Form 6-K Sol-Gel Technologies For: May 16
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
Report of Foreign Private Issuer
Pursuant to Rule 13a-16 or 15d-16
Under the Securities Exchange Act of 1934
For the month of May 2022
Commission File Number 001-38367
SOL-GEL TECHNOLOGIES LTD.
(Translation of registrant’s name into English)
7 Golda Meir Street
Ness Ziona 7403650, Israel
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover Form 20-F or
Form 40-F.
Form 20-F ☒ Form 40-F ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule
101(b)(1): ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule
101(b)(7): ☐
INFORMATION CONTAINED IN THIS REPORT ON FORM 6-K
Sol-Gel Technologies Ltd. (the “Company”) has made available an updated presentation about its business, a copy of which is furnished
herewith as Exhibit 99.1 and incorporated by reference.
2
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the Registrant has duly caused this report
to be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
SOL-GEL TECHNOLOGIES LTD.
|
||
|
|
|
|
|
|
Date: May 16, 2022
|
By:
|
/s/ Gilad Mamlok
|
|
|
|
|
Gilad Mamlok
|
|
|
|
|
Chief Financial Officer
|
|
3
Exhibit 99.1

NASDAQ: SLGL

This presentation contains “forward-looking statements” within the meaning of the Private Securities
Litigation Reform Act of 1995. All statements contained in this presentation that do not relate to matters of historical fact should be considered forward-looking statements, including, but not limited to, statements regarding the commercial
launch of TWYNEO, the regulatory approval of EPSOLAY, our expected cash runway, and the benefits we expect to receive under our agreement with Galderma. These forward-looking statements include information about possible or assumed future
results of our business, financial condition, results of operations, liquidity, plans and objectives. In some cases, you can identify forward-looking statements by terminology such as “believe,” “may,” “estimate,” “continue,” “anticipate,”
“intend,” “should,” “plan,” “expect,” “predict,” “potential,” or the negative of these terms or other similar expressions. Forward-looking statements are based on information we have when those statements are made or our management’s current
expectations and are subject to risks and uncertainties that could cause actual performance or results to differ materially from those expressed in or suggested by the forward-looking statements. Important factors that could cause such
differences include, but are not limited to, the risk that we will not receive all of the anticipated benefits under our agreement with Galderma, the risk of a delay in the commercial availability of TWYNEO and/or EPSOLAY, the risk that
TWYNEO will not provide treatment to the number of patients anticipated, risks relating to the effects of COVID 19 (coronavirus) as well as the following factors: (i) the adequacy of our financial and other resources, particularly in light of
our history of recurring losses and the uncertainty regarding the adequacy of our liquidity to pursue our complete business objectives; (ii) our ability to complete the development of our product candidates; (iii) our ability to find suitable
co-development partners; (iv) our ability to obtain and maintain regulatory approvals for our product candidates in our target markets, the potential delay in receiving such regulatory approvals and the possibility of adverse regulatory or
legal actions relating to our product candidates even if regulatory approval is obtained; (v) our ability to commercialize our pharmaceutical product candidates; (vi) our ability to obtain and maintain adequate protection of our intellectual
property; (vii) our ability to manufacture our product candidates in commercial quantities, at an adequate quality or at an acceptable cost; (viii) our ability to establish adequate sales, marketing and distribution channels; (ix) acceptance
of our product candidates by healthcare professionals and patients; (x) the possibility that we may face third-party claims of intellectual property infringement; (xi) the timing and results of clinical trials that we may conduct or that our
competitors and others may conduct relating to our or their products; (xii) intense competition in our industry, with competitors having substantially greater financial, technological, research and development, regulatory and clinical,
manufacturing, marketing and sales, distribution and personnel resources than we do; (xiii) potential product liability claims; (xiv) potential adverse federal, state and local government regulation in the United States, Europe or Israel; and
(xv) loss or retirement of key executives and research scientists. These and other important factors discussed in the Company's Annual Report on Form 20-F filed with the Securities and Exchange Commission (“SEC”) on April 4, 2022, as amended,
and our other reports filed with the SEC could cause actual results to differ materially from those indicated by the forward-looking statements made in this presentation Except as required by law, we undertake no obligation to update any
forward-looking statements in this presentation. FORWARD-LOOKING STATEMENTS

PIONEERING TOPICAL DERMATOLOGICAL DRUGS Two FDA Approvals Within One Year Not a real patient Not a
real patient

Say hello to thisnever-before-seen combo TWYNEO® FOR ACNE WAS LAUNCHED First and only FDA Approved
Fixed‑Dose Combination of Tretinoin and Benzoyl Peroxide
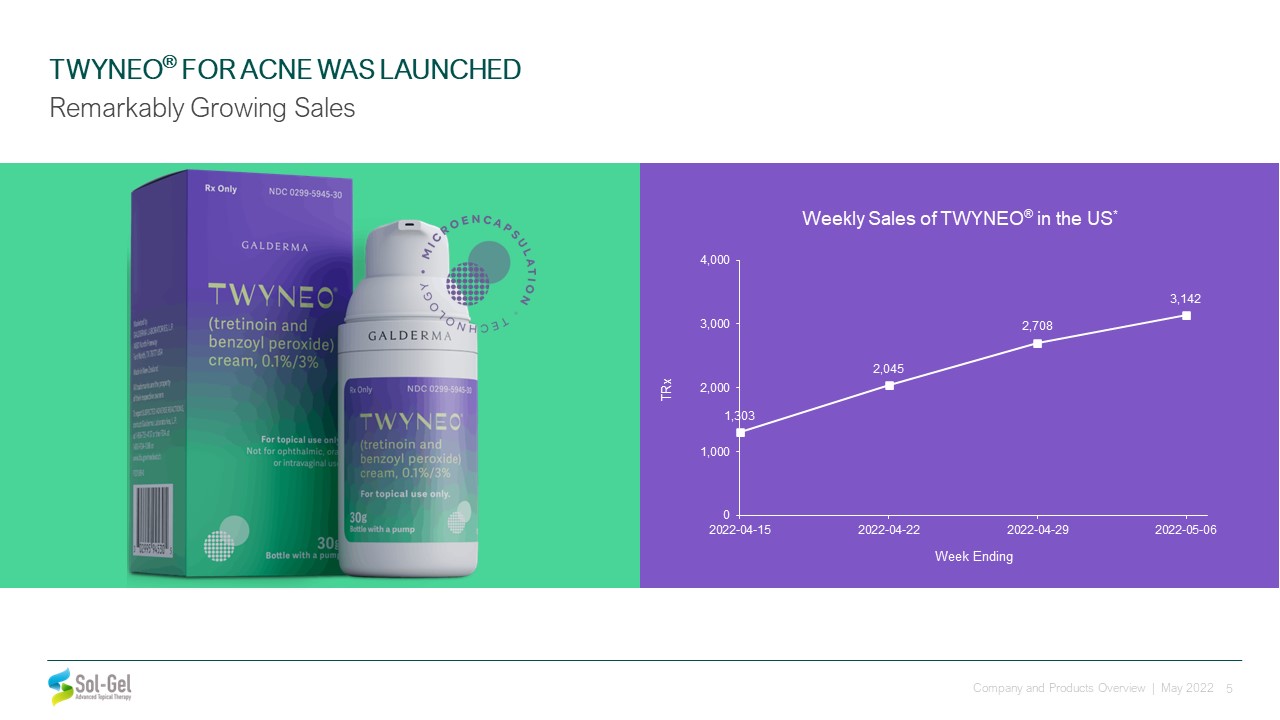
TWYNEO® FOR ACNE WAS LAUNCHED Remarkably Growing Sales

Acne vulgaris is a multifactorial disease. Even though benzoyl peroxide and tretinoin are widely
prescribed separately and have a complementary mechanism of action, so far, they could not be applied concomitantly because benzoyl peroxide decomposes tretinoin TWYNEO contains a fixed‑dose combination of tretinoin and benzoyl peroxide.
TWYNEO uses Sol‑Gel’s patented technology to prevent tretinoin from being degraded by benzoyl peroxide and slowly releases each of the active drug ingredients over time to provide a favorable efficacy and safety profile Patent protected
until 2038 by granted patents and until 2041 by a pending patent application TWYNEO® OFFERS COMPREHENSIVE TREATMENT FOR ACNE PATIENTS Potential to Become 1st Line Treatment
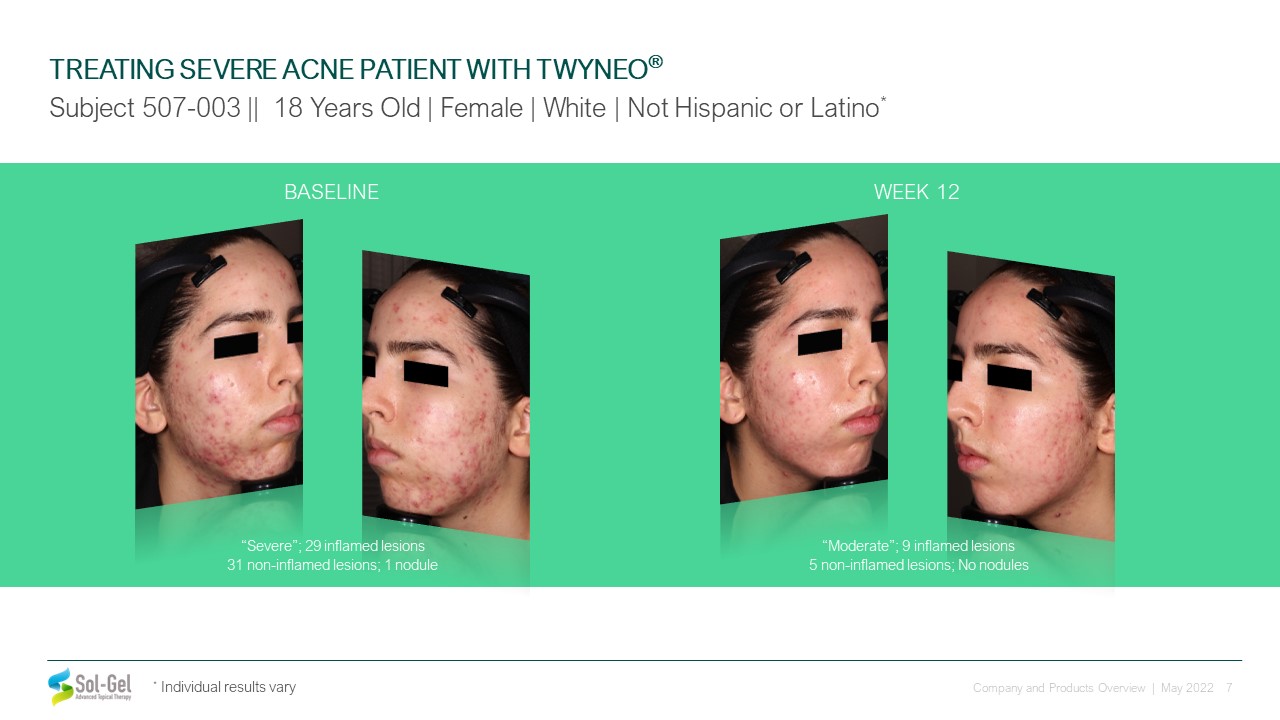
* Individual results vary BASELINE “Severe”; 29 inflamed lesions 31 non-inflamed lesions; 1
nodule WEEK 12 “Moderate”; 9 inflamed lesions 5 non-inflamed lesions; No nodules TREATING SEVERE ACNE PATIENT WITH TWYNEO® Subject 507-003 || 18 Years Old | Female | White | Not Hispanic or Latino*

EPSOLAY® FOR INFLAMMATORY LESIONS OF ROSACEA WAS APPROVED First and only Benzoyl Peroxide in
Rosacea Rosacea care with the brilliant touch of technology

Inflammatory lesions of rosacea resemble acne vulgaris, except that comedones (whiteheads and
blackheads) are absent and only inflammatory lesions exist EPSOLAY contains encapsulated benzoyl peroxide, using Sol‑Gel’s patented technology. Benzoyl peroxide is an effective antibacterial drug that is not associated with bacterial
resistance and is used to treat acne but not rosacea as it is assumed that rosacea patients cannot tolerate benzoyl peroxide. In Phase III clinical studies, EPSOLAY demonstrated statistically significant higher efficacy than the vehicle and
favorable safety and tolerability profile, similar to vehicle Patent protected until 2040 by granted patents and until 2041 by a pending patent application EPSOLAY® OFFERS EFFECTIVE TOPICAL TREATMENT FOR ROSACEA PATIENTS Potential to
Change Treatment Landscape
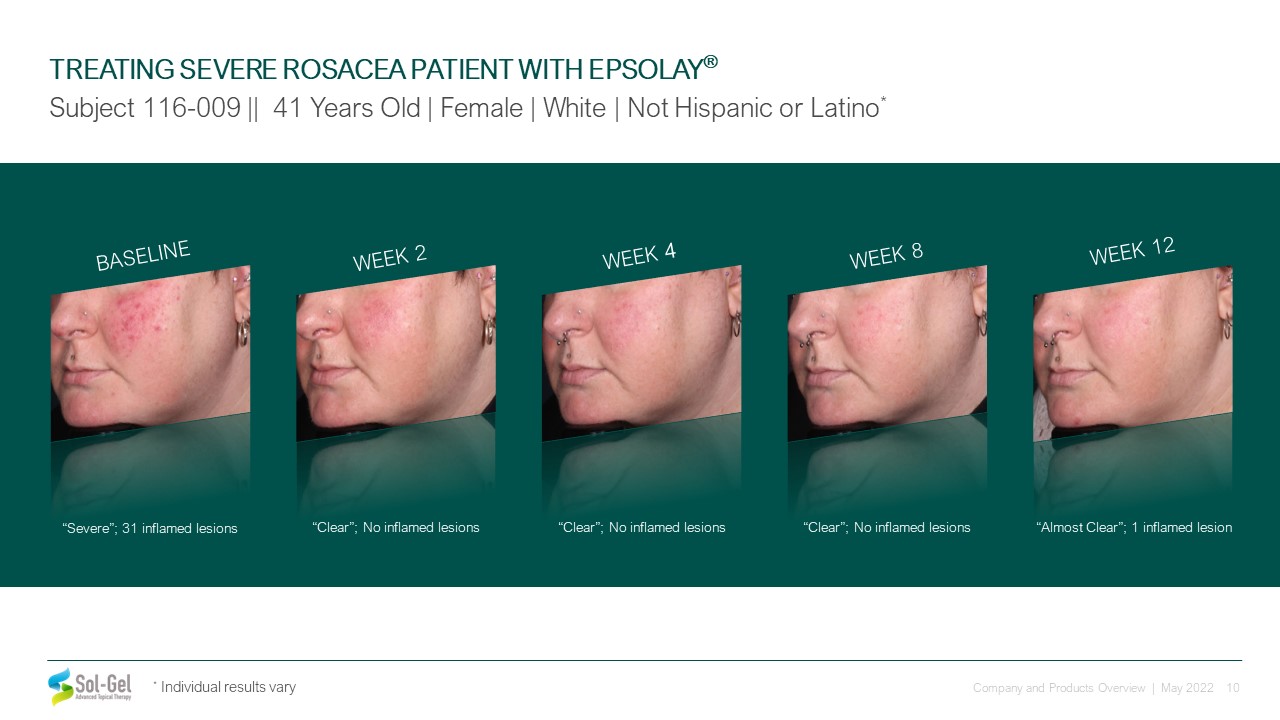
BASELINE “Severe”; 31 inflamed lesions WEEK 2 “Clear”; No inflamed lesions “Clear”; No inflamed
lesions WEEK 12 “Almost Clear”; 1 inflamed lesion WEEK 8 “Clear”; No inflamed lesions TREATING SEVERE ROSACEA PATIENT WITH EPSOLAY® Subject 116-009 || 41 Years Old | Female | White | Not Hispanic or Latino* WEEK 4 * Individual
results vary

PARTNERING WITH MARKET LEADER GALDERMA Galderma has Heritage of Successful Drugs in Acne and Rosacea

REACHING FAVORABLE AGREEMENT WITH GALDERMA Option to Regain Commercialization Rights at No Cost 5
Years following 1st Sale $11 million in upfront and product approval payments Mid- to high-teen percentage of royalties on net sales Up to additional $9 million in sales milestone payments Option to regain commercialization rights 5
years following first sale at no cost Cash-flow positive deal as of launch Allows for focus on innovative pipeline

IMPLEMENTING INNOVATION Enabling Microencapsulation Technology Proprietary silica-based
microencapsulation technology allows development of drugs that have the potential to be more effective and tolerable than existing drugs Core/shell structure designed to boost tolerability High encapsulation efficiency aimed to improve
stability Particle size and release rate tuned to allow efficient delivery of the entrapped API Patented platform strengthens our IP and creates barrier to entry for generic drugs
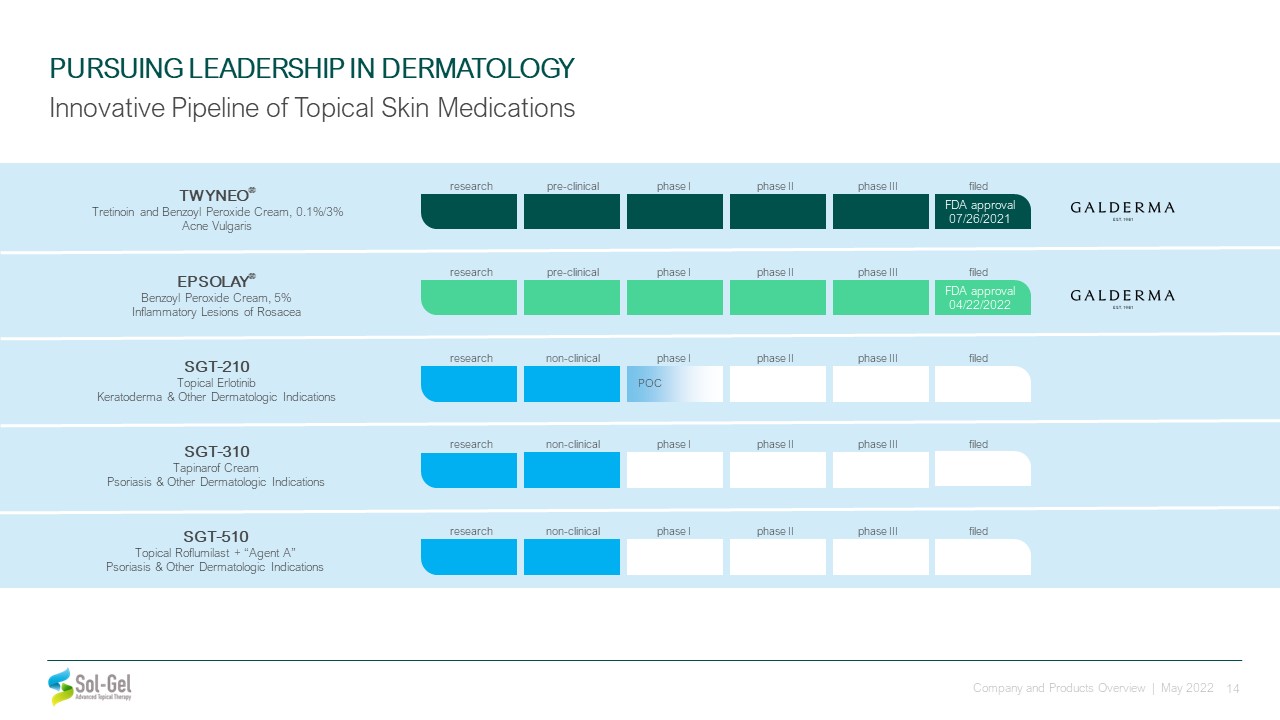
SGT-510 Topical Roflumilast + “Agent A”Psoriasis & Other Dermatologic Indications PURSUING
LEADERSHIP IN DERMATOLOGY Innovative Pipeline of Topical Skin Medications SGT-210 Topical ErlotinibKeratoderma & Other Dermatologic Indications SGT-310 Tapinarof CreamPsoriasis & Other Dermatologic
Indications non-clinical phase II phase III filed research phase I POC non-clinical phase II phase III filed research phase I non-clinical phase II phase III filed research phase I TWYNEO® Tretinoin and Benzoyl Peroxide
Cream, 0.1%/3%Acne Vulgaris EPSOLAY® Benzoyl Peroxide Cream, 5%Inflammatory Lesions of Rosacea FDA approval 04/22/2022 pre-clinical phase II phase III filed research phase I pre-clinical phase II phase III filed research phase
I FDA approval 07/26/2021

Initiate clinical development with a higher concentration of erlotinib in H1 23 DEVELOPING FIRST
ERLOTINIB TOPICAL DRUG SGT-210 for Palmoplantar Keratoderma and other Skin Conditions NEXT STEPS SGT-210 20 patent applications for erlotinib in various skin conditions (as of May 12, 2021)Potential IP protection until 2041 A
proof-of-concept clinical study in palmoplantar keratoderma patients indicated a potential modest improvement with a favorable safety profile
Initiate clinical development in H2 22 DEVELOPING NOVEL TAPINAROF CREAM SGT-310 for Psoriasis and
other Skin Conditions NEXT STEPS SGT-310 42 patent applications for tapinarof in various skin conditions (as of May 12, 2021)Potential IP protection until 2042 SGT-310 is intended to be an alternative to an investigational tapinarof
cream, 1%, for which an NDA was already submitted to the FDA

DEVELOPING NOVEL ROFLUMILAST COMBINATION TOPICAL DRUG SGT-510 for Psoriasis and other Skin
Conditions NEXT STEPS SGT-510 Initiate clinical development in H2 22 A patent application was filed Potential IP protection until 2039 SGT-510 (roflumilast + “Agent A”) is designed to be potentially more effective than roflumilast
cream, 0.3%, for which an NDA was already submitted to the FDA

FOCUSING ON INNOVATIVE PIPELINE WHILE SECURING NON-DILUTIVE FUNDING Sale of Generic Assets to
Padagis Sale of Generic Assets to Padagis in return for $21 million in quarterly installments over 24 months Proceed with 50/50 gross profit-sharing collaboration on 2 programs encompassing 4 high-value generic drug candidates Allows for
focus on innovative pipeline Reduces the need to raise dilutive capital
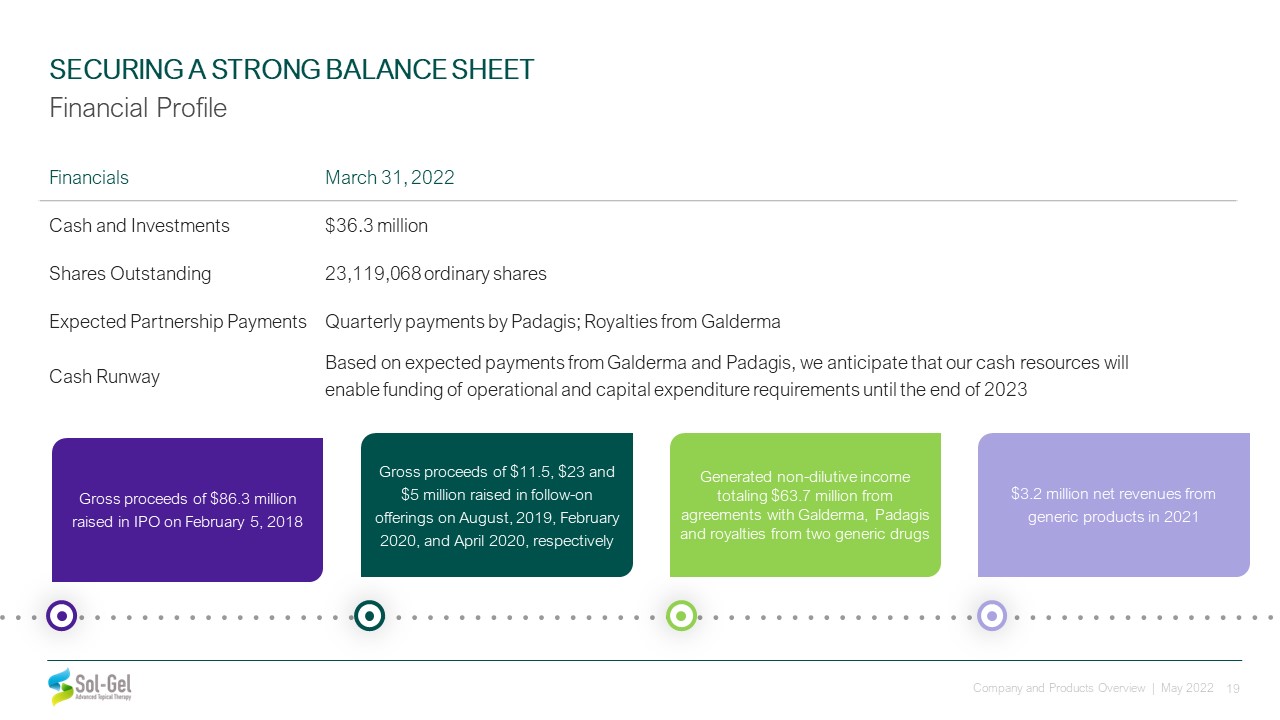
Financials March 31, 2022 Cash and Investments $36.3 million Shares Outstanding 23,119,068
ordinary shares Expected Partnership Payments Quarterly payments by Padagis; Royalties from Galderma Cash Runway Based on expected payments from Galderma and Padagis, we anticipate that our cash resources will enable funding of
operational and capital expenditure requirements until the end of 2023 Gross proceeds of $86.3 million raised in IPO on February 5, 2018 Gross proceeds of $11.5, $23 and $5 million raised in follow-on offerings on August, 2019, February
2020, and April 2020, respectively Generated non-dilutive income totaling $63.7 million from agreements with Galderma, Padagis and royalties from two generic drugs $3.2 million net revenues from generic products in 2021 SECURING A STRONG
BALANCE SHEET Financial Profile

BUILDING OUR FUTURE Investor Highlights Completed development of EPSOLAY and TWYNEO and gained FDA
approvals Maximized likelihood of market success for EPSOLAY and TWYNEO through commercialization agreements with US market leader, while retaining the option to regain commercialization rights 5 years following 1st commercial sale Defined
innovative product pipeline which targets multiple significant US markets and paves the way, together with the market success of EPSOLAY and TWYNEO, to transform Sol-Gel into a leading dermatology company

Indication: TWYNEO® (tretinoin and benzoyl peroxide) Cream, 0.1%/3% is indicated for the topical
treatment of acne vulgaris in adults and pediatric patients 9 years of age and older. Adverse Events: The most common adverse reactions (incidence ≥ 1%) in patients treated with TWYNEO Cream were pain (stinging, burning, or pain), dryness,
exfoliation, erythema (redness), dermatitis, pruritus (itching) and irritation - all at the application site. Warnings/Precautions: Patients using TWYNEO Cream may experience hypersensitivity reactions, including anaphylaxis (acute allergic
reaction), angioedema (rapid swelling), and urticaria (hives). If serious hypersensitivity reaction occurs, discontinue use of TWYNEO Cream immediately and seek medical attention. Skin irritation may be experienced, including application site
dryness, pain (stinging, burning or pain), exfoliation, erythema (redness), dermatitis, pruritus (itching) and irritation. Depending upon the severity, use a moisturizer, reduce the frequency of the application, or discontinue use. Avoid
application to cuts, abrasions, eczematous, or sunburned skin. TWYNEO Cream may increase photosensitivity, sensitivity to ultraviolet light. Minimize or avoid exposure to natural or artificial sunlight (tanning beds or UVA/B treatment). Use
sunscreen or protective clothing when sun exposure cannot be avoided. Discontinue use of TWYNEO Cream at the first evidence of sunburn. You are encouraged to report negative side effects of prescription drugs to the FDA.
Visit www.fda.gov/medwatch or call 1-800-FDA-1088 IMPORTANT SAFETY INFORMATION Indication: EPSOLAY® (benzoyl peroxide) Cream, 5% is indicated for the treatment of inflammatory lesions of rosacea in adults. Adverse Events: The most common
adverse reactions (incidence ≥ 1%) in patients treated with EPSOLAY Cream were pain, erythema (redness), pruritus (itching) and edema (swelling), all at the application site. Warnings/Precautions: Patients using EPSOLAY Cream may experience
hypersensitivity reactions, including anaphylaxis (acute allergic reaction), angioedema (rapid swelling), and urticaria (hives). If serious hypersensitivity reaction occurs, discontinue use of EPSOLAY Cream immediately and seek medical
attention/initiate appropriate therapy. Skin Irritation/contact dermatitis may be experienced, including erythema (redness), scaling, dryness, and stinging/burning. Irritation and contact dermatitis may occur. Use a moisturizer and
discontinue EPSOLAY Cream if symptoms do not improve. Avoid application to cuts, abrasions, eczematous, or sunburned skin. EPSOLAY Cream may increase photosensitivity, sensitivity to ultraviolet light. Minimize or avoid exposure to natural or
artificial sunlight (tanning beds or UVA/B treatment). Use sunscreen or protective clothing when sun exposure cannot be avoided. Discontinue use of EPSOLAY Cream at the first evidence of sunburn. You are encouraged to report negative side
effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088

NASDAQ: SLGL
Serious News for Serious Traders! Try StreetInsider.com Premium Free!
You May Also Be Interested In
- Maximize Your Forex Trading Strategy - Harness the Market Momentum for Enhanced Results.
- Hexagon acquires Xwatch to strengthen its construction safety portfolio
- GWM Focuses on New Energy Development with SVOLT Energy
Create E-mail Alert Related Categories
SEC FilingsSign up for StreetInsider Free!
Receive full access to all new and archived articles, unlimited portfolio tracking, e-mail alerts, custom newswires and RSS feeds - and more!



 Tweet
Tweet Share
Share