Form 8-K TCR2 THERAPEUTICS INC. For: Sep 17
Exhibit 99.1

TCR2 Therapeutics Announces Positive Interim Results from Ongoing Phase 1/2 Trial of Gavo-cel for Treatment Refractory Mesothelin-Expressing Solid Tumors
- Clinical activity observed in all three mesothelin-expressing tumor types treated
- Gavo-cel disease control rate (DCR) 81% with tumor regression in 15 of 16 evaluable patients
- Overall response rate (ORR) 31% in patients infused with gavo-cel following lymphodepletion
- Meaningful survival benefit at 11.2 months for patients with refractory mesothelioma
- Recommended Phase 2 Dose (RP2D) being refined after identification of the Maximum Tolerated Dose (MTD)
- TCR2 to host a conference call on Friday, September 17 at 9:00a.m. ET
CAMBRIDGE, Mass., Sept. 17, 2021 - TCR2 Therapeutics Inc. (Nasdaq: TCRR), a clinical-stage cell therapy company with a pipeline of novel T cell therapies for patients suffering from cancer, today announced positive interim results from the ongoing Phase 1 portion of the gavo-cel Phase 1/2 clinical trial for mesothelin-expressing solid tumors. A dataset will also be featured in an oral presentation at the European Society for Medical Oncology (ESMO) Congress 2021 on September 17 at 14:20 CEST (8:20am EST) (Presentation #959O) and is part of the official ESMO Press Programme.
As of the June 30, 2021 data cutoff, 17 patients (12 mesothelioma, 4 ovarian cancer and 1 cholangiocarcinoma) had received a single gavo-cel infusion in the dose escalation portion of the gavo-cel Phase 1 clinical trial. The median number of prior lines of therapy was 5, including immune checkpoint inhibitors (n=11) and mesothelin-directed therapies (n=5). Gavo-cel was administered up to dose level 5 (DL5) (5x108/m2 following lymphodepletion). Two dose limiting toxicities were reported: one Grade 3 pneumonitis at DL1 that resolved with supportive measures, which permitted the continuation of dose escalation, and one Grade 5 bronchoalveolar hemorrhage at DL5, which along with the development of severe CRS in all 3 patients treated at this dose level, led the Safety Review Team to declare 5x108/m2 as the MTD. Following identification of the MTD, one patient has received gavo-cel at 3x108/m2 after lymphodepletion using a split dosing approach to refine the RP2D and an additional patient has been treated at DL3 (1x108/m2 following lymphodepletion). In both cases gavo-cel was well tolerated with only Grade 1 non-hematological toxicities being reported.
15 of the 16 patients evaluable for efficacy experienced regression of their target lesions, ranging in magnitude from 5% to 75%. Six patients achieved partial responses (PRs) by target lesion assessment, four of whom (3 with mesothelioma and 1 with ovarian cancer) achieved a PR according to RECIST 1.1 criteria, including one who also achieved a complete metabolic response. One patient with cholangiocarcinoma was also considered to have achieved a PR by investigator assessment, for an ORR of 31%. By independent review assessment, the ORR was 25% with a DCR Rate of 81%. The median overall survival for patients with mesothelioma is 11.2 months, whereas the median progression free survival is 5.9 months.
“The interim gavo-cel data reported today showed continued clinical benefit and a manageable safety profile in a population of patients that previously achieved minimal or no improvement due to the advanced and aggressive state of their cancer,” said principal investigator David Hong, M.D., deputy chair of the Department of Investigational Cancer Therapeutics at The University of Texas MD Anderson Cancer Center. “Patients with treatment refractory cancer have very limited treatment options and will often need hospice and supportive care. We are encouraged by the early survival data from gavo-cel in patients previously treated with checkpoint inhibitors and other therapies.”
“Our ambition with gavo-cel from the start was to redefine treatment for solid tumors with cell therapies. We are excited to present data demonstrating clinical activity in all three mesothelin-expressing solid tumors treated to date and tumor regression in a majority of patients who are treatment refractory after numerous lines of therapy. We are very encouraged by the progression free survival and overall survival observed among patients with refractory mesothelioma treated so far with gavo-cel in the Phase 1 trial,” said Alfonso Quintás-Cardama, M.D., Chief Medical Officer of TCR2 Therapeutics. “Based on these data and the most recent patient experiencing a very mild safety profile at a cell dose of 3x108/m2, we believe the identification of the RP2D is close at hand. As we approach the Phase 2 expansion phase, our focus will shift to further optimizing outcomes for patients by studying combinations with immune checkpoint inhibitors, allowing gavo-cel re-treatment and evaluating different mesothelin expression thresholds.”
The primary objectives of the Phase 1 portion of the trial are to define the safety profile of gavo-cel in patients whose tumors overexpress mesothelin and to determine the RP2D. Secondary objectives include ORR and DCR. Exploratory objectives include the assessment of expansion, tumor infiltration, and persistence of gavo-cel.
Summary of trial conduct, baseline characteristics and gavo-cel dose:
| • | Screening: Forty-six percent of patients met the mesothelin expression cut-off as defined per protocol. |
| • | Patient Characteristics: 17 patients received gavo-cel including 12 with mesothelioma, 4 with ovarian cancer and 1 with cholangiocarcinoma with a median age of 57 years (range, 31-84 years). The median number of prior therapies was 5 (range, 1-9), including immune checkpoint inhibitor therapy (n=11) and anti-mesothelin therapies (n=5). |
| • | Gavo-cel Dose: The seventeen patients disclosed to date have received gavo-cel at the following dose level (DL): |
| • | DL 0: 5x107 cells/m2 without lymphodepletion – 1 mesothelioma patient |
| • | DL 1: 5x107 cells/m2 following lymphodepletion – 5 mesothelioma patients and 1 ovarian cancer patient |
| • | DL 2: 1x108 cells/m2 without lymphodepletion – 1 mesothelioma patient |
| • | DL 3: 1x108 cells/m2 following lymphodepletion – 1 mesothelioma patient, 1 cholangiocarcinoma patient, and 3 ovarian cancer patients |
| • | DL 4: 5x108 cells/m2 without lymphodepletion – 1 mesothelioma patient |
| • | DL 5: 5x108 cells/m2 following lymphodepletion – 3 mesothelioma patients |
Key clinical findings from patients treated with gavo-cel:
| • | Safety: gavo-cel was generally well tolerated with a manageable adverse event profile with no patients experiencing on-target, off-tumor toxicities. Two DLTs were observed: one case of Grade 3 pneumonitis at DL1 that resolved with anti-cytokine therapy, and one case of Grade 5 bronchoalveolar hemorrhage at DL5. Furthermore, all three patients treated at DL5 experienced Grade ³3 CRS which resulted in 5x108 cells/m2 following lymphodepletion being declared the MTD. |
| • | Clinical Activity: 16 patients were evaluable for response. Tumor regression was observed in 15 (94%) patients with a DCR of 81%. Six patients achieved partial responses (PRs) by target lesion assessment, four of whom (3 with mesothelioma and 1 with ovarian cancer) achieved a PR according to RECIST 1.1 criteria. The ORR by RECISTv1.1 criteria among patients infused with gavo-cel following lymphodepletion chemotherapy was 31% by independent review assessment, including one patient who achieved a complete metabolic response, and 38% by investigator assessment, which included a PR in a patient with metastatic cholangiocarcinoma. |
| • | Translational Data: Peak gavo-cel expansion (Cmax) increased when gavo-cel was administered following lymphodepletion in a dose dependent fashion. Cytokine elevations post-gavo-cel infusion were observed in all evaluable patients, which is indicative of mesothelin target engagement. |
About the Phase 1/2 Clinical Trial in Advanced Mesothelin-Expressing Solid Tumors
The Phase 1/2 clinical trial (NCT03907852) is evaluating the safety and efficacy of gavocabtagene autoleucel (“gavo-cel”; TC-210), TCR2’s T cell receptor fusion construct directed against mesothelin. The trial is enrolling patients with either mesothelin expressing non-small cell lung cancer (NSCLC), ovarian cancer, cholangiocarcinoma, or malignant pleural/peritoneal mesothelioma. The Phase 1 dose escalation portion of the clinical trial utilizes a modified 3+3 design with four increasing gavo-cel doses. At each dose, gavo-cel will be tested in two separate dose levels: first without lymphodepletion and then following lymphodepleting chemotherapy. The Phase 1 portion of the clinical trial is ongoing.
About Mesothelin-Expressing Solid Tumors
Mesothelin is a cell-surface glycoprotein highly expressed in a wide range of solid tumors, including malignant pleural/peritoneal mesothelioma, ovarian cancer, cholangiocarcinoma, breast cancer, pancreatic cancer and others. Overexpression of mesothelin is associated with poorer prognosis in some cancers due to its active role in both malignant transformation and tumor aggressiveness by promoting cancer cell proliferation, invasion, and metastasis. Of the wide range of solid tumors expressing mesothelin, non-small cell lung cancer, ovarian cancer, mesothelioma and cholangiocarcinoma represent a patient population up to 80,000 annually in the United States alone.
TCR2 Therapeutics Conference Call and Webcast
TCR2 Therapeutics will host a conference call and webcast on Friday, September 17 at 9:00am E.T. In order to participate in the conference call, please dial 866-220-8062 (domestic) or 470-495-9169 (international) and refer to confirmation number 1597681. The webcast and presentation will be made available on the TCR2 Therapeutics website in the Investors section under Events at investors.tcr2.com/events. Following the live audio webcast, a replay will be available on the Company’s website for approximately 30 days.
About TCR2 Therapeutics
TCR2 Therapeutics Inc. is a clinical-stage cell therapy company developing a pipeline of novel T cell therapies for patients suffering from cancer. TCR2’s proprietary T cell receptor (TCR) Fusion Construct T cells (TRuC®-T cells) specifically recognize and kill cancer cells by harnessing signaling from the entire TCR, independent of human leukocyte antigens (HLA). In preclinical studies, TRuC-T cells have demonstrated superior anti-tumor activity compared to chimeric antigen receptor T cells (CAR-T cells), while secreting lower levels of cytokine release. The Company’s lead TRuC-T cell product candidate targeting solid tumors, gavo-cel, is currently being studied in a Phase 1/2 clinical trial to treat patients with mesothelin-positive non-small cell lung cancer (NSCLC), ovarian cancer, malignant pleural/peritoneal mesothelioma, and cholangiocarcinoma. The Company’s lead TRuC-T cell product candidate targeting hematological malignancies, TC-110, is currently being studied in a Phase 1/2 clinical trial to treat patients with CD19-positive adult acute lymphoblastic leukemia (aALL) and with aggressive or indolent non-Hodgkin lymphoma (NHL). For more information about TCR2, please visit www.tcr2.com.
Forward-looking Statements
This press release contains forward-looking statements and information within the meaning of the Private Securities Litigation Reform Act of 1995 and other federal securities laws. The use of words such as “may,” “will,” “could”, “should,” “expects,” “intends,” “plans,” “anticipates,” “believes,” “estimates,” “predicts,” “projects,” “seeks,” “endeavor,” “potential,” “continue” or the negative of such words or other similar expressions can be used to identify forward-looking statements. These forward-looking statements include, but are not limited to, express or implied statements regarding TCR2’s expectations for the Phase 1/2 clinical trials of gavo-cel and TC-110; TCR2’s expectations for the safety and efficacy of its product candidates and enhancements, including gavo-cel and TC-110, compared to current T-cell therapy approaches; TCR2’s expectations regarding the timing of determining an RP2D for gavo-cel and TCR2’s expectations regarding the estimated patient populations and related market opportunities in gavo-cel’s and TC-110’s targeted indications.
The expressed or implied forward-looking statements included in this press release are only predictions and are subject to a number of risks, uncertainties and assumptions, including, without limitation: uncertainties inherent in clinical studies and in the availability and timing of data from ongoing clinical studies; whether interim results from a clinical trial will be predictive of the final results of a trial; the possibility that positive results from preclinical studies and correlative studies may not necessarily be predictive of the results of TCR2’s planned clinical trials, including the Phase 1/2 clinical trials of gavo-cel and TC-110; the risk that the results from the Phase 1/2 clinical trials of gavo-cel and TC-110 will not support further development and marketing approval; the risk that TCR2may be unable to gain approval of gavo-cel, TC-110 and its other product candidates on a timely basis, if at all; the risk that TCR2 has over-estimated the potential patient population for its product candidates, if approved; the risk that the current COVID-19 pandemic will impact TCR2’s clinical trials and other operations; and other risks set forth under the caption “Risk Factors” in TCR2’s most recent Annual Report on Form 10-K, most recent Quarterly Report on Form 10-Q and its other filings with the Securities and Exchange Commission. In light of these risks, uncertainties and assumptions, the forward-looking events and circumstances discussed in this press release may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements. You should not rely upon forward-looking statements as predictions of future events. Although TCR2 believes that the expectations reflected in the forward-looking statements are reasonable, it cannot guarantee that the future results, levels of activity, performance or events and circumstances reflected in the forward-looking statements will be achieved or occur.
Moreover, except as required by law, neither TCR2 nor any other person assumes responsibility for the accuracy and completeness of the forward-looking statements included in this press release. Any forward-looking statement included in this press release speaks only as of the date on which it was made. We undertake no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required by law.
Investor and Media Contact:
Carl Mauch
Director, Investor Relations and Corporate Communications
TCR2 Therapeutics Inc.
(617) 949-5667

gavo-cel Phase 1/2 Clinical Update September 2021 Exhibit 99.2

Forward Looking Statements This presentation has been prepared by TCR2 Therapeutics Inc. (“we,” “us,” or “our”) and contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and other federal securities laws. Forward-looking statements are neither historical facts nor assurances of future performance. Instead, they are based on our current beliefs, expectations and assumptions regarding the future of our business, future plans and strategies, our development plans, our clinical results and other future conditions. All statements, other than statements of historical facts, contained in this presentation, including express or implied statements regarding our expectations for the Phase 1/2 clinical trials of gavo-cel and TC-110, our expectations for the safety and efficacy of our product candidates and enhancements, including gavo-cel and TC-110, compared to current T-cell therapy approaches, our expectations regarding the timing of determining an RP2D for gavo-cel, our expectations regarding the estimated patient populations and related market opportunities in gavo-cel’s and TC-110’s targeted indications, and our expectations regarding manufacturing of our product candidates are forward-looking statements. These statements are based on management’s current expectations and beliefs and are forward-looking statements which involve risks and uncertainties that could cause actual results to differ materially from those discussed in such forward-looking statements. Such risks and uncertainties include, among others: uncertainties inherent in clinical studies and in the availability and timing of data from ongoing clinical studies; whether interim results from a clinical trial will be predictive of the final results of a trial; the possibility that positive results from preclinical studies and correlative studies may not necessarily be predictive of the results of our planned clinical trials, including the Phase 1/2 clinical trials of gavo-cel and TC-110; the risk that the results from the Phase 1/2 clinical trials of gavo-cel and TC-110 will not support further development and marketing approval; the risk that we may be unable to gain approval of gavo-cel, TC-110 and our other product candidates on a timely basis, if at all; the risk that we have over-estimated the potential patient population for our product candidates, if approved; the risk that the current COVID-19 pandemic will impact our clinical trials and other operations; and the other risks set forth under the caption “Risk Factors” in our most recent Annual Report on Form 10-K for the year ended December 31, 2020, as filed with the SEC on March 16, 2021, and in our future filings with the SEC available at the SEC’s website at www.sec.gov. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. You should not place undue reliance on any forward‐looking statements, which speak only as of the date they are made. While we may elect to update these forward-looking statements at some point in the future, we assume no obligation to update or revise any forward-looking statements except to the extent required by applicable law. Although we believe the expectations reflected in such forward-looking statements are reasonable, we can give no assurance that such expectations will prove to be correct. Accordingly, readers are cautioned not to place undue reliance on these forward-looking statements. No representations or warranties (expressed or implied) are made about the accuracy of any such forward-looking statements.

Introduction Garry Menzel, PhD Chief Executive Officer
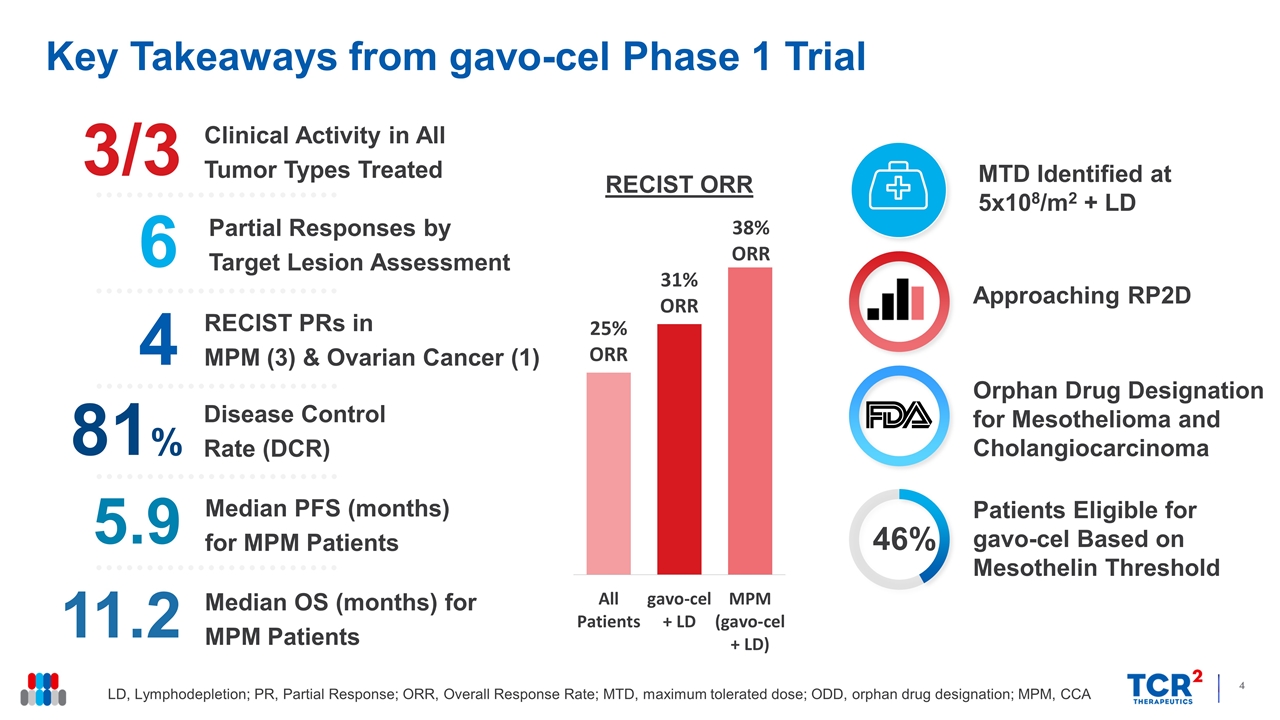
Key Takeaways from gavo-cel Phase 1 Trial LD, Lymphodepletion; PR, Partial Response; ORR, Overall Response Rate; MTD, maximum tolerated dose; ODD, orphan drug designation; MPM, CCA Approaching RP2D Orphan Drug Designation for Mesothelioma and Cholangiocarcinoma Median OS (months) for MPM Patients 11.2 Disease Control Rate (DCR) 81% Clinical Activity in All Tumor Types Treated 3/3 RECIST PRs in MPM (3) & Ovarian Cancer (1) 6 Partial Responses by Target Lesion Assessment 4 Median PFS (months) for MPM Patients 5.9 MTD Identified at 5x108/m2 + LD 46% Patients Eligible for gavo-cel Based on Mesothelin Threshold RECIST ORR

gavo-cel Clinical Trial Review Alfonso Quintás-Cardama, MD Chief Medical Officer
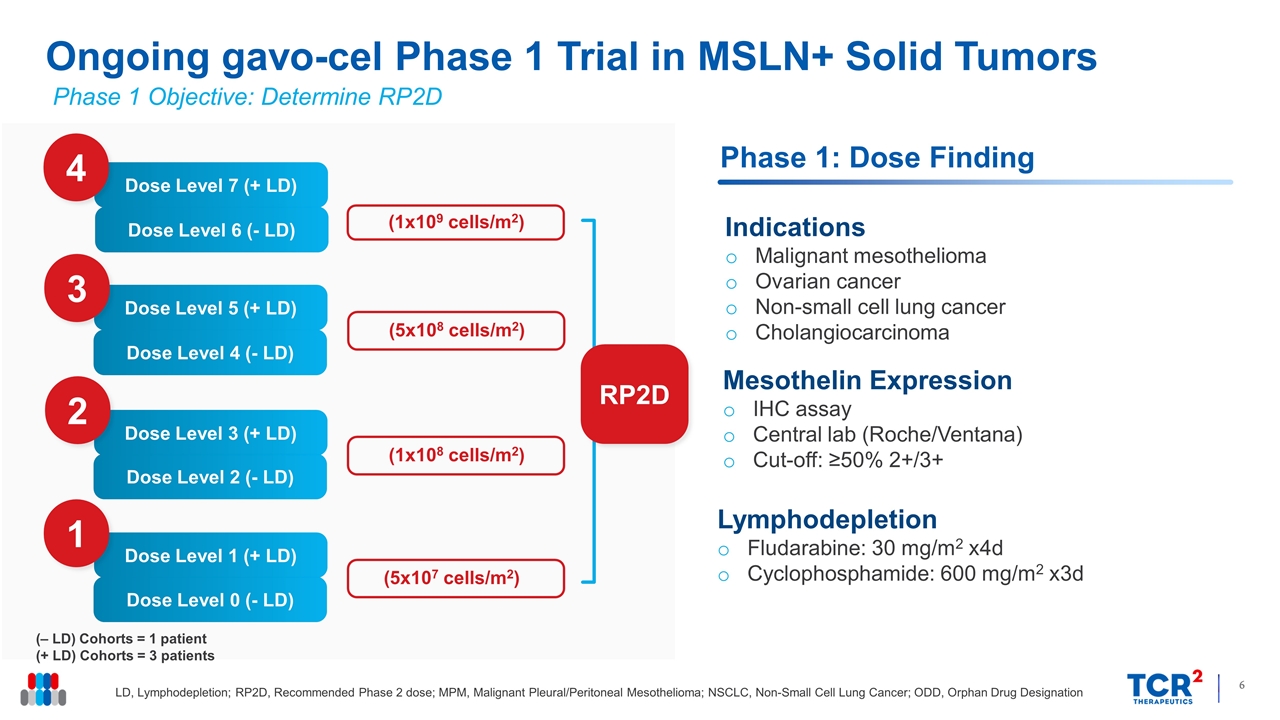
Ongoing gavo-cel Phase 1 Trial in MSLN+ Solid Tumors LD, Lymphodepletion; RP2D, Recommended Phase 2 dose; MPM, Malignant Pleural/Peritoneal Mesothelioma; NSCLC, Non-Small Cell Lung Cancer; ODD, Orphan Drug Designation RP2D Phase 1: Dose Finding (‒ LD) Cohorts = 1 patient (+ LD) Cohorts = 3 patients Dose Level 1 (+ LD) Dose Level 0 (- LD) 1 Dose Level 3 (+ LD) Dose Level 2 (- LD) 2 Dose Level 5 (+ LD) Dose Level 4 (- LD) 3 Dose Level 7 (+ LD) Dose Level 6 (- LD) 4 (5x107 cells/m2) (1x108 cells/m2) (5x108 cells/m2) (1x109 cells/m2) Lymphodepletion Fludarabine: 30 mg/m2 x4d Cyclophosphamide: 600 mg/m2 x3d Mesothelin Expression IHC assay Central lab (Roche/Ventana) Cut-off: ≥50% 2+/3+ Indications Malignant mesothelioma Ovarian cancer Non-small cell lung cancer Cholangiocarcinoma Phase 1 Objective: Determine RP2D
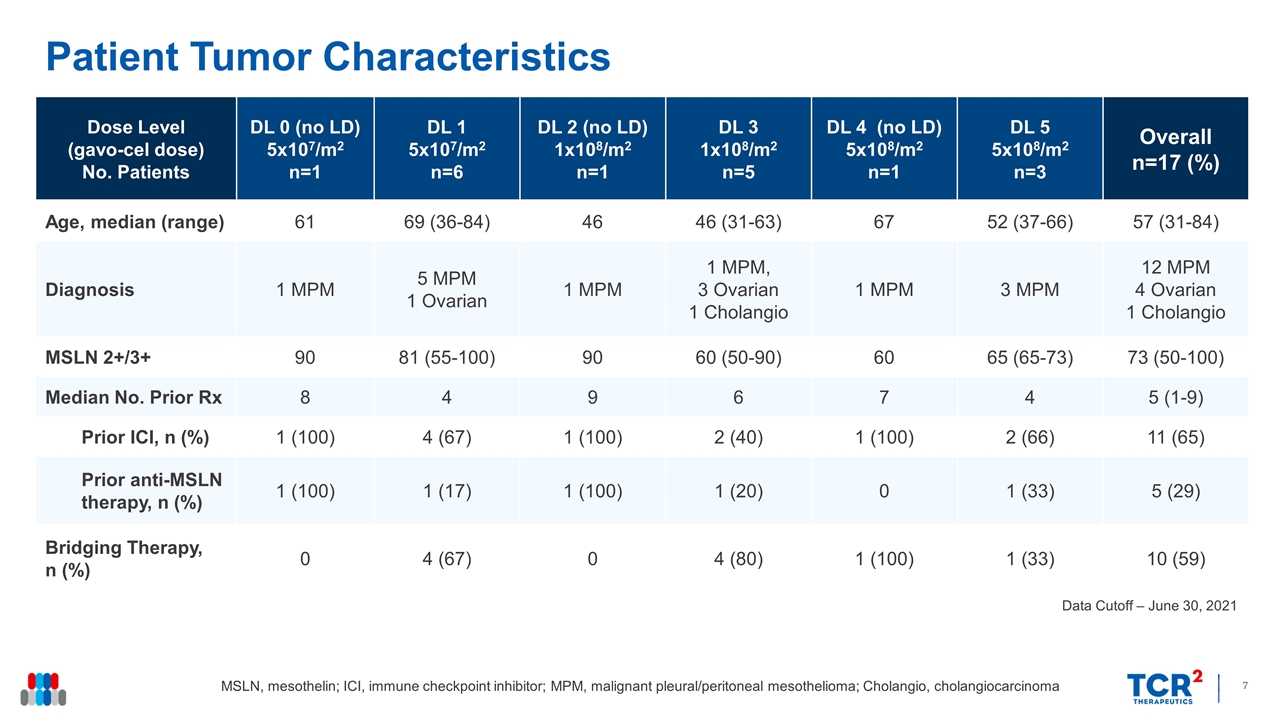
Patient Tumor Characteristics Data Cutoff – June 30, 2021 Dose Level (gavo-cel dose) No. Patients DL 0 (no LD) 5x107/m2 n=1 DL 1 5x107/m2 n=6 DL 2 (no LD) 1x108/m2 n=1 DL 3 1x108/m2 n=5 DL 4 (no LD) 5x108/m2 n=1 DL 5 5x108/m2 n=3 Overall n=17 (%) Age, median (range) 61 69 (36-84) 46 46 (31-63) 67 52 (37-66) 57 (31-84) Diagnosis 1 MPM 5 MPM 1 Ovarian 1 MPM 1 MPM, 3 Ovarian 1 Cholangio 1 MPM 3 MPM 12 MPM 4 Ovarian 1 Cholangio MSLN 2+/3+ 90 81 (55-100) 90 60 (50-90) 60 65 (65-73) 73 (50-100) Median No. Prior Rx 8 4 9 6 7 4 5 (1-9) Prior ICI, n (%) 1 (100) 4 (67) 1 (100) 2 (40) 1 (100) 2 (66) 11 (65) Prior anti-MSLN therapy, n (%) 1 (100) 1 (17) 1 (100) 1 (20) 0 1 (33) 5 (29) Bridging Therapy, n (%) 0 4 (67) 0 4 (80) 1 (100) 1 (33) 10 (59) MSLN, mesothelin; ICI, immune checkpoint inhibitor; MPM, malignant pleural/peritoneal mesothelioma; Cholangio, cholangiocarcinoma
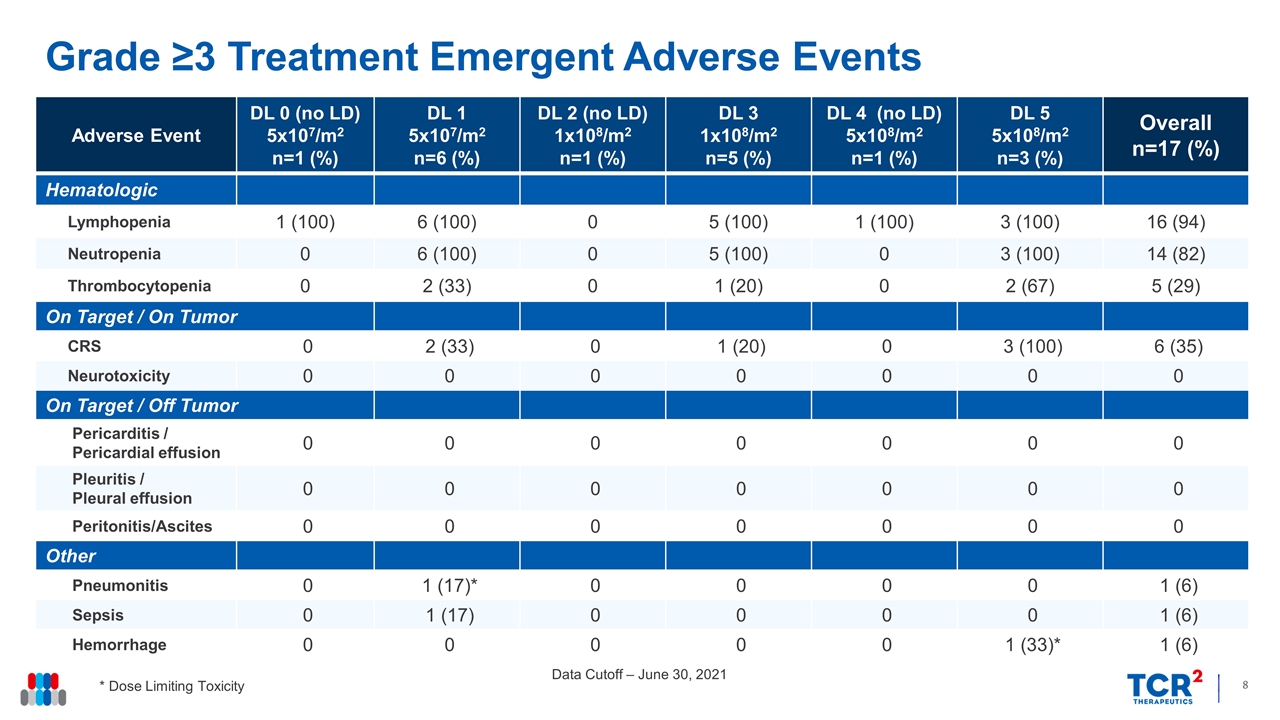
Grade ≥3 Treatment Emergent Adverse Events Data Cutoff – June 30, 2021 * Dose Limiting Toxicity Adverse Event DL 0 (no LD) 5x107/m2 n=1 (%) DL 1 5x107/m2 n=6 (%) DL 2 (no LD) 1x108/m2 n=1 (%) DL 3 1x108/m2 n=5 (%) DL 4 (no LD) 5x108/m2 n=1 (%) DL 5 5x108/m2 n=3 (%) Overall n=17 (%) Hematologic Lymphopenia 1 (100) 6 (100) 0 5 (100) 1 (100) 3 (100) 16 (94) Neutropenia 0 6 (100) 0 5 (100) 0 3 (100) 14 (82) Thrombocytopenia 0 2 (33) 0 1 (20) 0 2 (67) 5 (29) On Target / On Tumor CRS 0 2 (33) 0 1 (20) 0 3 (100) 6 (35) Neurotoxicity 0 0 0 0 0 0 0 On Target / Off Tumor Pericarditis / Pericardial effusion 0 0 0 0 0 0 0 Pleuritis / Pleural effusion 0 0 0 0 0 0 0 Peritonitis/Ascites 0 0 0 0 0 0 0 Other Pneumonitis 0 1 (17)* 0 0 0 0 1 (6) Sepsis 0 1 (17) 0 0 0 0 1 (6) Hemorrhage 0 0 0 0 0 1 (33)* 1 (6)
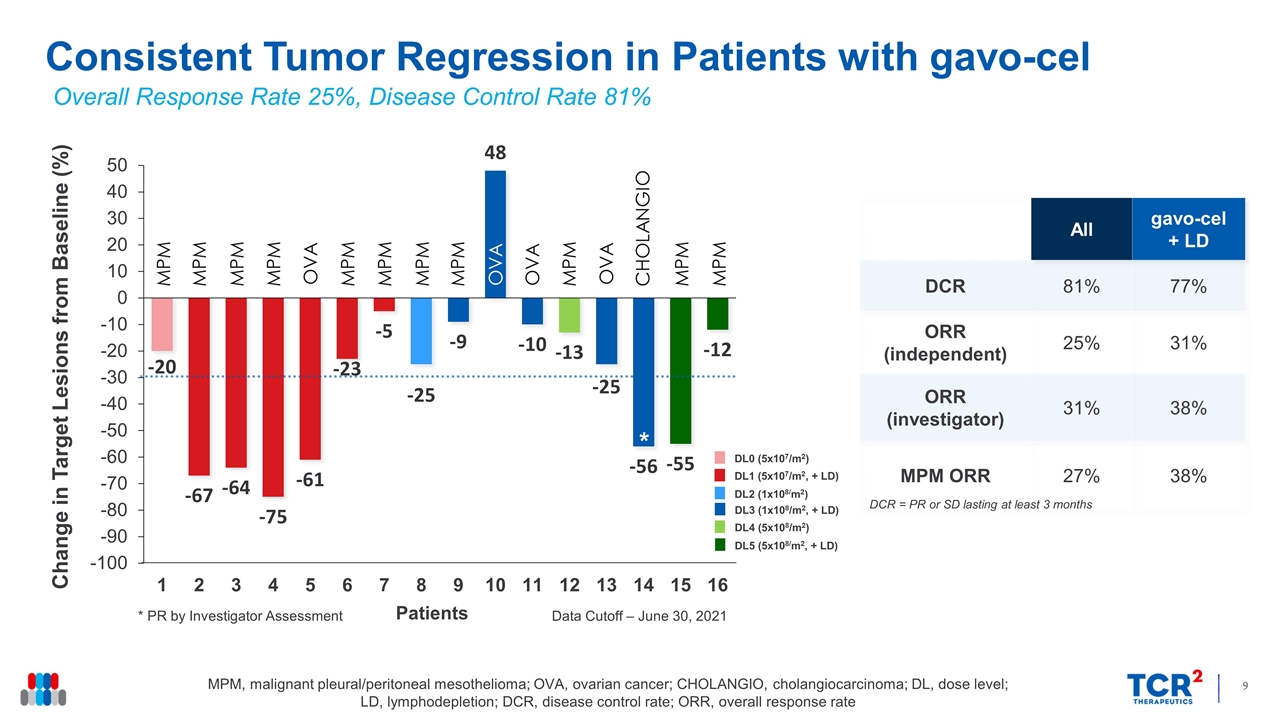
Consistent Tumor Regression in Patients with gavo-cel Change in Target Lesions from Baseline (%) MPM, malignant pleural/peritoneal mesothelioma; OVA, ovarian cancer; CHOLANGIO, cholangiocarcinoma; DL, dose level; LD, lymphodepletion; DCR, disease control rate; ORR, overall response rate All gavo-cel + LD DCR 81% 77% ORR (independent) 25% 31% ORR (investigator) 31% 38% MPM ORR 27% 38% Overall Response Rate 25%, Disease Control Rate 81% * * PR by Investigator Assessment Data Cutoff – June 30, 2021 DCR = PR or SD lasting at least 3 months DL0 (5x107/m2) DL1 (5x107/m2, + LD) DL2 (1x108/m2) DL3 (1x108/m2, + LD) DL4 (5x108/m2) DL5 (5x108/m2, + LD) * MPM MPM MPM MPM OVA MPM MPM MPM MPM OVA OVA MPM OVA CHOLANGIO MPM MPM Patients
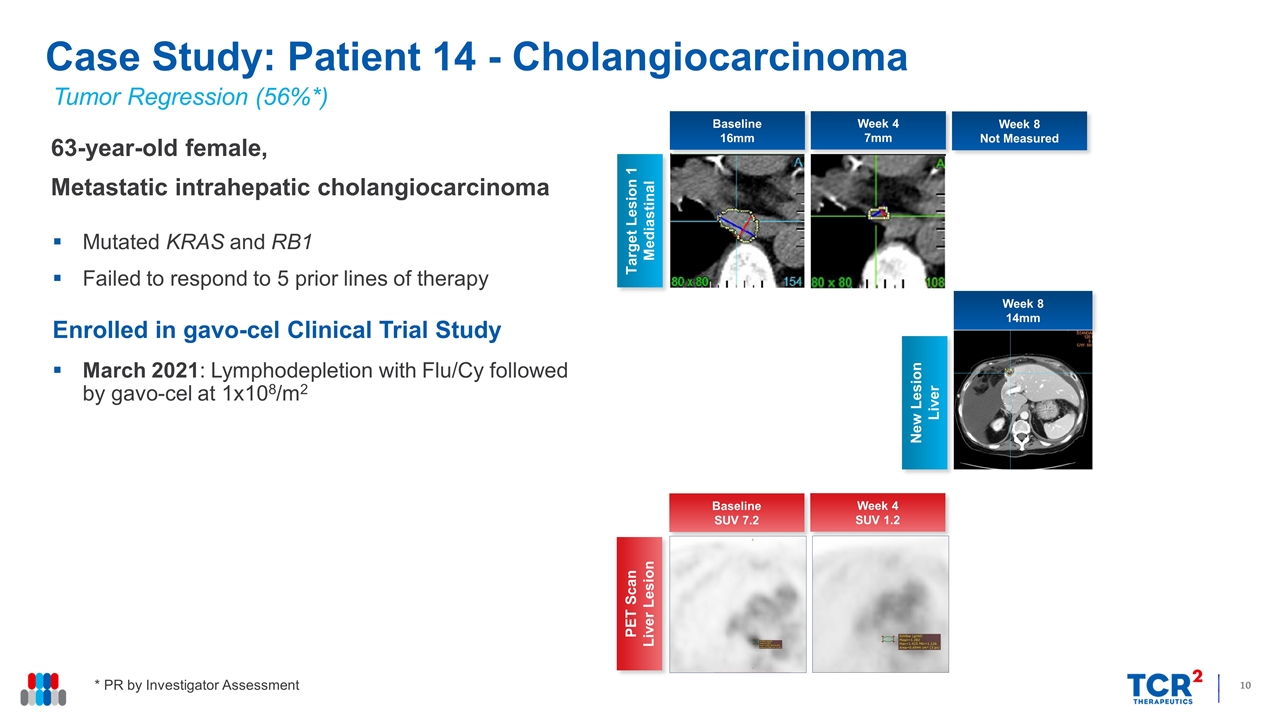
Tumor Regression (56%*) 63-year-old female, Metastatic intrahepatic cholangiocarcinoma Mutated KRAS and RB1 Failed to respond to 5 prior lines of therapy Enrolled in gavo-cel Clinical Trial Study March 2021: Lymphodepletion with Flu/Cy followed by gavo-cel at 1x108/m2 Case Study: Patient 14 - Cholangiocarcinoma Week 4 7mm Target Lesion 1 Mediastinal Week 8 Not Measured New Lesion Liver Baseline 16mm Week 8 14mm * PR by Investigator Assessment Week 4 SUV 1.2 PET Scan Liver Lesion Baseline SUV 7.2
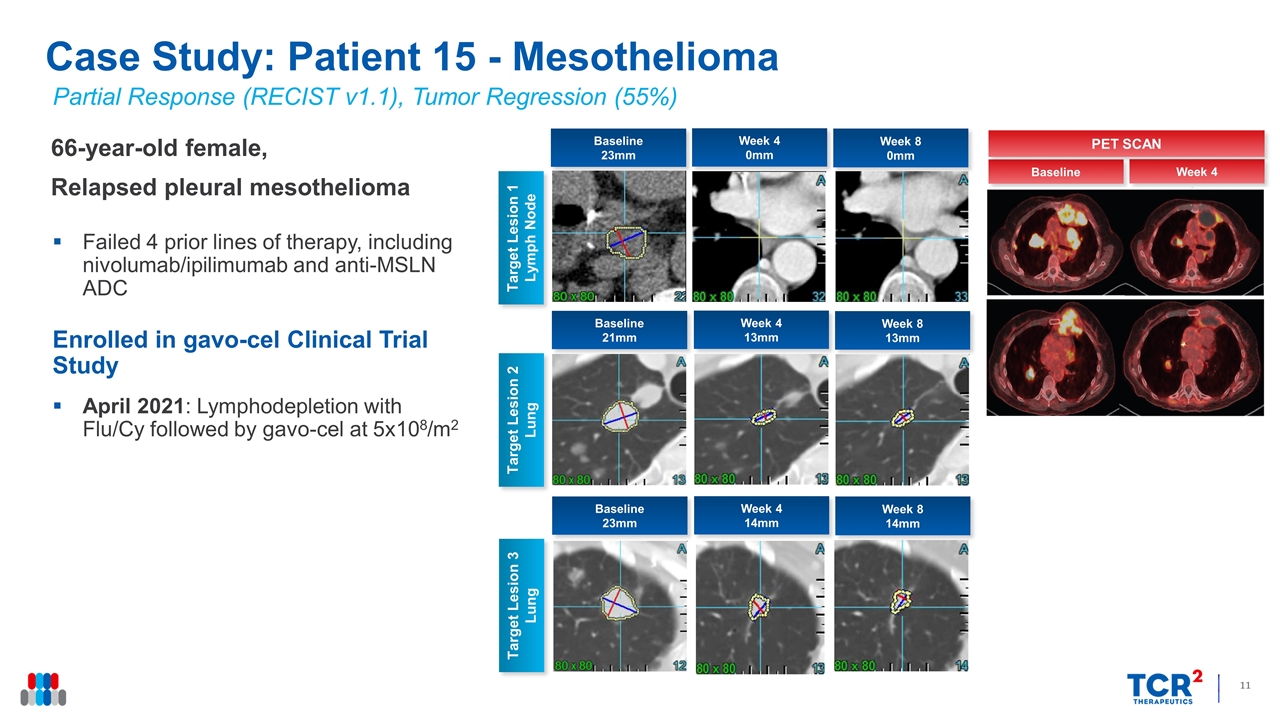
Partial Response (RECIST v1.1), Tumor Regression (55%) 66-year-old female, Relapsed pleural mesothelioma Failed 4 prior lines of therapy, including nivolumab/ipilimumab and anti-MSLN ADC Enrolled in gavo-cel Clinical Trial Study April 2021: Lymphodepletion with Flu/Cy followed by gavo-cel at 5x108/m2 Case Study: Patient 15 - Mesothelioma Week 4 0mm Target Lesion 1 Lymph Node Week 8 0mm Target Lesion 2 Lung Baseline 23mm Week 4 13mm Week 8 13mm Baseline 21mm Target Lesion 3 Lung Week 4 14mm Week 8 14mm Baseline 23mm Week 4 Baseline PET SCAN
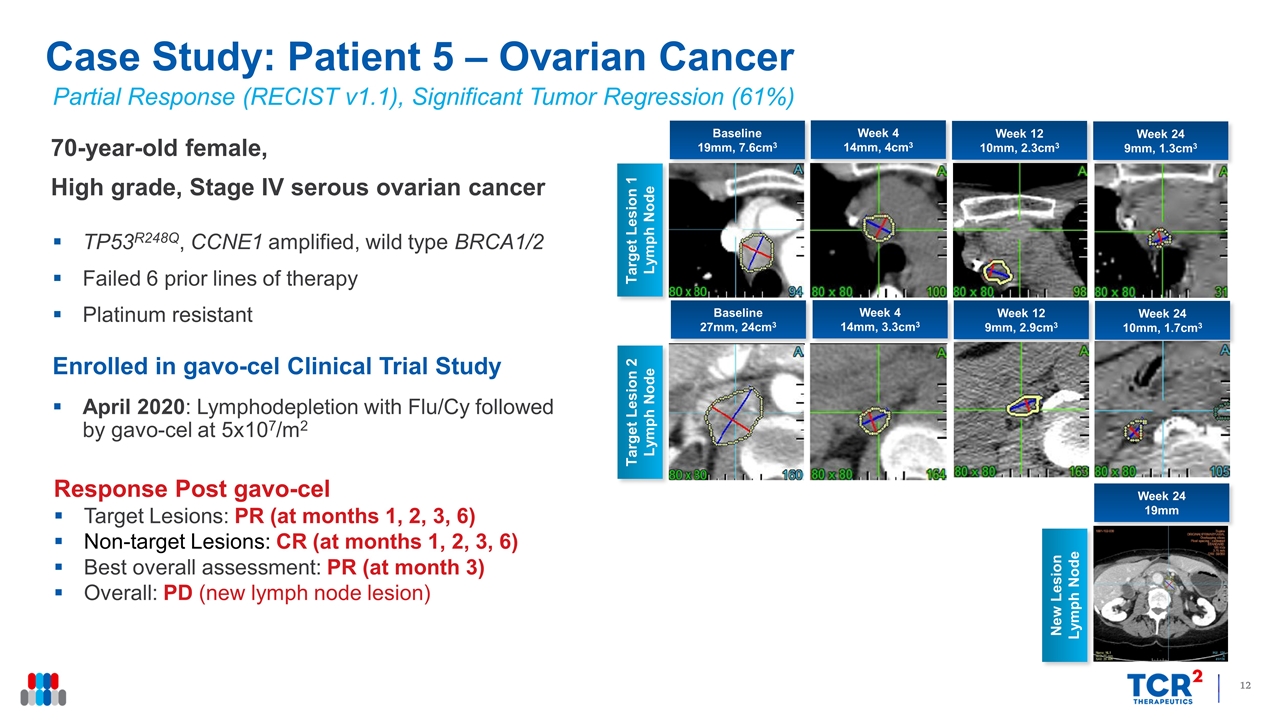
Partial Response (RECIST v1.1), Significant Tumor Regression (61%) 70-year-old female, High grade, Stage IV serous ovarian cancer TP53R248Q, CCNE1 amplified, wild type BRCA1/2 Failed 6 prior lines of therapy Platinum resistant Enrolled in gavo-cel Clinical Trial Study April 2020: Lymphodepletion with Flu/Cy followed by gavo-cel at 5x107/m2 Response Post gavo-cel Target Lesions: PR (at months 1, 2, 3, 6) Non-target Lesions: CR (at months 1, 2, 3, 6) Best overall assessment: PR (at month 3) Overall: PD (new lymph node lesion) Case Study: Patient 5 – Ovarian Cancer Week 4 14mm, 4cm3 Target Lesion 1 Lymph Node Week 12 10mm, 2.3cm3 Week 24 9mm, 1.3cm3 Target Lesion 2 Lymph Node Baseline 19mm, 7.6cm3 Week 4 14mm, 3.3cm3 Week 12 9mm, 2.9cm3 Week 24 10mm, 1.7cm3 Baseline 27mm, 24cm3 Week 24 19mm New Lesion Lymph Node
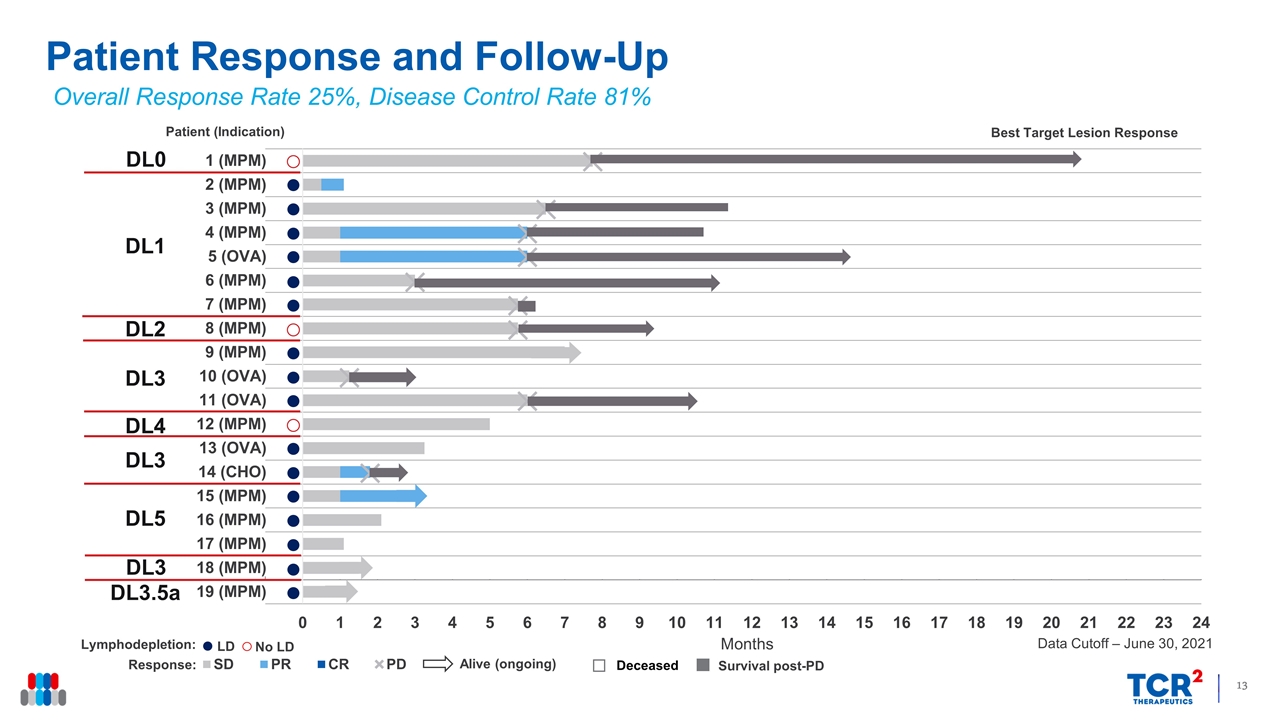
LD No LD Patient Response and Follow-Up Overall Response Rate 25%, Disease Control Rate 81% Data Cutoff – June 30, 2021 Alive (ongoing) Survival post-PD DL1 DL2 DL3 DL4 DL3 DL5 DL0 DL3 DL3.5a
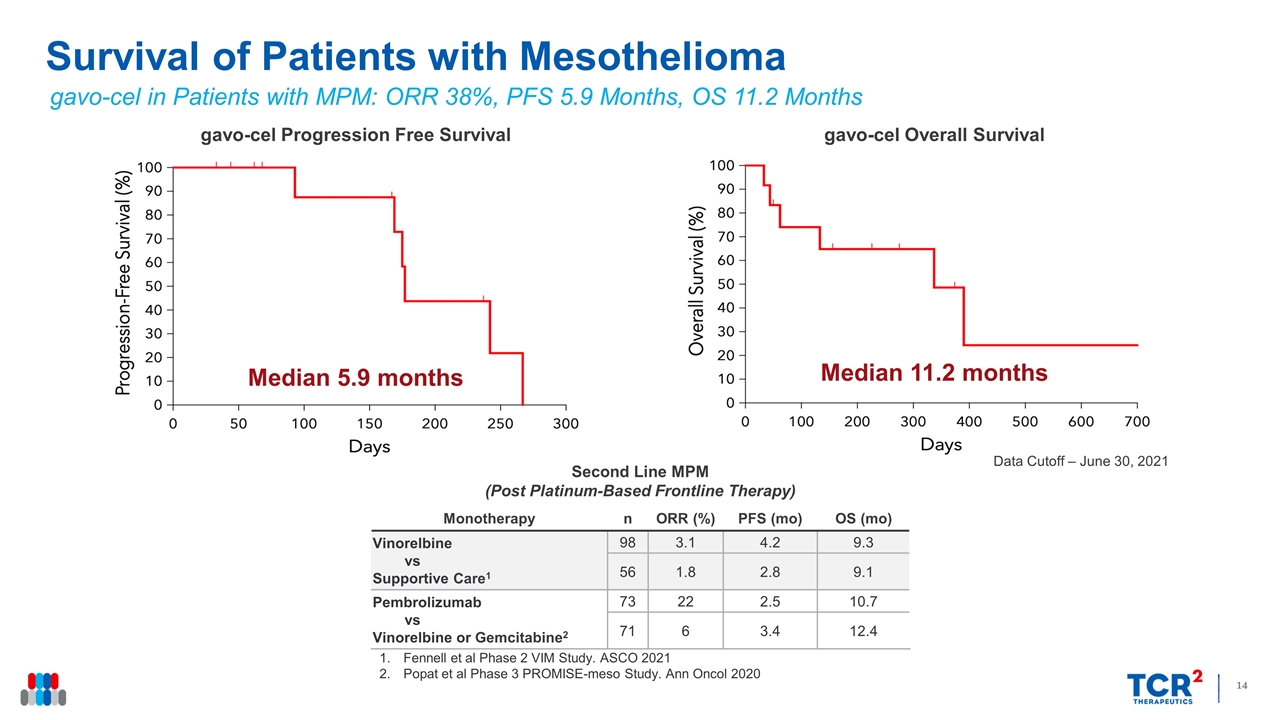
Data Cutoff – June 30, 2021 Survival of Patients with Mesothelioma Monotherapy n ORR (%) PFS (mo) OS (mo) Vinorelbine vs Supportive Care1 98 3.1 4.2 9.3 56 1.8 2.8 9.1 Pembrolizumab vs Vinorelbine or Gemcitabine2 73 22 2.5 10.7 71 6 3.4 12.4 Second Line MPM (Post Platinum-Based Frontline Therapy) gavo-cel Overall Survival Median 11.2 months gavo-cel Progression Free Survival Median 5.9 months Fennell et al Phase 2 VIM Study. ASCO 2021 Popat et al Phase 3 PROMISE-meso Study. Ann Oncol 2020 gavo-cel in Patients with MPM: ORR 38%, PFS 5.9 Months, OS 11.2 Months
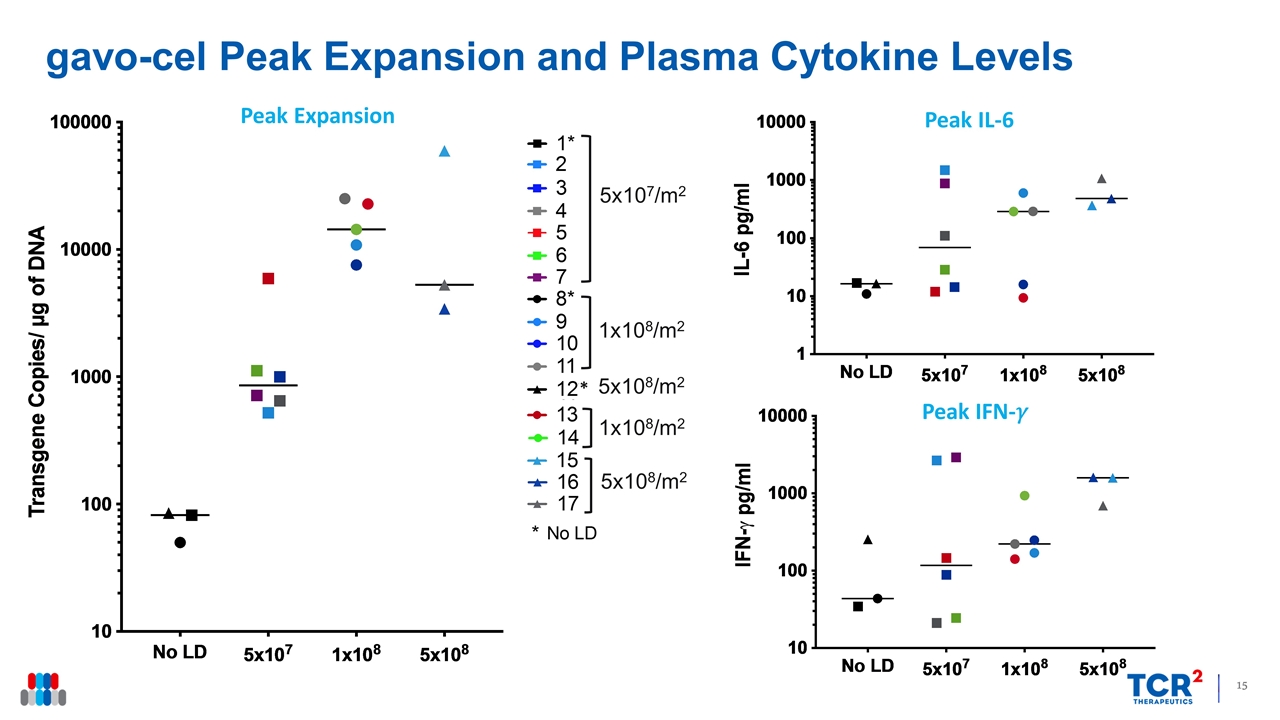
gavo-cel Peak Expansion and Plasma Cytokine Levels Peak Expansion Peak IFN-�� Peak IL-6 5x107/m2 1x108/m2 5x108/m2 1x108/m2 5x108/m2 *
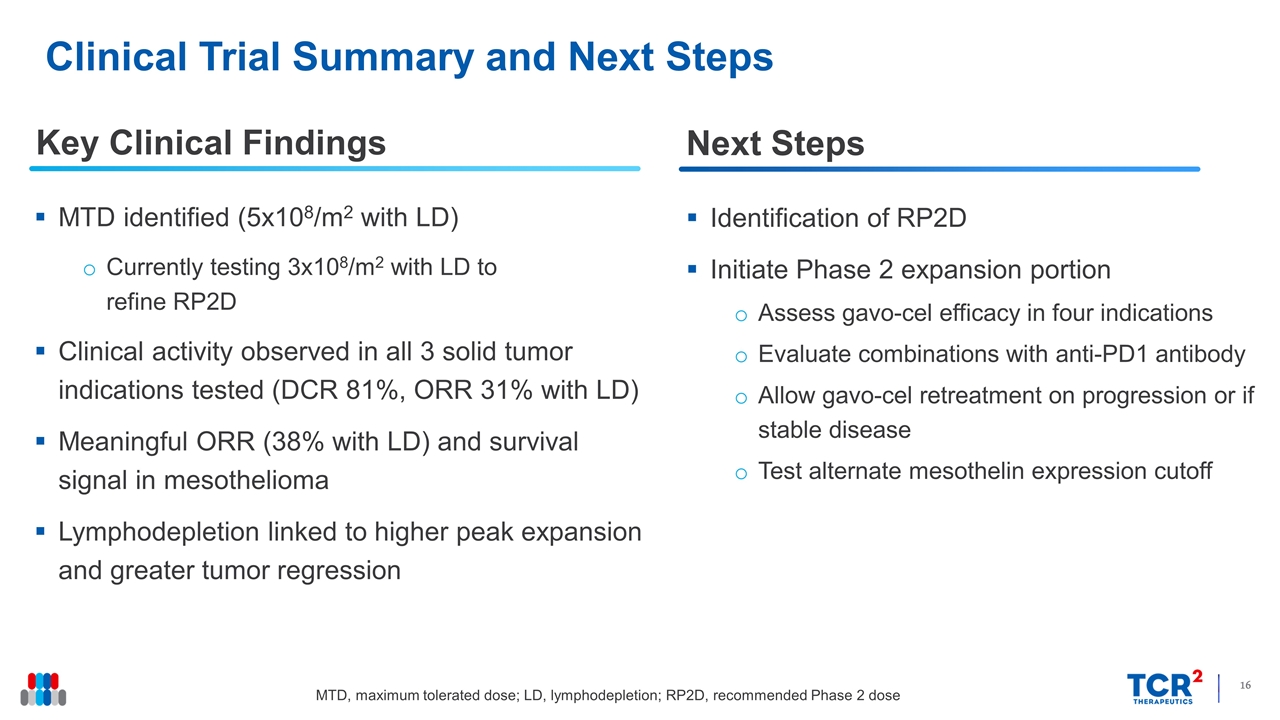
Clinical Trial Summary and Next Steps Key Clinical Findings MTD identified (5x108/m2 with LD) Currently testing 3x108/m2 with LD to refine RP2D Clinical activity observed in all 3 solid tumor indications tested (DCR 81%, ORR 31% with LD) Meaningful ORR (38% with LD) and survival signal in mesothelioma Lymphodepletion linked to higher peak expansion and greater tumor regression Identification of RP2D Initiate Phase 2 expansion portion Assess gavo-cel efficacy in four indications Evaluate combinations with anti-PD1 antibody Allow gavo-cel retreatment on progression or if stable disease Test alternate mesothelin expression cutoff Next Steps MTD, maximum tolerated dose; LD, lymphodepletion; RP2D, recommended Phase 2 dose
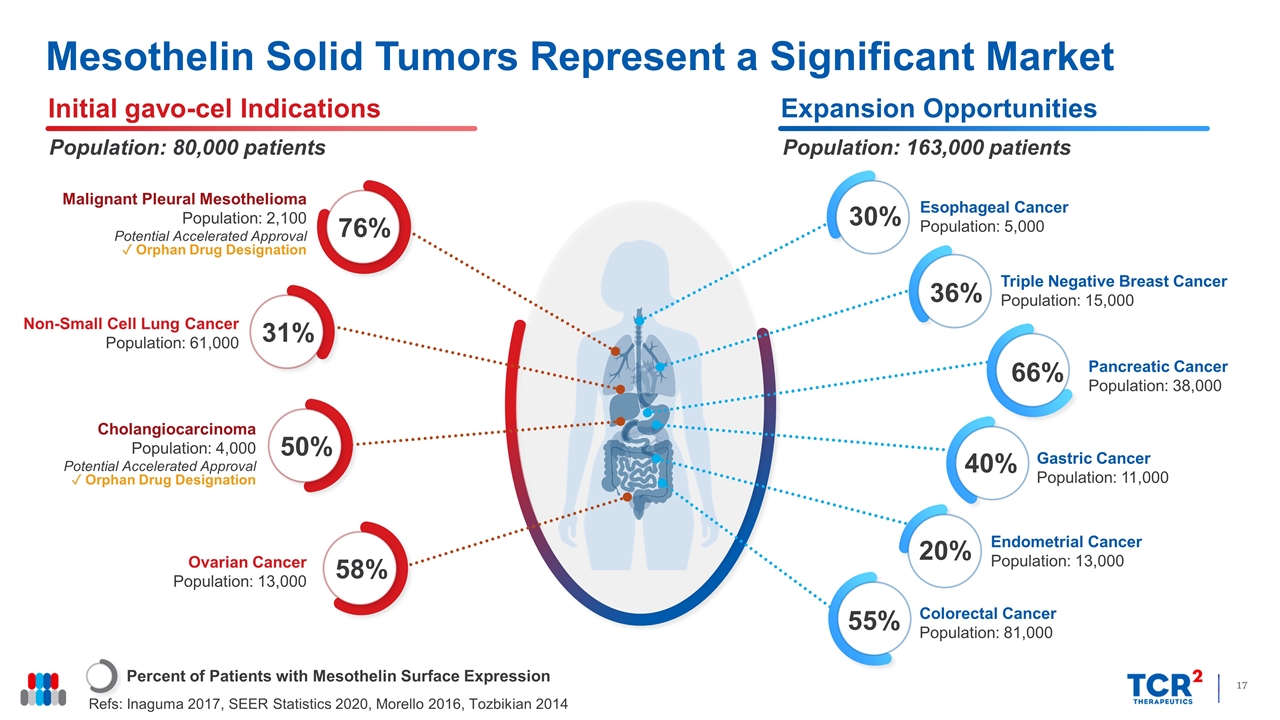
Mesothelin Solid Tumors Represent a Significant Market Percent of Patients with Mesothelin Surface Expression Refs: Inaguma 2017, SEER Statistics 2020, Morello 2016, Tozbikian 2014 Initial gavo-cel Indications Population: 80,000 patients Expansion Opportunities Population: 163,000 patients 76% Malignant Pleural Mesothelioma Population: 2,100 Potential Accelerated Approval ✓ Orphan Drug Designation 58% Ovarian Cancer Population: 13,000 Cholangiocarcinoma Population: 4,000 Potential Accelerated Approval ✓ Orphan Drug Designation 50% Non-Small Cell Lung Cancer Population: 61,000 31% Gastric Cancer Population: 11,000 40% Endometrial Cancer Population: 13,000 20% Colorectal Cancer Population: 81,000 55% Esophageal Cancer Population: 5,000 30% Triple Negative Breast Cancer Population: 15,000 36% Pancreatic Cancer Population: 38,000 66%
MSLN, CD70, GPC3 PD-1 Switch, IL-15, Dual TRuCs POC for Lead Candidate Solid Tumor Franchise Enhancements Allogeneic Platform Versatile TRuC Platform World Class Cell Therapy-Specific Leadership In-House Manufacturing Capabilities Cash as of 2Q21 ~$317M Expansion into Autoimmune Disease TRuC T-Regs The Path Forward – Powering a Solid Tumor Franchise TRuC, T Cell Receptor Fusion Construct; MSLN, mesothelin; POC, proof of concept

R&D Day – October 20, 2021 Further update on gavo-cel clinical trial and Phase 2 strategy Individual program reviews and pipeline strategy Allogeneic development candidate and strategy/timelines Development and data for TRuC T-reg T cells

Thank You
Serious News for Serious Traders! Try StreetInsider.com Premium Free!
You May Also Be Interested In
- Port Houston Regular Commission Meeting
- Ridgeline Minerals Announces Upsized Non-Brokered Private Placement to $2.2M
- FendX Announces Corporate Update
Create E-mail Alert Related Categories
SEC FilingsSign up for StreetInsider Free!
Receive full access to all new and archived articles, unlimited portfolio tracking, e-mail alerts, custom newswires and RSS feeds - and more!



 Tweet
Tweet Share
Share