Form 8-K TELA Bio, Inc. For: Aug 15
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of The Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
(Exact name of registrant as specified in its charter)
|
(State or other jurisdiction of incorporation) |
(Commission File Number) |
(I.R.S. Employer Identification No.) |
|
|
|
|
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including
area code: (
Not Applicable
(Former name or former address, if changed since last report.)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant
to Section 13(a) of the Exchange Act.
| Item 7.01 | Regulation FD Disclosure. |
On August 15, 2022, TELA Bio, Inc. (the “Company”) updated information reflected in a corporate slide deck, which representatives of the Company will use in various meetings with investors from time to time. A copy of the presentation is attached hereto as Exhibit 99.1 and incorporated herein by reference.
The information furnished pursuant to Item 7.01, including Exhibit 99.1, shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, and shall not be deemed to be incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as expressly set forth by specific reference in such filing.
| Item 9.01 | Financial Statements and Exhibits. |
(d) Exhibits
The following exhibits are being furnished herewith:
| Exhibit No. | Document | |
| 99.1 | Corporate Slide Deck, dated August 15, 2022. | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document). |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| TELA BIO, INC. | ||
| By: | /s/ Antony Koblish | |
| Name: | Antony Koblish | |
| Title: | President, Chief Executive Officer and Director | |
Date: August 15, 2022
Exhibit 99.1

INVESTOR PRESENTATION August 2022

Forward Looking Statements This presentation contains forward - looking statements within the meaning of The Private Securities Litigation Reform Act of 1995 . All statements other than statements of historical facts contained in this document, including but not limited to statements regarding possible or assumed future res ults of operations, business strategies, development plans, regulatory activities, market opportunity competitive position, potential growth opportunities, and the ef fec ts of competition, are forward - looking statements. These statements involve known and unknown risks, uncertainties and other important factors that may cause TELA B io, Inc.’s (the “Company”) actual results, performance or achievements to be materially different from any future results, performance or achievements expresse d o r implied by the forward - looking statements. In some cases, you can identify forward - looking statements by terms such as “may,” “will,” “should,” “expect,” “plan ,” “aim,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential” or “continue” or the negative of these term s o r other similar expressions. The forward - looking statements in this presentation are only predictions. The Company has based these forward - looking statements largely on its curr ent expectations and projections about future events and financial trends that it believes may affect the Company’s business, financial condition and results of operations. These forward - looking statements speak only as of the date of this presentation and are subject to a number of risks, uncertainties and assumptions , s ome of which cannot be predicted or quantified and some of which are beyond the Company’s control, including, among others: the impact to the Company's business of the ongoing COVID - 19 pandemic and the development of new variants of COVID - 19, including but not limited to any impact on the Company's ability to market its products, demand for the Company's products due to deferral of procedures using the Company's products, the labor and staffing environment in the healthcare ind ust ry, or disruption in the Company's supply chain, the Company's ability to achieve or sustain profitability, the Company's ability to gain market acceptance for the Company's products and to accurately forecast and meet customer demand, the Company's ability to compete successfully, that data from earlier studies related to t he Company’s products and interim data from ongoing studies may not be replicated in later studies or indicative of future data, that data obtained from clinical st udi es utilizing the Company’s products may not be indicative of outcomes in other surgical settings, the Company's ability to enhance the Company's product offerings, devel opm ent and manufacturing problems, capacity constraints or delays in production of the Company's products, maintenance of coverage and adequate reimbursement fo r p rocedures using the Company's products, product defects or failures. These and other risks and uncertainties are described more fully in the "Risk Factors" se ction and elsewhere in the Company's filings with the Securities and Exchange Commission and available at www.sec.gov. You should not rely on these forward - looking s tatements as predictions of future events. The events and circumstances reflected in the Company’s forward - looking statements may not be achieved or occur, and act ual results could differ materially from those projected in the forward - looking statements. Moreover, the Company operates in a dynamic industry and economy. New ri sk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties that the Compan y m ay face. Except as required by applicable law, we do not plan to publicly update or revise any forward - looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.

TELA Bio, Inc. • ~$2.2B US market opportunity 1 • Multiple innovative products offering attractive value propositions for hospitals • Growing portfolio of offerings • Compelling, expanding set of clinical data • Increasing GPO/market access 1 Management estimate. $2.2B total includes $1.5B hernia & abdominal wall reconstruction, $0.7B plastic reconstructive surgery. Redefining soft tissue preservation and restoration with a differentiated category of tissue reinforcement materials

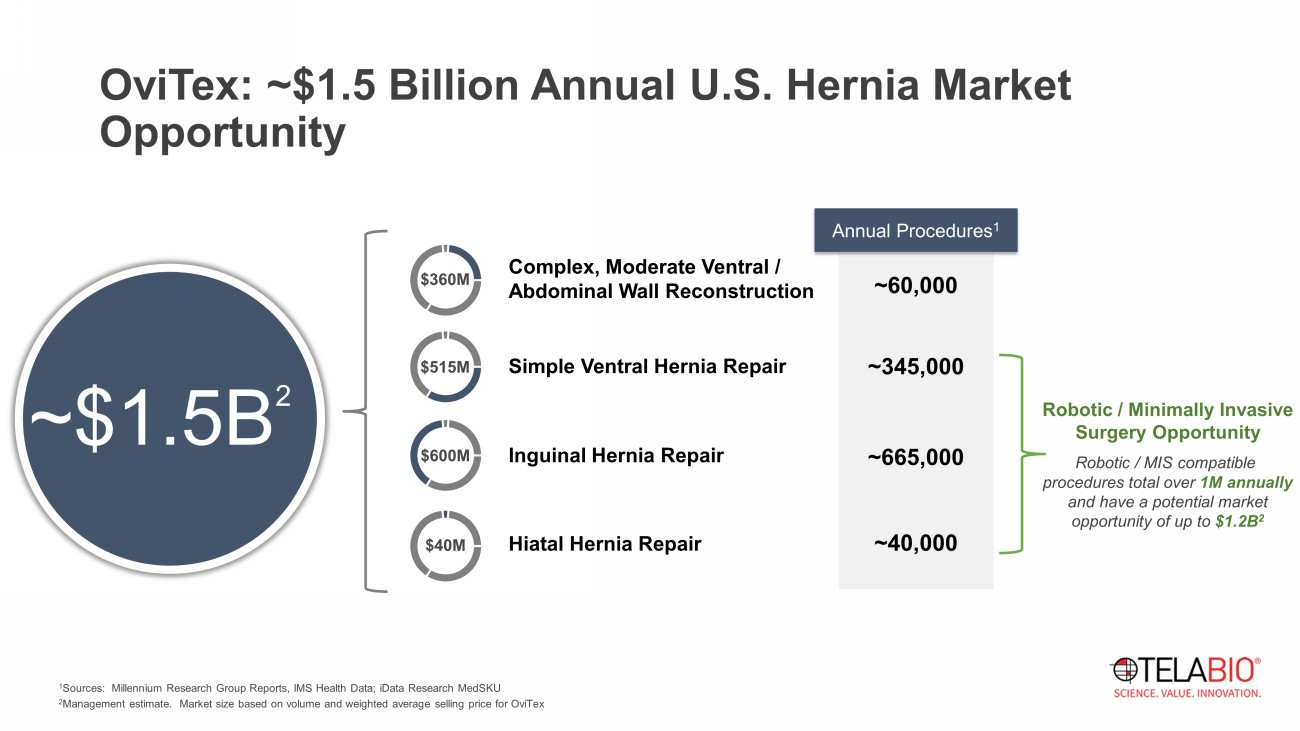
Complex, Moderate Ventral / Abdominal Wall Reconstruction Simple Ventral Hernia Repair Inguinal Hernia Repair Hiatal Hernia Repair ~60,000 ~345,000 ~665,000 ~40,000 $360M $515M $600M $40M 1 Sources: Millennium Research Group Reports, IMS Health Data; iData Research MedSKU 2 Management estimate. Market size based on volume and weighted average selling price for OviTex Annual Procedures 1 ~$1.5B 2 Robotic / Minimally Invasive Surgery Opportunity OviTex: ~$1.5 Billion Annual U.S. Hernia Market Opportunity Robotic / MIS compatible procedures total over 1M annually and have a potential market opportunity of up to $1.2B 2

OviTex Portfolio: Designed for a Range of Hernia Patients and Surgical Techniques • Coated resorbable synthetic meshes • Biologic meshes • Biologic meshes CONFIGURATION OviTex 2S 8 - layer device, with 2 “smooth sides” suitable for intraperitoneal placement Robot Compatible 1 : No Strength 2 : +++ Viscera Contact 2 : Yes OviTex 1S 6 - layer device, with “smooth side” suitable for intraperitoneal placement Robot Compatible 1 : Yes Strength 2 : ++ Viscera Contact 2 : Yes COMPETITIVE SET • Coated resorbable synthetic meshes • Biologic meshes OviTex LPR 4 - layer device, with “smooth side” suitable for intraperitoneal placement Robot Compatible 1 : Yes Strength 2 : + Viscera Contact 2 : Yes • Resorbable synthetic meshes • Biologic meshes OviTex 4 - layer device, not intended for intraperitoneal placement Robot Compatible 1 : Yes Strength 2 : + Viscera Contact 2 : Not recommended Images represent permanent polymer OviTex products. Resorbable polymer products have clear polymer. All trademarks and regist ere d marks are property of their respective owners. 1. Robot compatibility based on use of 10mm trocar. Robot compatibility of LPR and OviTex include sizes 400 cm 2 or less. Robot compatibility of OviTex 1S includes sizes 200 cm 2 or less 2. Biomechanical data on file. + denotes relative level of strength. Strattice Laparoscopic Phasix ST Bio - A SurgiMend Phasix Strattice XenMatrix SurgiMend Strattice XenMatrix SurgiMend Strattice XenMatrix Phasix ST
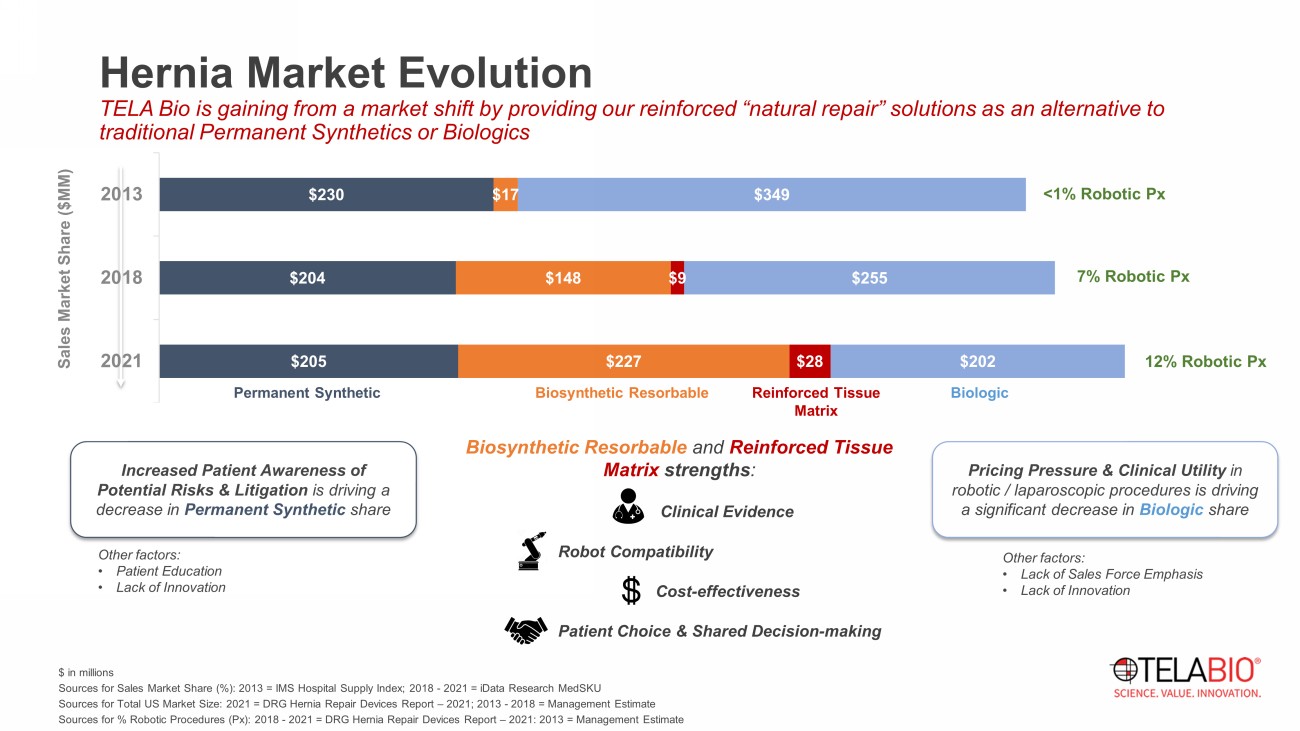
Permanent Synthetic Biologic Biosynthetic Resorbable Reinforced Tissue Matrix Other factors: • Lack of Sales Force Emphasis • Lack of Innovation Other factors: • Patient Education • Lack of Innovation Pricing Pressure & Clinical Utility in robotic / laparoscopic procedures is driving a significant decrease in Biologic share Increased Patient Awareness of Potential Risks & Litigation is driving a decrease in Permanent Synthetic share $ in millions Sources for Sales Market Share (%): 2013 = IMS Hospital Supply Index; 2018 - 2021 = iData Research MedSKU Sources for Total US Market Size: 2021 = DRG Hernia Repair Devices Report – 2021; 2013 - 2018 = Management Estimate Sources for % Robotic Procedures ( Px ): 2018 - 2021 = DRG Hernia Repair Devices Report – 2021: 2013 = Management Estimate $205 $204 $230 $227 $148 $17 $28 $9 $202 $255 $349 2021 2018 2013 Sales Market Share ($MM) Biosynthetic Resorbable and Reinforced Tissue Matrix strengths : Clinical Evidence Robot Compatibility Cost - effectiveness Patient Choice & Shared Decision - making <1% Robotic Px 7% Robotic Px 12% Robotic Px Hernia Market Evolution TELA Bio is gaining from a market shift by providing our reinforced “natural repair” solutions as an alternative to traditional Permanent Synthetics or Biologics

Lawsuits against permanent synthetic meshes estimated to be assembled across the U.S. 3 of surgeons agree that use of permanent synthetic mesh puts patients at long - term risk of complications 1 1. Hernia and Abdominal Surgeries Survey (Oct 2020). A group of 71 surgeons were surveyed regarding use of mesh in various hernia repai r surgeries. 2. Figures derived from Company - sponsored poll of approximately 1,100 potential patients for hernia procedures. 3. www.drugwatch.com (September 2021) 59% Hernia patients want proactive control in their care 2 3 of 4 ~30K Need for Alternative to Permanent Synthetic Mesh
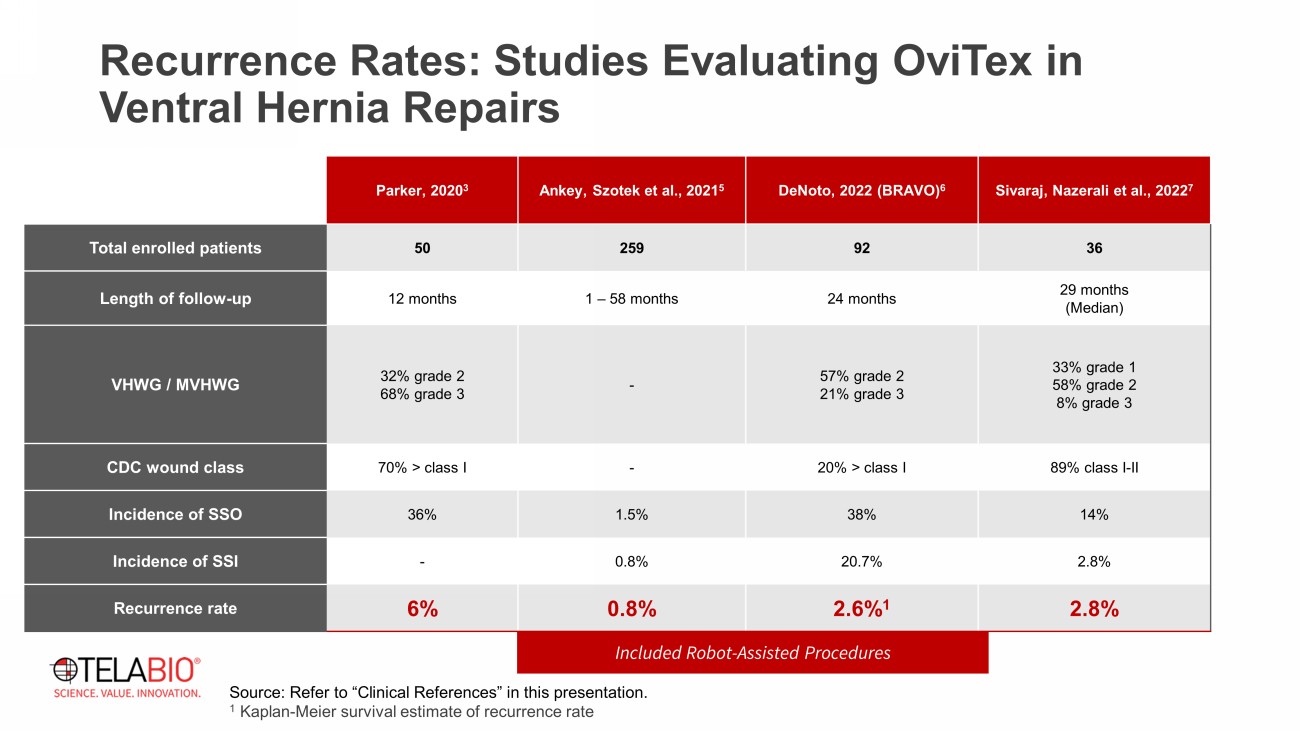
Parker, 2020 3 Ankey , Szotek et al., 2021 5 DeNoto, 2022 (BRAVO) 6 Sivaraj , Nazerali et al., 2022 7 Total enrolled patients 50 259 92 36 Length of follow - up 12 months 1 – 58 months 24 months 29 months (Median) VHWG / MVHWG 32% grade 2 68% grade 3 - 57% grade 2 21% grade 3 33% grade 1 58% grade 2 8% grade 3 CDC wound class 70% > class I - 20% > class I 89% class I - II Incidence of SSO 36% 1.5% 38% 14% Incidence of SSI - 0.8% 20.7% 2.8% Recurrence rate 6% 0.8% 2.6% 1 2.8% Included Robot - Assisted Procedures Source: Refer to “Clinical References” in this presentation. 1 Kaplan - Meier survival estimate of recurrence rate Recurrence Rates: Studies Evaluating OviTex in Ventral Hernia Repairs
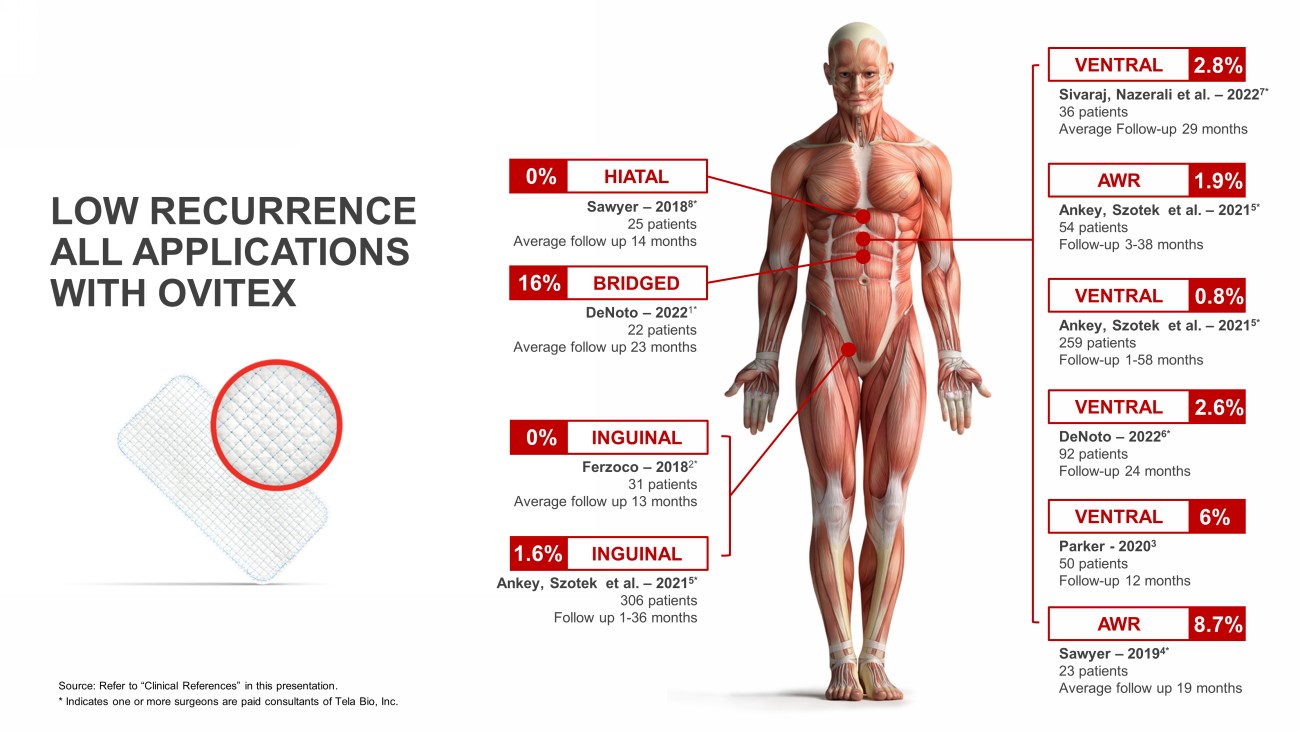
LOW RECURRENCE ALL APPLICATIONS WITH OVITEX Ankey , Szotek et al. – 2021 5* 54 patients Follow - up 3 - 38 months Ferzoco – 2018 2* 31 patients Average follow up 13 months INGUINAL 0% Ankey , Szotek et al. – 2021 5* 306 patients Follow up 1 - 36 months INGUINAL 1.6% Sawyer – 2018 8* 25 patients Average follow up 14 months HIATAL 0% DeNoto – 2022 1* 22 patients Average follow up 23 months BRIDGED 16% AWR 1.9% Parker - 2020 3 50 patients Follow - up 12 months VENTRAL 6% Sawyer – 2019 4* 23 patients Average follow up 19 months AWR 8.7% DeNoto – 2022 6* 92 patients Follow - up 24 months VENTRAL 2.6% Ankey , Szotek et al. – 2021 5* 259 patients Follow - up 1 - 58 months VENTRAL 0.8% Sivaraj , Nazerali et al. – 2022 7* 36 patients Average Follow - up 29 months VENTRAL 2.8% Source: Refer to “Clinical References” in this presentation. * Indicates one or more surgeons are paid consultants of Tela Bio, Inc.

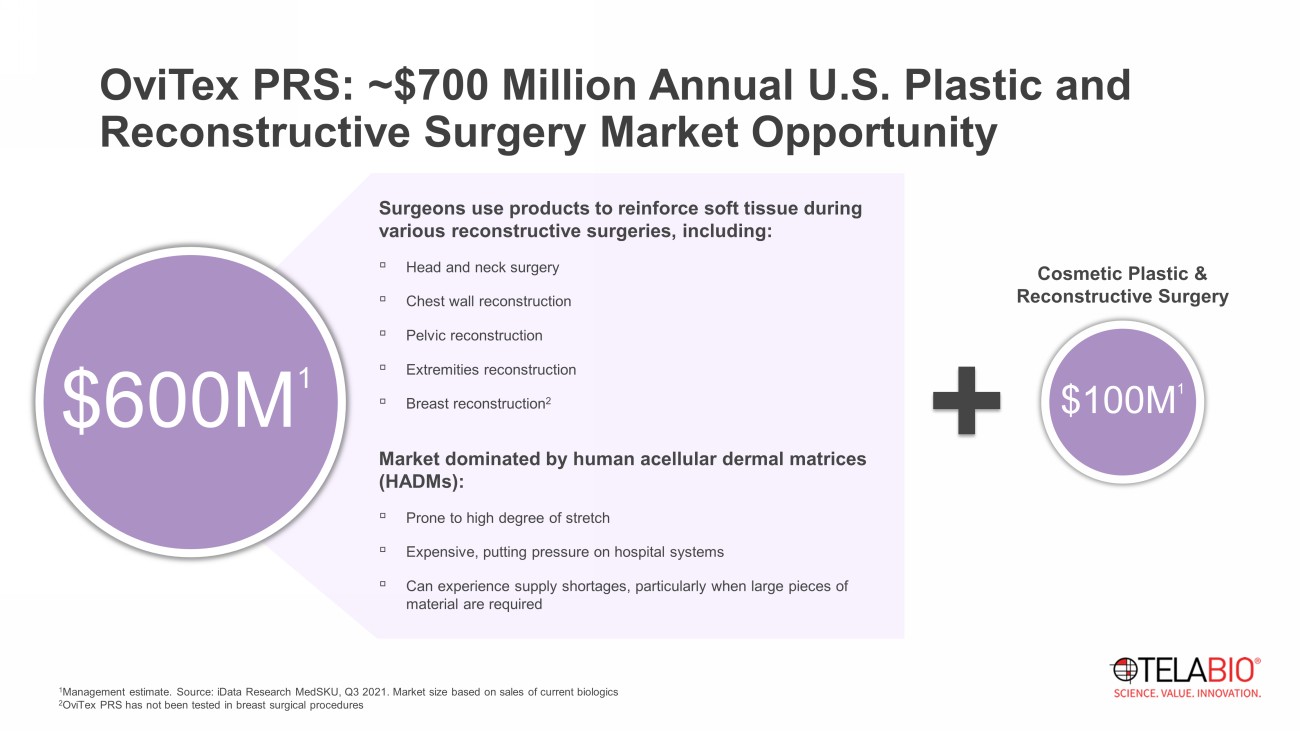
OviTex PRS: ~$700 Million Annual U.S. Plastic and Reconstructive Surgery Market Opportunity Surgeons use products to reinforce soft tissue during various reconstructive surgeries, including: ▫ Head and neck surgery ▫ Chest wall reconstruction ▫ Pelvic reconstruction ▫ Extremities reconstruction ▫ Breast reconstruction 2 1 Management estimate. Source: iData Research MedSKU , Q3 2021. Market size based on sales of current biologics 2 OviTex PRS has not been tested in breast surgical procedures Cosmetic Plastic & Reconstructive Surgery $600M 1 $100M 1 Market dominated by human acellular dermal matrices (HADMs): ▫ Prone to high degree of stretch ▫ Expensive, putting pressure on hospital systems ▫ Can experience supply shortages, particularly when large pieces of material are required
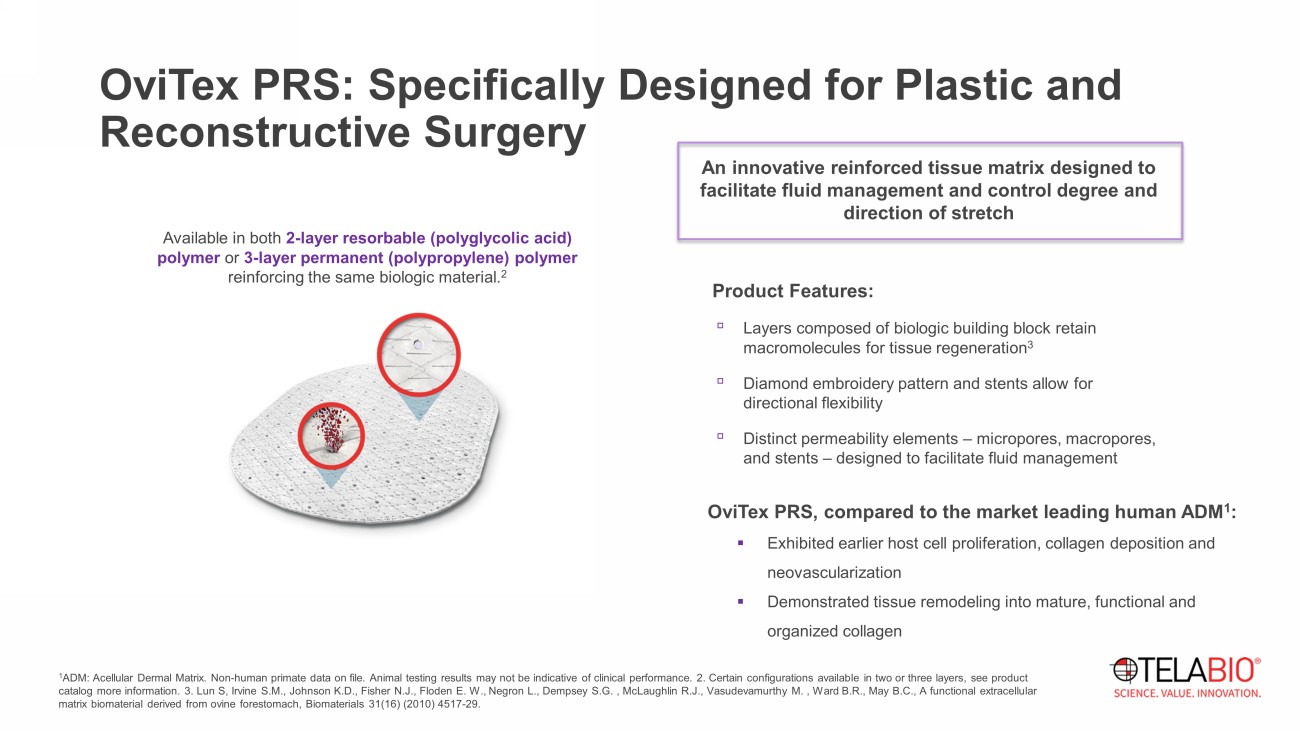
OviTex PRS: Specifically Designed for Plastic and Reconstructive Surgery An innovative reinforced tissue matrix designed to facilitate fluid management and control degree and direction of stretch Product Features: ▫ Layers composed of biologic building block retain macromolecules for tissue regeneration 3 ▫ Diamond embroidery pattern and stents allow for directional flexibility ▫ Distinct permeability elements – micropores , macropores, and stents – designed to facilitate fluid management Available in both 2 - layer resorbable (polyglycolic acid) polymer or 3 - layer permanent (polypropylene) polymer reinforcing the same biologic material. 2 1 ADM: Acellular Dermal Matrix. Non - human primate data on file. Animal testing results may not be indicative of clinical performan ce. 2. Certain configurations available in two or three layers, see product catalog more information. 3. Lun S, Irvine S.M., Johnson K.D., Fisher N.J., Floden E. W., Negron L., Dempsey S.G. , McLaughlin R.J., Vasudevamurthy M. , Ward B.R., May B.C., A functional extracellular matrix biomaterial derived from ovine forestomach, Biomaterials 31(16) (2010) 4517 - 29. OviTex PRS, compared to the market leading human ADM 1 : ▪ Exhibited earlier host cell proliferation, collagen deposition and neovascularization ▪ Demonstrated tissue remodeling into mature, functional and organized collagen
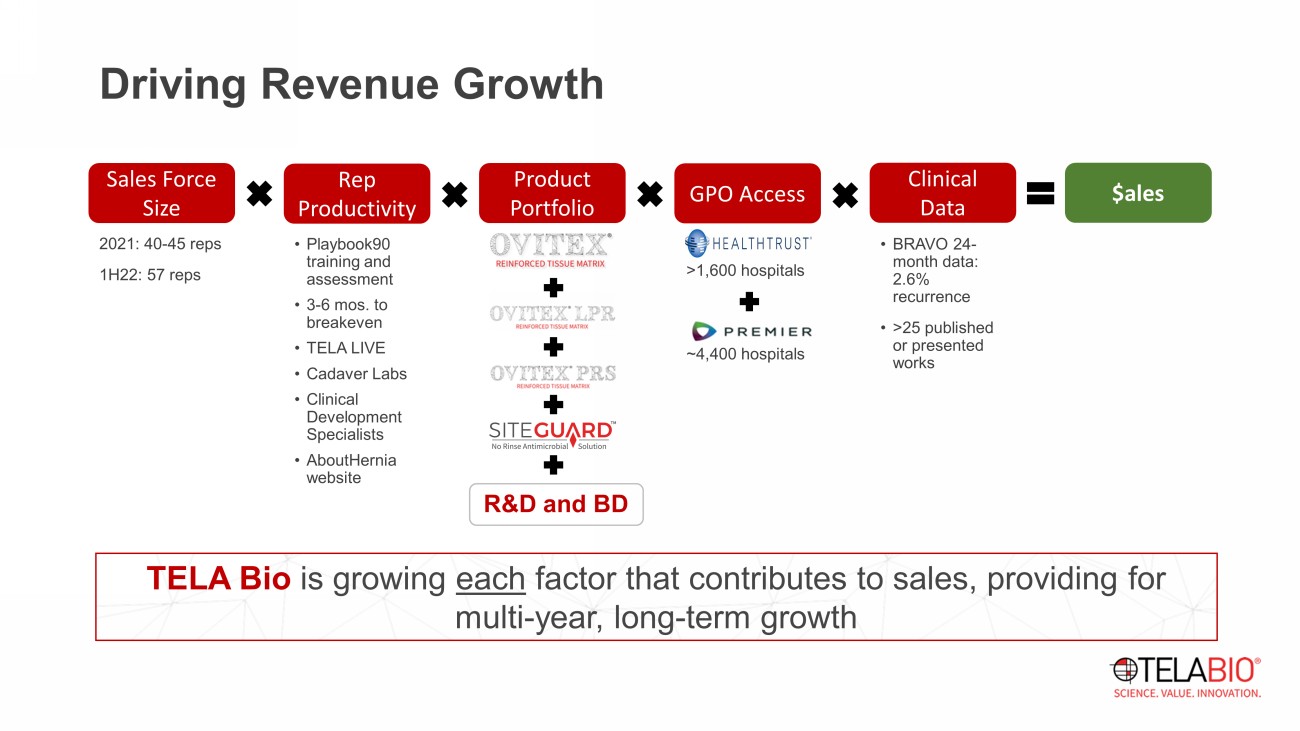
TELA Bio is growing each factor that contributes to sales, providing for multi - year, long - term growth 2021: 40 - 45 reps 1H22: 57 reps Driving Revenue Growth Rep Productivity Sales Force Size Product Portfolio GPO Access Clinical Data $ales • Playbook90 training and assessment • 3 - 6 mos. to breakeven • TELA LIVE • Cadaver Labs • Clinical Development Specialists • AboutHernia website • BRAVO 24 - month data: 2.6% recurrence • >25 published or presented works >1,600 hospitals ~4,400 hospitals R&D and BD
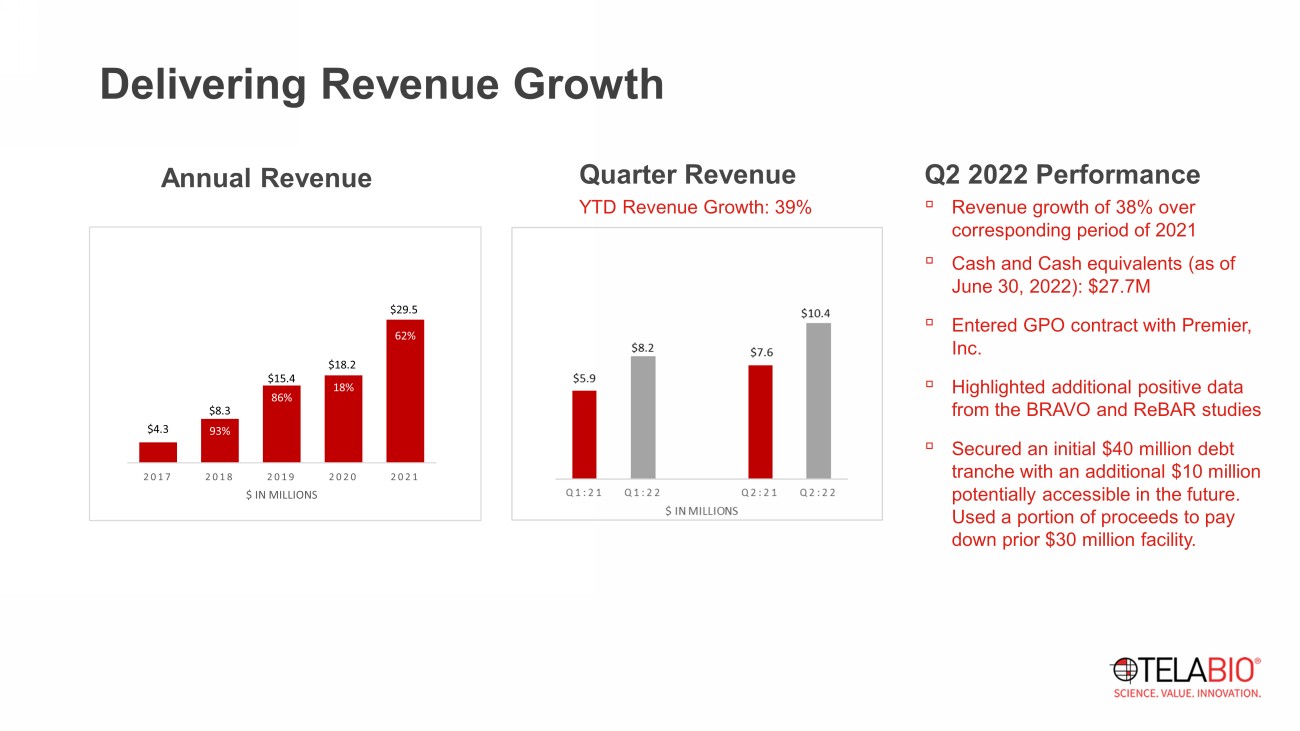
Delivering Revenue Growth Q2 2022 Performance ▫ Revenue growth of 38 % over corresponding period of 2021 ▫ Cash and Cash equivalents (as of June 30, 2022): $ 27.7 M ▫ Entered GPO contract with Premier, Inc. ▫ Highlighted additional positive data from the BRAVO and ReBAR studies ▫ Secured an initial $40 million debt tranche with an additional $10 million potentially accessible in the future. Used a portion of proceeds to pay down prior $30 million facility. Annual Revenue Quarter Revenue YTD Revenue Growth: 39% $4.3 $8.3 $15.4 $18.2 $29.5 93% 86% 18% 62% 2017 2018 2019 2020 2021 $ IN MILLIONS

Investment Rationale Advanced reinforced tissue matrix portfolio supported by compelling clinical evidence Focused on ~$2.2 billion annual U.S. total addressable markets – still in early stages of growth Driving commercial adoption with targeted direct - sales approach Recent product launches in growing markets: robotic hernia surgery + plastic and reconstructive surgery Broad intellectual property portfolio Established DRG - based reimbursement pathway for hernia repair and robust GPO access Industry leading executive team with proven track record

CLINICAL REFERENCES 1. DeNoto G 3rd. Bridged repair of large ventral hernia defects using an ovine reinforced biologic: A case series. Ann Med Surg ( Lond ). 2022 Mar 2;75:103446. doi : 10.1016/j.amsu.2022.103446. PMID: 35386793; PMCID: PMC8977941. 2. Ferzoco S.J. Early experience outcome of a reinforced Bioscaffold in inguinal hernia repair: A case series. Int. J. Surg. Open. 2018;12:9 – 11. doi : 10.1016/j.ijso.2018.06.001. 3. Parker MJ, Kim RC, Barrio M, Socas J, Reed LR, Nakeeb A, House MG, Ceppa EP (2021) A novel biosynthetic scaffold mesh reinforcement affords the lowest hernia recurrence in the highest - risk patients. Surg Endosc . 2021 Sep;35(9):5173 - 5178. doi : 10.1007/s00464 - 020 - 08009 - 1. Epub 2020 Sep 24. PMID: 32970208. 4. Sawyer M.A.J. (2019) Ovine Polymer - Reinforced BioScaffold in Abdominal Wall Reconstruction, Poster presented at: American Hernia Society (AHS) Annual Meeting 2019, Las Vegas, NV 5. Ankney C, Banaschak C, Sowers B, Szotek P (2021) Minimizing Retained Foreign Body in Hernia Repair Using a Novel Technique: Reinforced Biologic Augmented Repair ( ReBAR ). Maples. 2021 July 6. DeNoto G 3rd, Ceppa EP, Pacella SJ, Sawyer M, Slayden G, Takata M, Tuma G, Yunis J. 24 - Month results of the BRAVO Study: A prospective, multi - center study evaluating the clinical outcomes of ventral hernias treated with OviTex 1S Permanent Reinforced Tissue Matrix. Abstract to be presented at: 2022 American Hernia Society (AHS) Meeting, September 14 - 16, 2022, Charlotte, NC. 7. Sivaraj , D, Henn, D, Fischer, KS, Trudy S. Kim, TS, Black, CK, Lin, JQ, Barrera, JA, Leeolou , MC, Makarewicz , NS, Chen, K, Perrault, DP, Gurtner , GC, Lee, GK, Nazerali , R Reinforced Biologic Mesh Reduces Postoperative Complications Compared to Biologic Mesh after Ventral Hernia Repair: Plast Reconstr Surg Glob Open 2022;10:e4083; doi : 10.1097/GOX.0000000000004083 8. Sawyer M.A.J. (2018) New Ovine Polymer - Reinforced Bioscaffold in Hiatal Hernia Repair. JSLS J. Soc. Laparoendosc . Surg. 2018;22 doi : 10.4293/JSLS.2018.00057.
Serious News for Serious Traders! Try StreetInsider.com Premium Free!
You May Also Be Interested In
- AVXL INVESTOR DEADLINE: Robbins Geller Rudman & Dowd LLP Announces that Anavex Life Sciences Corporation Investors with Substantial Losses Have Opportunity to Lead Case
- Aurania Announces Increase in Size of Debt Settlement to C$2.07 Million
- Ensurge Micropower ASA - Grant of Incentive Subscription Rights
Create E-mail Alert Related Categories
SEC FilingsSign up for StreetInsider Free!
Receive full access to all new and archived articles, unlimited portfolio tracking, e-mail alerts, custom newswires and RSS feeds - and more!



 Tweet
Tweet Share
Share