Form 8-K PIERIS PHARMACEUTICALS, For: May 17

PRESS RELEASE PIERIS PHARMACEUTICALS REPORTS FIRST QUARTER 2021 FINANCIAL RESULTS AND PROVIDES CORPORATE UPDATE COMPANY TO HOST AN INVESTOR CONFERENCE CALL ON MONDAY, MAY 17, 2021 AT 8:00 AM EDT PRS-060/AZD1402 dosed to first patient in phase 2a study Cinrebafusp alfa (PRS-343) phase 2 study expected to begin this summer Announced license and collaboration agreement with Boston Pharmaceuticals for PRS- 342, a preclinical 4-1BB/GPC3 immuno-oncology bispecific; Pieris to receive $10 million upfront payment and is eligible to receive up to approximately $353 million in milestone payments, as well as tiered royalties and sublicense revenue share BOSTON, MA, May 17, 2021 - Pieris Pharmaceuticals, Inc. (NASDAQ: PIRS), a clinical-stage biotechnology company advancing novel biotherapeutics through its proprietary Anticalin® technology platform for respiratory diseases, cancer, and other indications, today reported financial results for the first quarter of 2021 ended March 31, 2021 and provided an update on the Company’s recent and anticipated future developments. “The last few months were marked by significant accomplishments. We leveraged existing and new alliances to materially bolster our balance sheet with $46 million in capital through recent upfront and milestone payments, focused equity stakes by partners AstraZeneca and Seagen, and ongoing funding support by our partners for several assets, ensuring that we are well positioned to execute on our strategic plans into 2023. These partnerships are helping us advance several therapeutic programs and further validate our Anticalin platform. We are pleased that dosing of asthmatic patients with PRS-060/AZD1402 has begun in the previously announced phase 2a study, and we expect to report data from this study next year. The phase 1 data we presented at AACR last month for cinrebafusp alfa, our 4-1BB/HER2 bispecific, support entry into a phase 2 study this summer in HER2-high and HER2-low gastric cancer, where we are setting a high bar to govern development beyond the initial cohort in each study arm. The preclinical data we presented at AACR with our co-development partner, Servier, for PRS-344/S095012, a 4-1BB/PD-L1 bispecific, provide a compelling rationale for clinical development, which will begin later this year. Expanding our roster of 4-1BB-based bispecifics advancing towards clinical development, we also recently announced an immuno-oncology collaboration with Boston Pharmaceuticals to advance PRS-342, a 4- 1BB/GPC3 bispecific, into the clinic,” said Stephen S. Yoder, President and Chief Executive Officer of Pieris. PRS-060/AZD1402 and AstraZeneca Collaboration: Dosing has begun in the first part of the global phase 2a study of PRS-060/AZD1402, an inhaled IL-4 receptor alpha inhibitor under development in collaboration with AstraZeneca for the treatment of moderate-to-severe asthma. Pieris and AstraZeneca expect to announce data from the phase 2a study next year. Upon completion of the study, which is being sponsored and funded by AstraZeneca, Pieris will have options to co-develop and, subsequently, co-commercialize PRS-060/AZD1402 in the United States. Pieris and AstraZeneca continue to advance each of the four programs in the collaboration beyond PRS-060/AZD1402. Cinrebafusp Alfa (PRS-343): Pieris is preparing a two-arm phase 2 study for cinrebafusp alfa, a 4-1BB/HER2 bispecific for the treatment of HER2-expressing solid tumors, in gastric cancer that will begin this summer and will set a high bar for clinical benefit, durability of response, and safety to determine further development beyond the planned initial 20 patients in each study arm. Pieris presented updated data for this program in an oral presentation at the American Association for

Cancer Research (AACR) Annual Meeting 2021. In clinical trials to date, cinrebafusp alfa has shown an acceptable safety profile at all doses tested with no dose-limiting toxicities. Single-agent activity has been most pronounced on a Q2W regimen, which includes one confirmed complete response and four confirmed partial responses, and which will be the basis for phase 2 development. The bispecific also showed a clear dose response and a 4-1BB-driven mechanism of action based on clinical benefit and pharmacodynamic correlates. Additionally, clinical activity was observed in patients with "cold" tumors, as well as those with HER2-low expressing tumors. Supported by these data, the first arm of the announced phase 2 study will be in combination with ramucirumab and paclitaxel in HER2-high gastric cancer, while the second arm will be in combination with tucatinib in HER2-low gastric cancer. Collaboration partners Lilly and Seagen will supply ramucirumab and tucatinib, respectively, under previously announced drug supply agreements. PRS-344/S095012 and Servier Collaboration: Pieris and Servier presented preclinical data for PRS-344/S095012, a 4-1BB/PD-L1 bispecific, as part of a poster session at the AACR Annual Meeting 2021. PRS-344/S095012 induced a dose-dependent anti-tumor response and significantly extended survival in an anti-PD-L1-resistant mouse model. The data also demonstrated PRS- 344/S095012 to be superior to the combination of individual PD-L1- and 4-1BB- targeting molecules. PRS-344/S095012 is expected to enter phase 1 development later this year. Pieris holds exclusive commercialization rights for PRS-344 in the United States and will receive royalties on ex-U.S. sales for this program. As part of our multi-program immuno-oncology collaboration, Servier is also responsible for further development of PRS-352, an undisclosed bispecific. PRS-342/BOS-342 and Boston Pharmaceuticals Collaboration: Pieris entered into an exclusive product license agreement wherein Boston Pharmaceuticals will develop PRS-342, a 4-1BB/GPC3 immuno-oncology Anticalin-antibody bispecific fusion protein in the IND-enabling phase. Under the terms of the agreement, Boston Pharmaceuticals exclusively licensed worldwide rights to PRS-342. Pieris will receive an upfront payment of $10 million and is further entitled to receive up to approximately $353 million in development, regulatory, and sales-based milestone payments, and tiered royalties. Pieris will also contribute an undisclosed amount and input for manufacturing activities to support IND-readiness. Preclinical Respiratory Pipeline: Pieris continues to advance several proprietary early-stage respiratory programs and expects to share data and rationale for advancement of one of its lead innovative proprietary programs this year. First Quarter Financial Update: Cash Position – Cash and cash equivalents totaled $66.8 million for the quarter ended March 31, 2021, compared to a cash and cash equivalents balance of $70.4 million for the quarter ended December 31, 2020. The increase was due to $13 million received from Seagen in March 2021 offset by operating cash needs. The quarter end cash balance excludes both $23 million received from AstraZeneca in April and $10 million to be received from Boston Pharmaceuticals. R&D Expense – R&D expenses were $16.6 million for the quarter ended March 31, 2021, compared to $12.8 million for the quarter ended March 31, 2020. The increase reflects higher spending on preclinical activities for our proprietary respiratory program and manufacturing costs for our immuno-oncology programs, while maintaining flat spending on other non-project related R&D costs. G&A Expense – G&A expenses were $4.1 million for the quarter ended March 31, 2021, compared to $4.4 million for the quarter ended March 31, 2020. The decrease reflects more one-time costs incurred in 2020 related to the move to a new R&D facility in Hallbergmoos, Germany, partially offset by higher consulting expenses in the first quarter of 2021.

Net Loss - Net loss was $4.2 million or $(0.07) per share for the quarter ended March 31, 2021, compared to a net loss of $3.6 million or $(0.07) per share for the quarter ended March 31, 2020. First Quarter Financial Update Conference Call: Pieris management will host a conference call beginning at 8:00 AM EDT on Monday, May 17, 2021, to discuss the first quarter of 2021 financial results and provide a corporate update. Individuals can join the call by dialing +1-877-407-8920 (US & Canada) or +1-412-902-1010 (International). Alternatively, a listen-only audio webcast of the call can be accessed here. For those unable to participate in the conference call or listen to the webcast, a replay will be available on the Investors section of the Company's website, www.pieris.com. About Pieris Pharmaceuticals: Pieris is a clinical-stage biotechnology company that discovers and develops Anticalin protein-based drugs to target validated disease pathways in a unique and transformative way. Our pipeline includes inhalable Anticalin proteins to treat respiratory diseases and immuno-oncology multi-specifics tailored for the tumor microenvironment. Proprietary to Pieris, Anticalin proteins are a novel class of therapeutics validated in the clinic and by partnerships with leading pharmaceutical companies, including AstraZeneca, Seagen, and Servier. Anticalin® is a registered trademark of Pieris. For more information, visit www.pieris.com. Forward Looking Statements: This press release contains forward-looking statements as that term is defined in Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Statements in this press release that are not purely historical are forward-looking statements. Such forward-looking statements include, among other things, whether the combination of cinrebafusp alfa with other therapies could address a high medical need in HER2 gastric cancer patients who do not respond to traditional HER2-targeted therapies; whether the effects of the combination of cinrebafusp alfa with other therapies seen in preclinical studies will be observed in clinical trials; whether data from patients enrolled to date will be sufficient to inform the recommended phase 2 dose for the Company’s planned proof of concept study of cinrebafusp alfa in gastric cancer; the expected timing and potential outcomes of the reporting by the Company of key clinical data from its programs, references to novel technologies and methods and our business and product development plans, including the Company’s cash resources, the advancement of our proprietary and co-development programs into and through the clinic and the expected timing for reporting data, making IND filings or achieving other milestones related to our programs, including PRS-060/AZD1402, cinrebafusp alfa, PRS-344, and PRS-352 and the expected timing of the initiation of the next stage of cinrebafusp alfa’s development in gastric cancer. Actual results could differ from those projected in any forward-looking statements due to numerous factors. Such factors include, among others, our ability to raise the additional funding we will need to continue to pursue our business and product development plans; the inherent uncertainties associated with developing new products or technologies and operating as a development stage company; our ability to develop, complete clinical trials for, obtain approvals for and commercialize any of our product candidates, including our ability to recruit and enroll patients in our studies; our ability to address the requests of the U.S. Food and Drug Administration; competition in the industry in which we operate; delays or disruptions due to COVID-19; and market conditions. These forward-looking statements are made as of the date of this press release, and we assume no obligation to update the forward-looking statements, or to update the reasons why actual results could differ from those projected in the forward-looking statements, except as required by law. Investors should consult all of the information set forth herein and should also refer to the risk factor disclosure set forth in the reports and other documents we file with the Securities and Exchange Commission available at www.sec.gov, including without limitation the Company's Annual Report on Form 10-K for the fiscal year ended December 31, 2020 and the Company's Quarterly Reports on Form 10-Q.

Investor Relations Contact: Pieris Pharmaceuticals, Inc. Maria Kelman Executive Director, Investor Relations +1 857 362 9635 [email protected]
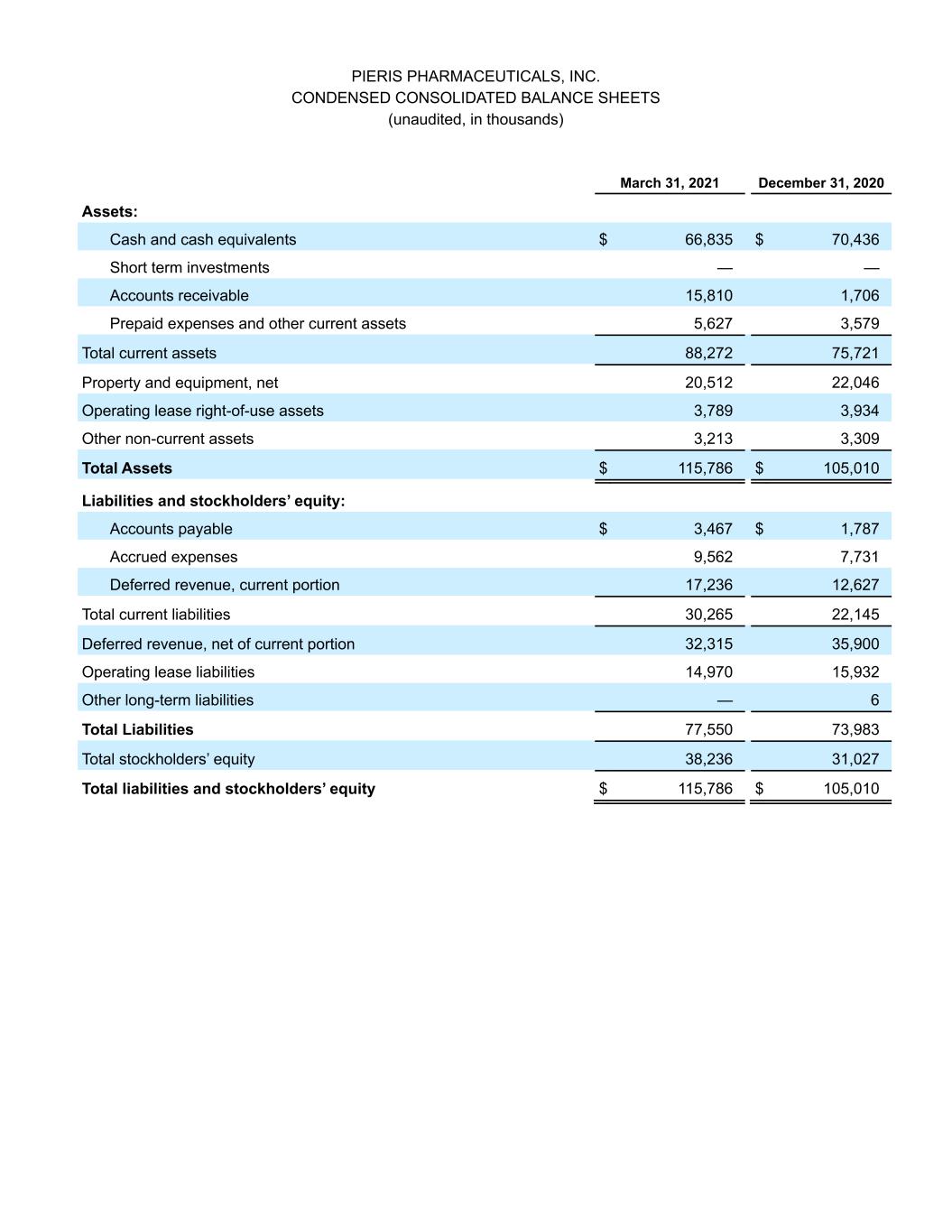
PIERIS PHARMACEUTICALS, INC. CONDENSED CONSOLIDATED BALANCE SHEETS (unaudited, in thousands) March 31, 2021 December 31, 2020 Assets: Cash and cash equivalents $ 66,835 $ 70,436 Short term investments — — Accounts receivable 15,810 1,706 Prepaid expenses and other current assets 5,627 3,579 Total current assets 88,272 75,721 Property and equipment, net 20,512 22,046 Operating lease right-of-use assets 3,789 3,934 Other non-current assets 3,213 3,309 Total Assets $ 115,786 $ 105,010 Liabilities and stockholders’ equity: Accounts payable $ 3,467 $ 1,787 Accrued expenses 9,562 7,731 Deferred revenue, current portion 17,236 12,627 Total current liabilities 30,265 22,145 Deferred revenue, net of current portion 32,315 35,900 Operating lease liabilities 14,970 15,932 Other long-term liabilities — 6 Total Liabilities 77,550 73,983 Total stockholders’ equity 38,236 31,027 Total liabilities and stockholders’ equity $ 115,786 $ 105,010
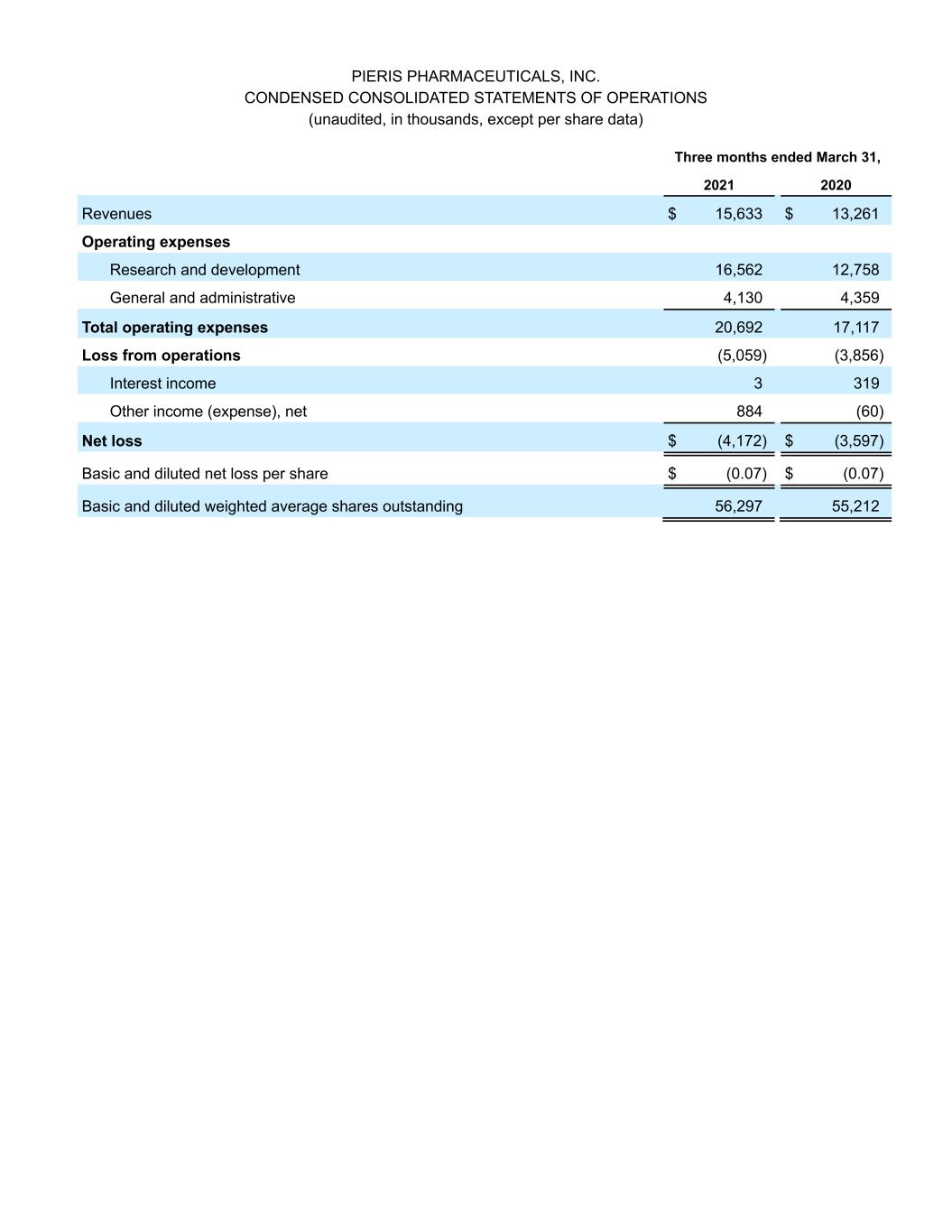
PIERIS PHARMACEUTICALS, INC. CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS (unaudited, in thousands, except per share data) Three months ended March 31, 2021 2020 Revenues $ 15,633 $ 13,261 Operating expenses Research and development 16,562 12,758 General and administrative 4,130 4,359 Total operating expenses 20,692 17,117 Loss from operations (5,059) (3,856) Interest income 3 319 Other income (expense), net 884 (60) Net loss $ (4,172) $ (3,597) Basic and diluted net loss per share $ (0.07) $ (0.07) Basic and diluted weighted average shares outstanding 56,297 55,212
Serious News for Serious Traders! Try StreetInsider.com Premium Free!
You May Also Be Interested In
- InventHelp Inventor Develops New Charging Station for Phones (PHO-4008)
- Cencia Exchange has purchased commercial insurance for liquidation, providing cash claim protection for Cencia users
- Premier’s PINC AI™ and Fortune Name the Nation’s 100 Top Hospitals®
Create E-mail Alert Related Categories
SEC FilingsSign up for StreetInsider Free!
Receive full access to all new and archived articles, unlimited portfolio tracking, e-mail alerts, custom newswires and RSS feeds - and more!



 Tweet
Tweet Share
Share