Three Months Ended March 31, | |||||||
2018 | 2017 | ||||||
(unaudited) | (unaudited) | ||||||
NET REVENUE | $ | 9,802 | $ | 6,618 | |||
COST OF GOODS SOLD, EXCLUDING DEPRECIATION AND AMORTIZATION | (1,276 | ) | (587 | ) | |||
GROSS PROFIT | 8,526 | 6,031 | |||||
RESEARCH, DEVELOPMENT AND MEDICAL AFFAIRS EXPENSES | 2,822 | 2,110 | |||||
GENERAL AND ADMINISTRATIVE EXPENSES | 3,855 | 3,264 | |||||
SALES AND MARKETING EXPENSES | 5,969 | 5,502 | |||||
DEPRECIATION AND AMORTIZATION | 649 | 666 | |||||
OPERATING EXPENSES | 13,295 | 11,542 | |||||
NET LOSS FROM OPERATIONS | (4,769 | ) | (5,511 | ) | |||
INTEREST EXPENSE AND OTHER | (1,151 | ) | (1,337 | ) | |||
UNREALIZED FOREIGN CURRENCY GAIN (LOSS), NET | 2 | (28 | ) | ||||
LOSS ON EARLY EXTINGUISHMENT OF DEBT | (1,766 | ) | — | ||||
CHANGE IN FAIR VALUE OF DERIVATIVE WARRANT LIABILITY | — | 167 | |||||
NET LOSS BEFORE TAXES | (7,684 | ) | (6,709 | ) | |||
PROVISION FOR TAXES | — | (26 | ) | ||||
NET LOSS | $ | (7,684 | ) | $ | (6,735 | ) | |
NET LOSS PER SHARE — Basic and diluted | $ | (0.11 | ) | $ | (0.10 | ) | |
WEIGHTED AVERAGE SHARES OUTSTANDING — Basic and diluted | 69,883,012 | 64,862,904 | |||||
RECONCILIATION OF GAAP MEASURES TO NON-GAAP ADJUSTED MEASURES | |||||||
GAAP NET LOSS TO NON-GAAP ADJUSTED EBITDA | |||||||
(in thousands) | |||||||
Three Months Ended March 31, | |||||||
2018 | 2017 | ||||||
(unaudited) | (unaudited) | ||||||
GAAP NET LOSS | $ | (7,684 | ) | $ | (6,735 | ) | |
Adjustments to net loss: | |||||||
Interest expense and other | 1,151 | 1,337 | |||||
Provision for taxes | 0 | 26 | |||||
Depreciation and amortization | 649 | 666 | |||||
Stock-based compensation expenses | 1,207 | 1,166 | |||||
Unrealized foreign currency exchange (Gains) Losses | (2 | ) | 28 | ||||
Change in the fair value of derivative warrant liability | — | (167 | ) | ||||
Loss on early extinguishment of debt | 1,766 | — | |||||
NON-GAAP ADJUSTED EBITDA | $ | (2,913 | ) | $ | (3,679 | ) | |
GAAP OPERATING EXPENSES TO NON-GAAP ADJUSTED OPERATING EXPENSES | |||||||
Three Months Ended March 31, | |||||||
2018 | 2017 | ||||||
(unaudited) | (unaudited) | ||||||
GAAP OPERATING EXPENSES | $ | 13,295 | $ | 11,542 | |||
Adjustments to Operating Expenses: | |||||||
Depreciation and Amortization | (649 | ) | (666 | ) | |||
Stock-Based Compensation | (1,207 | ) | (1,166 | ) | |||
NON-GAAP ADJUSTED OPERATING EXPENSES | $ | 11,439 | $ | 9,710 | |||
ALIMERA SCIENCES, INC.
CONSOLIDATING STATEMENTS OF OPERATIONS
FOR THE THREE MONTHS ENDED MARCH 31, 2018 AND 2017
(in thousands)
Three Months Ended March 31, 2018 | |||||||||||||||
U.S. | International | Other | Consolidated | ||||||||||||
(unaudited) | |||||||||||||||
NET REVENUE | $ | 6,977 | $ | 2,825 | $ | — | $ | 9,802 | |||||||
COST OF GOODS SOLD, EXCLUDING DEPRECIATION AND AMORTIZATION | (885 | ) | (391 | ) | — | (1,276 | ) | ||||||||
GROSS PROFIT | 6,092 | 2,434 | — | 8,526 | |||||||||||
RESEARCH, DEVELOPMENT AND MEDICAL AFFAIRS EXPENSES | 1,640 | 950 | 232 | 2,822 | |||||||||||
GENERAL AND ADMINISTRATIVE EXPENSES | 2,293 | 907 | 655 | 3,855 | |||||||||||
SALES AND MARKETING EXPENSES | 4,371 | 1,278 | 320 | 5,969 | |||||||||||
DEPRECIATION AND AMORTIZATION | — | — | 649 | 649 | |||||||||||
OPERATING EXPENSES | 8,304 | 3,135 | 1,856 | 13,295 | |||||||||||
SEGMENT LOSS FROM OPERATIONS | (2,212 | ) | (701 | ) | (1,856 | ) | (4,769 | ) | |||||||
OTHER INCOME AND EXPENSES, NET | (2,915 | ) | (2,915 | ) | |||||||||||
NET LOSS BEFORE TAXES | $ | (7,684 | ) | ||||||||||||
Three Months Ended March 31, 2017 | |||||||||||||||
U.S. | International | Other | Consolidated | ||||||||||||
(unaudited) | |||||||||||||||
NET REVENUE | $ | 4,445 | $ | 2,173 | $ | — | $ | 6,618 | |||||||
COST OF GOODS SOLD, EXCLUDING DEPRECIATION AND AMORTIZATION | (448 | ) | (139 | ) | — | (587 | ) | ||||||||
GROSS PROFIT | 3,997 | 2,034 | — | 6,031 | |||||||||||
RESEARCH, DEVELOPMENT AND MEDICAL AFFAIRS EXPENSES | 1,158 | 741 | 211 | 2,110 | |||||||||||
GENERAL AND ADMINISTRATIVE EXPENSES | 1,705 | 918 | 641 | 3,264 | |||||||||||
SALES AND MARKETING EXPENSES | 4,045 | 1,143 | 314 | 5,502 | |||||||||||
DEPRECIATION AND AMORTIZATION | — | — | 666 | 666 | |||||||||||
OPERATING EXPENSES | 6,908 | 2,802 | 1,832 | 11,542 | |||||||||||
SEGMENT LOSS FROM OPERATIONS | (2,911 | ) | (768 | ) | (1,832 | ) | (5,511 | ) | |||||||
OTHER INCOME AND EXPENSES, NET | (1,198 | ) | (1,198 | ) | |||||||||||
NET LOSS BEFORE TAXES | $ | (6,709 | ) | ||||||||||||

First Quarter 2018 Earnings Presentation
Thursday, May 3rd 2018
Exhibit 99.2

This presentation contains “forward-looking statements,” within the meaning of the Private Securities Litigation Reform Act of 1995, regarding, among
other things,, the potential approval and commercialization of ILUVIEN for the treatment of posterior uveitis in Europe. Such forward-looking
statements are based on current expectations and involve inherent risks and uncertainties, including factors that could delay, divert or change any of
them, and could cause actual results to differ materially from those projected in its forward-looking statements. Words such as “anticipate,” “believe,”
“estimate,” “expect,” “intend,” “may,” “plan,” “contemplate,” “predict,” “project,” “target,” “likely,” “potential,” “continue,” “ongoing,” “will,” “would,”
“should,” “could,” or the negative of these terms and similar expressions are intended to identify forward-looking statements, although not all forward-
looking statements contain these identifying words. Such forward-looking statements are based on current expectations and involve inherent risks and
uncertainties, including factors that could delay, divert or change any of them, and could cause actual results to differ materially from those projected in
its forward-looking statements. Meaningful factors which could cause actual results to differ include, but are not limited to, that the estimated size of
the DME market is not a $2 billion opportunity and the market for uveitis is not 20% of DME, continued market acceptance of ILUVIEN in the U.S. and
Europe, including physicians' ability to obtain reimbursement, as well as other factors discussed in the “Risk Factors” and “Management's Discussion
and Analysis of Financial Condition and Results of Operations” sections of Alimera's Annual Report on Form 10-K for the year ended December 31, 2017,
which is on file with the Securities and Exchange Commission (SEC) and available on the SEC's website at http://www.sec.gov. Additional factors may
also be set forth in those sections of Alimera’s Quarterly Report on Form 10-Q for the three months ended March 31, 2018, to be filed In addition to the
risks described above and in Alimera's Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and other filings with
the SEC, other unknown or unpredictable factors also could affect Alimera's results. There can be no assurance that the actual results or developments
anticipated by Alimera will be realized or, even if substantially realized, that they will have the expected consequences to, or effects on, Alimera.
Therefore, no assurance can be given that the outcomes stated in such forward-looking statements and estimates will be achieved. All forward-looking
statements contained in this presentation are expressly qualified by the cautionary statements contained or referred to herein. Alimera cautions
investors not to rely too heavily on the forward-looking statements Alimera makes or that are made on its behalf. These forward-looking statements
speak only as of the date of this presentation (unless another date is indicated). Alimera undertakes no obligation, and specifically declines any
obligation, to publicly update or revise any such forward-looking statements, whether as a result of new information, future events or otherwise.
Safe Harbor Statement
2
© 2018 Alimera Sciences | NASDAQ: ALIM

3
First Quarter 2018 Overview
Significant Revenue Growth
• 48% increase compared to 1Q17
© 2018 Alimera Sciences | NASDAQ: ALIM
U.S.
• $7.0 million in 1Q18; 59% revenue increase in
the U.S. compared to 1Q17
• 21% increase in end user demand
International
• $2.8 million in 1Q18
• 27% revenue increase internationally
compared to 1Q17
$6.7M
$9.8M
Revenue
+48%
Revenue
Growth
1Q17 1Q18

4
ILUVIEN Presentations at ARVO*
© 2018 Alimera Sciences | NASDAQ: ALIM
19 ILUVIEN presentations at ARVO 2018
Data from multiple countries represented
• Eight U.S. presentations on diabetic macular edema (DME)
• Ten EU/Middle East Presentations on DME
• One European presentation on uveitis
Real world support for ILUVIEN for DME
• Three-year experience
• Improved treatment burden for patients
• Predictability of intraocular pressure (IOP) response
• Improvement of the retinal nerve fiber layer
*Association for Research in Vision and Ophthalmology

5
ILUVIEN for Non-Infectious Posterior Uveitis
90.5%
59.6%
27.6%
25.7%
PSV-FAI-001 PSV-FAI-005
Percentage of Patients with Uveitis
Recurrence
FAI InsertSham
• Application for non-infectious posterior uveitis indication
submitted in 17 countries in Europe in December of 2017.
• Alimera plans to file additional requested follow-up
clinical data by end of 2018.
• Expected approval 1H19.
• Data from two phase 3 clinical trials of the FAI insert
demonstrated:
• A sustained prevention of recurrence of noninfectious
posterior uveitis.
• Significantly reduced need for adjunctive therapy.
• Estimated market size -- 20% of DME market.
• Steroids are the current standard of care, no anti-VEGF
competition (i.e. Lucentis, Eylea).
© 2018 Alimera Sciences | NASDAQ: ALIM

6
Growth of ILUVIEN Usage in United States
© 2018 Alimera Sciences | NASDAQ: ALIM
0
1000
2000
3000
4000
5000
6000
7000
8000
9000
10000
~Cumulative Eyes Treated with ILUVIEN - U.S.
7
U.S. Distributor and Demand Orders
© 2018 Alimera Sciences | NASDAQ: ALIM
167
467
536
502
533
777 788
827
705
850 837
896
851
293
460
608
475
501
873
750
1004
500
980
864
804
853
0
200
400
600
800
1000
1200
1Q15 2Q15 3Q15 4Q15 1Q16 2Q16 3Q16 4Q16 1Q17 2Q17 3Q17 4Q17 1Q18
Demand Revenue
Distributor Order Volume
• Distributor order
volumes differed from
end user demand on a
quarter by quarter basis
• 1Q18 distributor volume
was in line with end user
demand.

8
U.S. Demand Orders
© 2018 Alimera Sciences | NASDAQ: ALIM
167
467
536
502
533
777 788
827
705
850 837
896
851
0
100
200
300
400
500
600
700
800
900
1000
First Quarter Second Quarter Third Quarter Fourth Quarter
2015 2016 2017 2018
U.S. End User Demand
• Units sold from our distributor
partners to physicians and pharmacies
• Represents actual usage of ILUVIEN.
U.S. Usage Growth
• U.S. end user demand grew 21% in
1Q18.

9
Income Statement
© 2018 Alimera Sciences | NASDAQ: ALIM
($000’s)
Three Months Ended
March 31, 2018
Three Months Ended
March 31, 2017
Revenue $9,802 $6,618
Operating Expenses $13,295 $11,542
Net Loss ($7,684) ($6,735)
Adj. EBITDA* ($2,913) ($3,679)
*Adjusted EBITDA is earnings before interest taxes, depreciation, amortization, stock-based compensation expenses, net unrealized gains and losses from foreign currency
exchange transactions, gains and losses from the change in the fair value of derivative warrant liability and losses on extinguishment of debt. See slides 11 and 12 of this
presentation for reconciliation of this Non-GAAP financial measure.

10
First Quarter 2018 Overview
Significant Revenue Growth
• 48% increase compared to 1Q17
© 2018 Alimera Sciences | NASDAQ: ALIM
U.S. Revenue
• $7.0 million in 1Q18; 59% revenue increase in
the U.S. compared to 1Q17
• 21% increase in end user demand
International
• $2.8 million in 1Q18
• 27% revenue increase internationally
compared to 1Q17
$6.7M
$9.8M
Revenue
+48%
Revenue
Growth
1Q17 1Q18

11
Reconciliation of GAAP Net Loss to NON-GAAP Adjusted EBITDA
© 2018 Alimera Sciences | NASDAQ: ALIM
Alimera believes that the non-GAAP financial information provided in this presentation can assist investors in the overall understanding of its financial
performance when considered together with GAAP figures.
This presentation contains a discussion of non-GAAP financial measures, as defined in Regulation G of the Securities Exchange Act of 1934, as amended.
Alimera reports its financial results in compliance with GAAP, but believes that the non-GAAP measures of Adjusted EBITDA will be a more relevant
measure of Alimera's operating performance. For the purpose of this presentation, “Adjusted EBITDA” is adjusted earnings before interest, taxes,
depreciation, amortization, non-cash stock-based compensation expense, and to the extent they are included in the calculation of earnings, net
unrealized gain (loss) from foreign currency exchange transactions, gains (losses) from the change in the fair value of derivative warrant liability and
losses on the extinguishment of debt. Alimera uses Adjusted EBITDA in the management of its business. Accordingly, Adjusted EBITDA for the first
quarter of 2018 has been presented in certain instances excluding items identified in the reconciliations provided.
This non-GAAP financial measure, as presented, may not be comparable to similarly titled measures reported by other companies since not all
companies may calculate these measures in an identical manner and, therefore, they are not necessarily an accurate measure of comparison between
companies.
The presentation of this non-GAAP financial measure is not intended to be considered in isolation or as a substitute for guidance prepared in accordance
with GAAP. The principal limitation of this non-GAAP financial measure is that they exclude significant elements that are required by GAAP to be
recorded in Alimera's financial statements. In addition, this measure is subject to inherent limitations as it reflects the exercise of judgments by
management in determining this non-GAAP financial measures. In order to compensate for these limitations, Alimera presents its non-GAAP financial
results in connection with its GAAP results. Investors are encouraged to review the reconciliation of this non-GAAP financial measures to its most directly
comparable GAAP financial measure as set forth on page 12 of this presentation.

12
Reconciliation of GAAP Net Loss to NON-GAAP Adjusted EBITDA
© 2018 Alimera Sciences | NASDAQ: ALIM
Three Months Ended March 31,
($000’s) 2018 2017
GAAP Net Loss ($7,684) ($6,735)
Adjustments to Net Loss
Interest Expense, Net and Other $1,151 $1,337
Provision for Taxes - $26
Depreciation and Amortization $649 $666
Stock-Based Compensation $1,207 $1,166
Unrealized Foreign Currency Exchange
(Gains) Losses
(2) 28
Change in the Fair Value of Derivative
Warrant Liability
- ($167)
Loss on Early Extinguishment of Debt $1,766 -
Non-GAAP Adjusted EBITDA ($2,913) ($3,679)

First Quarter 2018 Earnings Presentation
Thursday, May 3rd 2018
Serious News for Serious Traders! Try StreetInsider.com Premium Free!
You May Also Be Interested In
- Craig-Hallum Assumes Alimera Sciences (ALIM) at Buy
- Australian Oilseeds, Largest APAC Producer of Non-Chemical, Non-GMO “Cold-Processing” Vegetable Oil, Enters into Contract with Woolworths and Costco Australia.
- Aristocrat Leisure Completes Acquisition of Neo Group Ltd. (f/k/a NeoGames) for $29.50 per Share
Create E-mail Alert Related Categories
SEC FilingsSign up for StreetInsider Free!
Receive full access to all new and archived articles, unlimited portfolio tracking, e-mail alerts, custom newswires and RSS feeds - and more!



 Tweet
Tweet Share
Share