Form 8-K KERYX BIOPHARMACEUTICALS For: Nov 09
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of report (Date of earliest event reported): November 9, 2016
Keryx Biopharmaceuticals, Inc.
(Exact Name of Registrant as Specified in Charter)
| Delaware | 000-30929 | 13-4087132 | ||
| (State or Other Jurisdiction of Incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
One Marina Park Drive, 12th Floor
Boston, Massachusetts 02210
(Address of Principal Executive Offices)
(617) 466-3500
(Registrant’s telephone number, including area code)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ | Written communications pursuant to Rule 425 under the Securities Act. |
| ☐ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act. |
| ☐ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. |
| ☐ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. |
| Item 2.02. | Results of Operations and Financial Condition. |
On November 9, 2016, Keryx Biopharmaceuticals, Inc. (“Keryx”) issued a press release announcing its results of operations for the third quarter ended September 30, 2016. Keryx also announced that on November 9, 2016 at 8:00 a.m. ET, it will host an investor conference call to discuss its third quarter financial results and provide a business update. A copy of such press release is being furnished as Exhibit 99.1 to this report and is incorporated herein by reference. A copy of the investor presentation to be used on the investor conference call is being furnished as Exhibit 99.2 to this report and is incorporated herein by reference.
The information set forth in Exhibits 99.1 and 99.2 shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liability of that section, and shall not be incorporated by reference into any registration statement or other document filed under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
| Item 9.01. | Financial Statements And Exhibits. |
| (d) | Exhibits. |
The following exhibits are furnished herewith:
| 99.1 | Press release issued by Keryx Biopharmaceuticals, Inc., dated November 9, 2016. | |
| 99.2 | Investor presentation to be presented by Keryx Biopharmaceuticals, Inc. on November 9, 2016. | |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Keryx Biopharmaceuticals, Inc. (Registrant) | ||||||
| Date: November 9, 2016 |
By: | /s/ Brian Adams | ||||
| Brian Adams | ||||||
| General Counsel and Corporate Secretary | ||||||
INDEX TO EXHIBITS
| Exhibit Number |
Description | |
| 99.1 | Press release issued by Keryx Biopharmaceuticals, Inc., dated November 9, 2016. | |
| 99.2 | Investor presentation to be presented by Keryx Biopharmaceuticals, Inc. on November 9, 2016. | |

|
Exhibit 99.1 |
Keryx Biopharmaceuticals Announces Third Quarter 2016 Financial Results and Provides Corporate Update
| • | The company has established adequate supply of Auryxia®(ferric citrate) and is prepared to make this medicine available to people on dialysis pending U.S. FDA approval of second contract manufacturer |
| • | Keryx has completed the sNDA (supplemental new drug application) seeking to expand Auryxia’s label and will submit it pending final U.S. FDA agreement on the company’s proposed pediatric plan |
BOSTON, MA, November 9, 2016 – Keryx Biopharmaceuticals, Inc. (Nasdaq: KERX), a biopharmaceutical company focused on bringing innovative medicines to people with renal disease, today announced its financial results for the third quarter ended September 30, 2016 and provided a corporate update.
“Our second contract manufacturer is successfully producing Auryxia,” said Greg Madison, president and chief executive officer of Keryx Biopharmaceuticals. “We are ready to promptly make Auryxia available to pharmacies, pending approval of this manufacturer, which has a November 13, 2016 PDUFA date. Since August, we have been working closely with the kidney community to support them through the supply interruption. We are committed to maintaining a consistent supply of Auryxia, and we are working to ensure that the renal care teams will confidently prescribe Auryxia again.”
BUSINESS UPDATE
| • | The company reported Auryxia net U.S. product sales for the third quarter of 2016 of $5.1 million compared to $3.2 million in the third quarter of 2015. Third quarter 2016 product sales reflect approximately 9,700 reported prescriptions of Auryxia during the period. In addition, Keryx announced it will change its method of revenue recognition from the sell-through (deferred) method to the pull-through (ex-factory) method beginning in the fourth quarter of 2016. |
| • | On August 1, 2016, Keryx announced a temporary interruption in the supply of Auryxia was imminent due to a production-related issue at its existing manufacturer in converting active pharmaceutical ingredient (API) to finished drug product. Keryx is working with its existing manufacturer to resolve the issue and resume production. Keryx filed a post-approval supplement application with the U.S. FDA for approval of a second contract manufacturer to supply finished drug product. The application has a Prescription Drug User Fee Act, or PDUFA, action date of November 13, 2016. The company is prepared to promptly supply Auryxia to pharmacies, pending FDA approval of its second contract manufacturer. In October 2016, Keryx entered into a long-term agreement with its second contract manufacturer for the manufacture of commercial supply of finished drug product. |
| • | Keryx announced that it has completed the sNDA seeking an additional indication for the treatment of iron deficiency anemia in adults with stage 3-5 non-dialysis dependent chronic kidney disease (CKD), and is ready to submit the application to the FDA, pending final agreement on its pediatric plan. |
| • | Five abstracts related to ferric citrate have been accepted for poster presentation at the upcoming American Society of Nephrology’s (ASN) 2016 Kidney Week taking place November 15 – 20, 2016. Four of the five abstracts describe data from the pivotal Phase 3 trial evaluating ferric citrate for the treatment of iron deficiency anemia in adults with stage 3-5 non-dialysis dependent CKD. The accepted abstracts are available online on the ASN conference website. In addition, the company plans to submit Phase 3 results for publication in a peer reviewed medical journal. |
Page 1 of 7

FINANCIAL SECTION
Third Quarter Ended September 30, 2016 Financial Results
“In the third quarter, we focused on rebuilding supply of Auryxia while at the same time continuing to invest appropriately in future growth areas, including pre-launch activities for the potential label expansion of ferric citrate for the treatment of adults with IDA and stage 3-5 NDD-CKD,” said Scott Holmes, chief financial officer of Keryx. “As we look ahead, we will be focused on how quickly we can return Auryxia to patients who had previously been prescribed our medicine, and on bringing Auryxia to new patients who could potentially benefit from treatment.”
At September 30, 2016, the company had cash and cash equivalents of $132.2 million.
Total revenues for the quarter ended September 30, 2016 were approximately $6.3 million, compared with $4.2 million during the same period in 2015. Total revenues for the third quarter of 2016 consisted of Auryxia net U.S. product sales of $5.1 million, and license revenue of $1.3 million associated with royalties received on ferric citrate net sales from Keryx’s Japanese partner. Beginning in the fourth quarter of 2016, the company will change its revenue recognition policy from the sell-through method based on patient prescription fulfillments to the pull-through or ex-factory method based on sales of Auryxia to wholesalers and pharmacy customers. This decision was made following the company’s ability to reasonably estimate product returns. Under the ex-factory method, revenue will be recognized when wholesalers and direct customers receive orders of Auryxia.
Cost of goods sold for the quarter ended September 30, 2016 was $18.2 million as compared with $3.1 million during the same period in 2015. Cost of goods sold during the third quarter of 2016 included $13.8 million in write-offs of work-in-process inventory that was determined to no longer be suitable for commercial manufacture. Additionally, cost of goods sold during the quarter also included $2.3 million related to manufacturing charges incurred as a result of not fully utilizing planned production at the company’s existing third-party drug product manufacturer.
Research and development expenses for the quarter ended September 30, 2016 decreased by 22 percent to $8.7 million as compared to $11.2 million during the same period in 2015. The decrease was primarily due to a reduction in clinical expenses associated with the company’s completed Phase 3 clinical trial evaluating ferric citrate for the treatment of IDA in adults with stage 3-5 non-dialysis dependent CKD.
Selling, general and administrative expenses for the quarter ended September 30, 2016 were $20.5 million as compared with $20.2 million during the same period in 2015.
Net loss for the third quarter ended September 30, 2016 was $41.7 million, or $0.39 per share, compared to a net loss of $30.7 million, or $0.29 per share, for the comparable quarter in 2015.
Conference Call Information
Keryx will host an investor conference call today, Wednesday, November 9, 2016, at 8:00 a.m. ET to discuss financial results for the third quarter of 2016. In order to participate in the conference call, please call 1-(888) 396-2320 (U.S.), 1-(774) 264-7560 (outside the U.S.), call-in ID: 118829223. The call will also be webcast with slides, which will be accessible through the Investors section of the company’s website at www.keryx.com. The audio replay will be available at http://www.keryx.com for a period of 15 days after the call.
Page 2 of 7

About Non-Dialysis Dependent Chronic Kidney Disease and Iron Deficiency Anemia
It is estimated that approximately one in 10 U.S. adults are affected by CKD, which has no cure. Treatment today consists of measures to help control signs and symptoms, reduce the impact of many complications to make a person more comfortable and slow disease progression.
Iron deficiency anemia is one of the most common complications of chronic kidney disease. IDA develops early in the course of CKD and worsens with disease progression, is extremely prevalent in the non-dialysis dependent CKD population and is associated with fatigue, lethargy, decreased quality of life and is also believed to be associated with cardiovascular complications, hospitalizations, and increased mortality. There are five stages of CKD; in stages 1 and 2 people are typically under the care of a primary care physician and have a mild loss of kidney function. Typically, as people progress to stage 3 hemoglobin levels begin to fall, the patient experiences moderate to severe loss of kidney function and is generally referred to a nephrologist. Stage 4 is characterized as advanced disease with multiple complications. Stage 5 is considered kidney failure and the stage in which a patient would often initiate dialysis. It is estimated that approximately 1.6 million adults with stage 3-5 CKD in the U.S. alone are also afflicted with iron deficiency anemia. Currently available oral iron supplements are associated with limited efficacy and dose-limiting tolerability issues. No oral iron medicines are currently FDA approved to treat iron deficiency anemia in non-dialysis dependent CKD patients, and the NDD-CKD patient population remains underserved.
About Auryxia®
Auryxia® (ferric citrate) was approved by the U.S. Food and Drug Administration on September 5, 2014 and is indicated in the U.S. for the control of serum phosphorus levels in patients with CKD on dialysis. The U.S. approval of Auryxia was based on data from the company’s Phase 3 registration program. In the Phase 3 clinical trials, Auryxia effectively reduced serum phosphorus levels to within the KDOQI guidelines range of 3.5 to 5.5 mg/dL.
Auryxia binds with dietary phosphate in the GI tract and precipitates as ferric phosphate. The unbound portion of Auryxia has been shown to increase serum iron parameters including ferritin and transferrin saturation (TSAT). Iron absorption from Auryxia may lead to excessive elevations in iron stores. Accordingly, physicians should assess and monitor iron parameters before starting and while on Auryxia, and may need to decrease or discontinue IV iron for these patients. The most common adverse events for Auryxia treated patients were gastrointestinal-related, including diarrhea, nausea, vomiting and constipation. For more information about Auryxia and the US full prescribing information, visit www.Auryxia.com.
IMPORTANT U.S. SAFETY INFORMATION FOR AURYXIA® (ferric citrate)
Contraindication: Patients with iron overload syndrome, e.g. hemochromatosis, should not take Auryxia® (ferric citrate).
Iron Overload: Iron absorption from Auryxia may lead to increased iron in storage sites. Iron parameters should be monitored prior to and while on Auryxia. Patients receiving IV iron may require a reduction in dose or discontinuation of IV iron therapy.
Accidental Overdose of Iron: Accidental overdose of iron containing products is a leading cause of fatal poisoning in children under 6 years of age. Keep Auryxia away from children as it contains iron. Call a poison control center or your physician in case of an accidental overdose in a child.
Patients with Gastrointestinal Bleeding or Inflammation: Safety has not been established for these patients.
Page 3 of 7

Adverse Events: The most common adverse events with Auryxia were diarrhea (21%), nausea (11%), constipation (8%), vomiting (7%) and cough (6%). Gastrointestinal adverse reactions were the most common reason for discontinuing Auryxia (14%). Auryxia contains iron and may cause dark stools, which is considered normal with oral medications containing iron.
Drug Interactions: Doxycycline should be taken at least 1 hour before Auryxia. Ciprofloxacin should be taken at least 2 hours before or after Auryxia.
For Full Prescribing Information for Auryxia, please visit http://auryxia.com/important-safety-information/
Page 4 of 7

Keryx Biopharmaceuticals, Inc.
Condensed Consolidated Statement of Operations
(In thousands, except share and per share amounts)
(unaudited)
| Three Months Ended September 30, |
Nine Months Ended September 30, |
|||||||||||||||
| 2016 | 2015 | 2016 | 2015 | |||||||||||||
| Revenues: |
||||||||||||||||
| Net U.S. Auryxia product sales |
$ | 5,050 | $ | 3,191 | $ | 18,945 | $ | 5,371 | ||||||||
| License revenue |
1,287 | 1,017 | 3,505 | 2,526 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total Revenues |
6,337 | 4,208 | 22,450 | 7,897 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Operating Expenses: |
||||||||||||||||
| Cost of goods sold |
18,196 | 3,065 | 24,365 | 3,445 | ||||||||||||
| License expenses |
772 | 611 | 2,103 | 1,516 | ||||||||||||
| Research and development |
8,674 | 11,150 | 23,320 | 28,704 | ||||||||||||
| Selling, general and administrative |
20,521 | 20,205 | 61,518 | 59,847 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total Operating Expenses |
48,163 | 35,031 | 111,306 | 93,512 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Operating Loss |
(41,826 | ) | (30,823 | ) | (88,856 | ) | (85,615 | ) | ||||||||
| Other Income (expense): |
||||||||||||||||
| Other income (expense), net |
150 | 100 | (38,395 | ) | 321 | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Loss Before Income Taxes |
(41,676 | ) | (30,723 | ) | (127,251 | ) | (85,294 | ) | ||||||||
| Income taxes |
20 | 22 | 60 | 67 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net Loss |
$ | (41,696 | ) | $ | (30,745 | ) | (127,311 | ) | (85,361 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net Loss Per Common Share Basic and diluted net loss per common share |
$ | (0.39 | ) | $ | (0.29 | ) | $ | (1.20 | ) | $ | (0.83 | ) | ||||
|
|
|
|
|
|
|
|
|
|||||||||
| Shares Used in Computing Net Loss Per Common Share |
||||||||||||||||
| Basic and diluted |
105,924,106 | 105,205,170 | 105,805,669 | 103,458,248 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
Page 5 of 7

Selected Consolidated Balance Sheet Data
(In thousands)
(unaudited)
| September 30, 2016 | December 31, 2015 | |||||||
| Assets |
||||||||
| Cash and cash equivalents |
$ | 132,172 | $ | 200,290 | ||||
| Inventory |
$ | 24,313 | $ | 41,881 | ||||
| Total assets |
$ | 168,625 | $ | 258,685 | ||||
| Liabilities and Stockholders’ Equity |
||||||||
| Accounts payable and accrued expenses |
$ | 18,038 | $ | 26,795 | ||||
| Deferred revenue |
$ | — | $ | 3,526 | ||||
| Derivative liability |
$ | — | $ | 46,686 | ||||
| Convertible senior notes, net of discount |
$ | 125,000 | $ | 90,773 | ||||
| Total liabilities |
$ | 146,636 | $ | 171,751 | ||||
| Stockholders’ equity |
$ | 21,989 | $ | 86,934 | ||||
Forward Looking Statements
Some of the statements included in this press release, particularly those regarding future revenues and expenses and the commercialization and ongoing clinical development of Auryxia, including those statements related to the interruption in the supply of Auryxia and when Auryxia may be available to patients again as well as the submission of an sNDA to the FDA to expend the label of ferric citrate to include the treatment of IDA in adults with stage 3-5 NDD-CKD, may be forward-looking statements that involve a number of risks and uncertainties. For those statements, we claim the protection of the safe harbor for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995. Among the factors that could cause our actual results to differ materially are the following: our ability to quickly and successfully identify and resolve the production-related issue; our ability to quickly and successfully identify and engage secondary suppliers of finished drug product; our ability to receive FDA approval of any secondary suppliers of finished drug product including approval of our second manufacturer by the assigned PDUFA action date; whether we can increase adoption of Auryxia in patients with CKD on dialysis; whether we can maintain our operating expenses to projected levels while continuing our current clinical, regulatory and commercial activities; whether we will able to identify and negotiate acceptable terms with a commercialization partner in the E.U.; whether we or a partner can successfully launch Fexeric® in the E.U.; whether Riona will be successfully marketed in Japan by our Japanese partner, Japan Tobacco, Inc. and its subsidiary Torii Pharmaceutical Co., Ltd; the risk that the FDA may not concur with our interpretation of our Phase 3 study results in NDD- CKD, supportive data, conduct of the studies, or any other part of our regulatory submission and could ultimately deny approval of ferric citrate for the treatment of IDA in adults with stage 3-5 NDD-CKD; the risk that if approved for use in NDD-CKD that we may not be able to successfully market Auryxia for use in this indication; and other risk factors identified from time to time in our reports filed with the Securities and Exchange Commission. Any forward looking statements set forth in this press release speak only as of the date of this press release. We do not undertake to update any of these forward-looking statements to reflect events or circumstances that occur after the date hereof. This press release and prior releases are available at http://www.keryx.com. The information found on our website is not incorporated by reference into this press release and is included for reference purposes only.
Page 6 of 7

About Keryx Biopharmaceuticals, Inc.
Keryx Biopharmaceuticals, with headquarters in Boston, is focused on bringing innovative medicines to market for people with renal disease. In December 2014, the company launched its first FDA-approved medicine, Auryxia® (ferric citrate) in the United States. In January 2014, ferric citrate was approved for use in Japan, where it is being marketed as Riona® by Keryx’s Japanese partner, Japan Tobacco Inc. and Torii Pharmaceutical Co. Ltd. In September 2015, the European Commission granted European market authorization for Fexeric® (ferric citrate coordination complex). For more information about Keryx, please visit www.keryx.com.
KERYX BIOPHARMACEUTICALS CONTACTS:
Amy Sullivan
Vice President, Strategic Operations and Corporate Affairs
T: 617.466.3519
Lora Pike
Senior Director, Investor Relations
T: 617.466.3511
Page 7 of 7

Third Quarter 2016 Financial Results Conference Call November 9, 2016 Keryx Biopharmaceuticals, Inc. Exhibit 99.2

Safe Harbor Statement Some of the statements included in this presentation, particularly those regarding future revenues and expenses and the commercialization and ongoing clinical development of Auryxia, including those statements related to the interruption in the supply of Auryxia and when Auryxia may be available to patients again as well as the submission of an sNDA to the FDA to expend the label of ferric citrate to include the treatment of IDA in adults with stage 3-5 NDD-CKD, may be forward-looking statements that involve a number of risks and uncertainties. For those statements, we claim the protection of the safe harbor for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995. Among the factors that could cause our actual results to differ materially are the following: our ability to quickly and successfully identify and resolve the production-related issue; our ability to quickly and successfully identify and engage secondary suppliers of finished drug product; our ability to receive FDA approval of any secondary suppliers of finished drug product including approval of our second manufacturer by the assigned PDUFA action date; whether we can increase adoption of Auryxia in patients with CKD on dialysis; whether we can maintain our operating expenses to projected levels while continuing our current clinical, regulatory and commercial activities; whether we will able to identify and negotiate acceptable terms with a commercialization partner in the E.U.; whether we or a partner can successfully launch Fexeric® in the E.U.; whether Riona will be successfully marketed in Japan by our Japanese partner, Japan Tobacco, Inc. and its subsidiary Torii Pharmaceutical Co., Ltd; the risk that the FDA may not concur with our interpretation of our Phase 3 study results in NDD-CKD, supportive data, conduct of the studies, or any other part of our regulatory submission and could ultimately deny approval of ferric citrate for the treatment of IDA in adults with stage 3-5 NDD-CKD; the risk that if approved for use in NDD-CKD that we may not be able to successfully market Auryxia for use in this indication; and other risk factors identified from time to time in our reports filed with the Securities and Exchange Commission. Any forward looking statements set forth in this press release speak only as of the date of this press release. We do not undertake to update any of these forward-looking statements to reflect events or circumstances that occur after the date hereof. This press release and prior releases are available at http://www.keryx.com. The information found on our website is not incorporated by reference into this press release and is included for reference purposes only.

Third Quarter 2016 Financial Results - Agenda Topic Speaker Introduction Amy Sullivan, VP Strategic Operations and Corporate Affairs Opening Remarks Greg Madison, President and CEO Third Quarter Financial Results Scott Holmes, Chief Financial Officer sNDA submission and ASN 2016 John Neylan, M.D., Chief Medical Officer Plans to make Auryxia available to patients Doug Jermasek, VP, Marketing & Strategy Question & Answer All

Business Focus Past Three Months Managed through temporary supply interruption Ready to make Auryxia available pending FDA approval of second manufacturer Completed sNDA to expand Auryxia’s label for potential treatment of iron deficiency anemia in adults with stage 3-5 NDD-CKD Ready to submit sNDA pending FDA agreement on pediatric plan Created plan to bring Auryxia back to dialysis patients Continued to develop launch plan for potential treatment of IDA in adults with stage 3-5 NDD-CKD Plans Auryxia sNDA
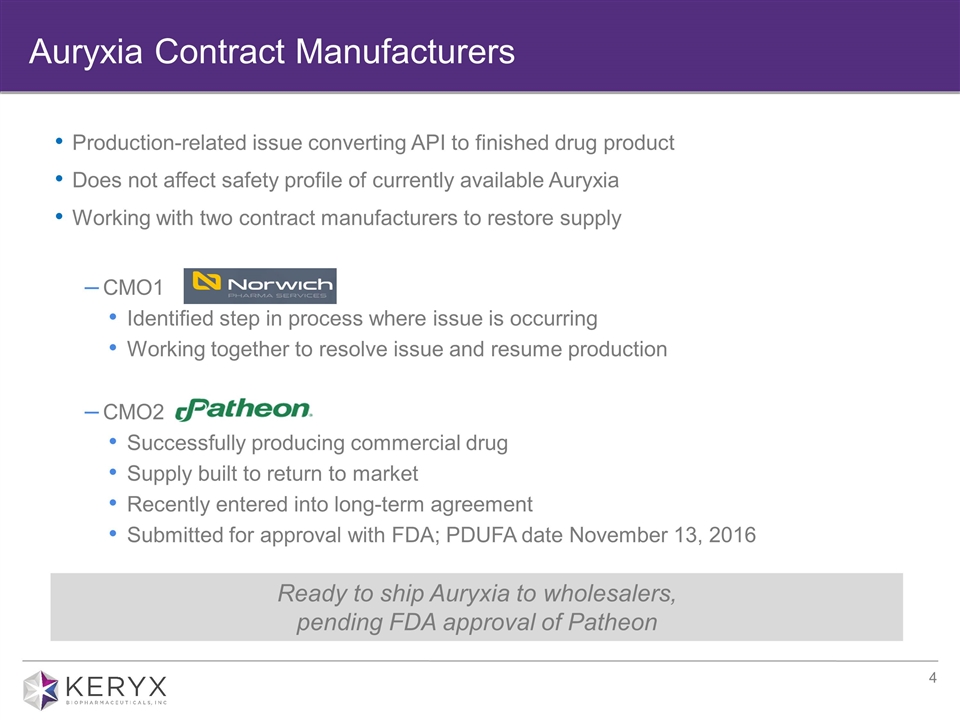
Production-related issue converting API to finished drug product Does not affect safety profile of currently available Auryxia Working with two contract manufacturers to restore supply CMO1 Identified step in process where issue is occurring Working together to resolve issue and resume production CMO2 Successfully producing commercial drug Supply built to return to market Recently entered into long-term agreement Submitted for approval with FDA; PDUFA date November 13, 2016 Auryxia Contract Manufacturers Ready to ship Auryxia to wholesalers, pending FDA approval of Patheon

Financial Highlights Scott Holmes Chief Financial Officer
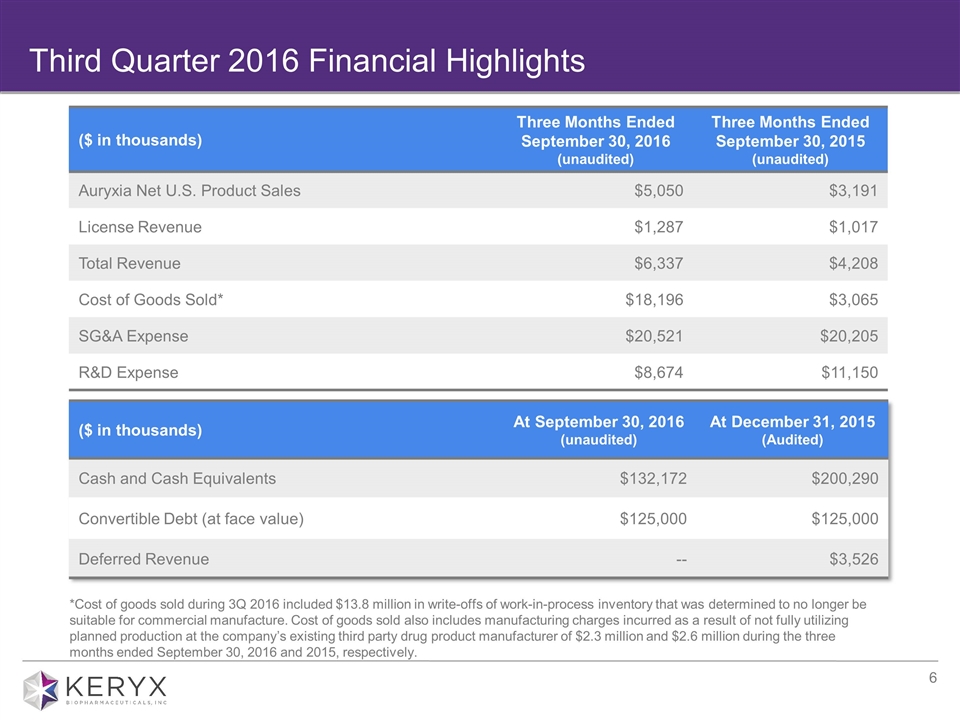
Third Quarter 2016 Financial Highlights ($ in thousands) Three Months Ended September 30, 2016 (unaudited) Three Months Ended September 30, 2015 (unaudited) Auryxia Net U.S. Product Sales $5,050 $3,191 License Revenue $1,287 $1,017 Total Revenue $6,337 $4,208 Cost of Goods Sold* $18,196 $3,065 SG&A Expense $20,521 $20,205 R&D Expense $8,674 $11,150 ($ in thousands) At September 30, 2016 (unaudited) At December 31, 2015 (Audited) Cash and Cash Equivalents $132,172 $200,290 Convertible Debt (at face value) $125,000 $125,000 Deferred Revenue -- $3,526 *Cost of goods sold during 3Q 2016 included $13.8 million in write-offs of work-in-process inventory that was determined to no longer be suitable for commercial manufacture. Cost of goods sold also includes manufacturing charges incurred as a result of not fully utilizing planned production at the company’s existing third party drug product manufacturer of $2.3 million and $2.6 million during the three months ended September 30, 2016 and 2015, respectively.

sNDA for Potential Label Expansion & ASN 2016 John Neylan, M.D. Chief Medical Officer
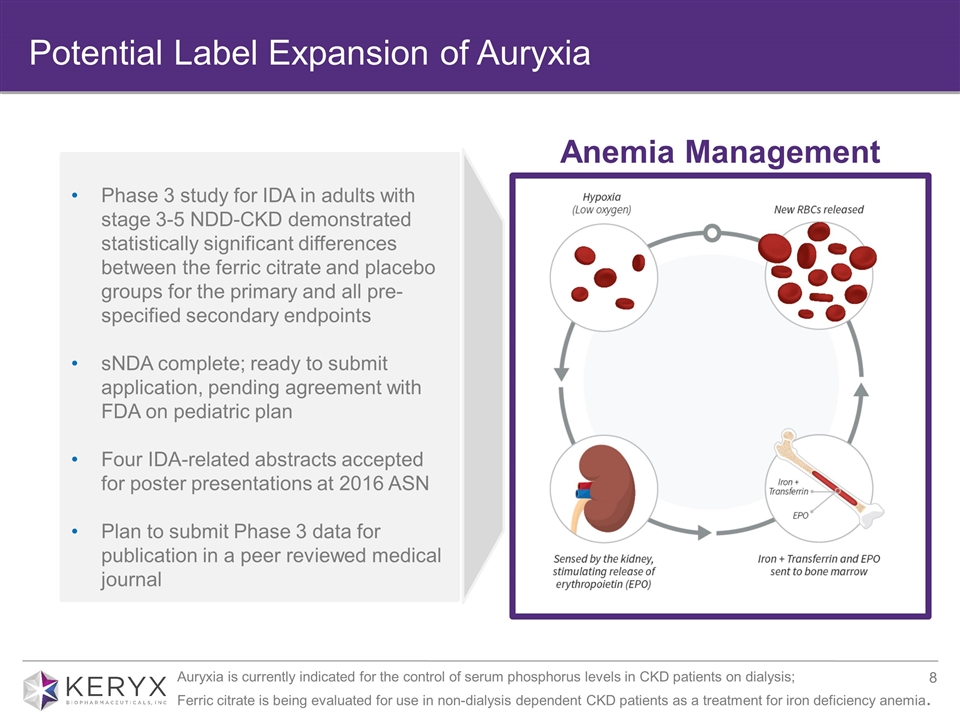
Potential Label Expansion of Auryxia Anemia Management Phase 3 study for IDA in adults with stage 3-5 NDD-CKD demonstrated statistically significant differences between the ferric citrate and placebo groups for the primary and all pre-specified secondary endpoints sNDA complete; ready to submit application, pending agreement with FDA on pediatric plan Four IDA-related abstracts accepted for poster presentations at 2016 ASN Plan to submit Phase 3 data for publication in a peer reviewed medical journal Auryxia is currently indicated for the control of serum phosphorus levels in CKD patients on dialysis; Ferric citrate is being evaluated for use in non-dialysis dependent CKD patients as a treatment for iron deficiency anemia.
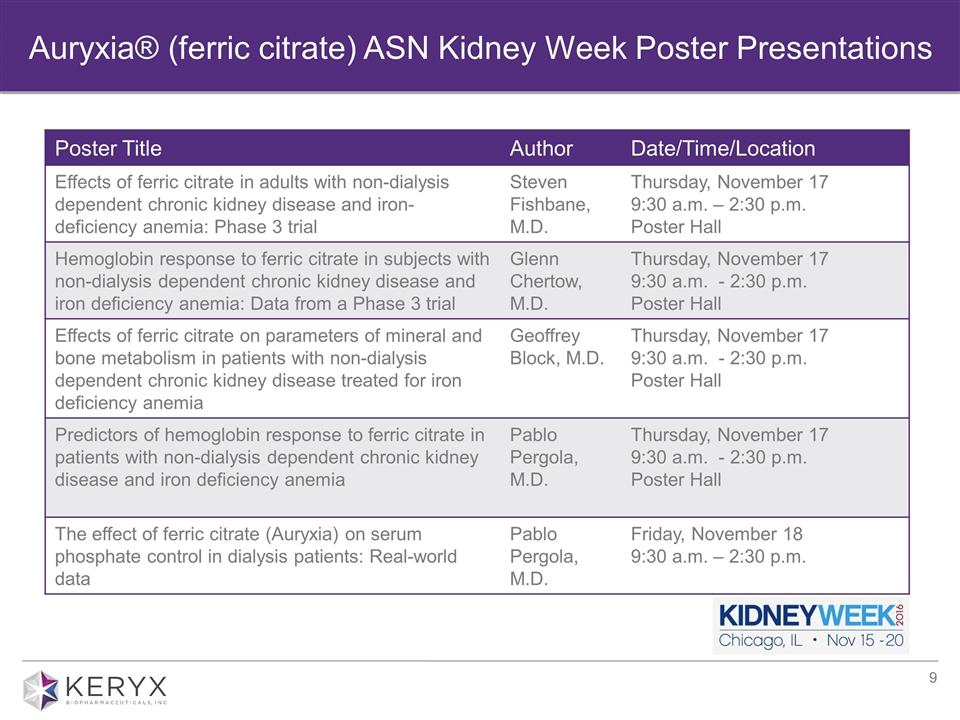
Auryxia® (ferric citrate) ASN Kidney Week Poster Presentations Poster Title Author Date/Time/Location Effects of ferric citrate in adults with non-dialysis dependent chronic kidney disease and iron-deficiency anemia: Phase 3 trial Steven Fishbane, M.D. Thursday, November 17 9:30 a.m. – 2:30 p.m. Poster Hall Hemoglobin response to ferric citrate in subjects with non-dialysis dependent chronic kidney disease and iron deficiency anemia: Data from a Phase 3 trial Glenn Chertow, M.D. Thursday, November 17 9:30 a.m. - 2:30 p.m. Poster Hall Effects of ferric citrate on parameters of mineral and bone metabolism in patients with non-dialysis dependent chronic kidney disease treated for iron deficiency anemia Geoffrey Block, M.D. Thursday, November 17 9:30 a.m. - 2:30 p.m. Poster Hall Predictors of hemoglobin response to ferric citrate in patients with non-dialysis dependent chronic kidney disease and iron deficiency anemia Pablo Pergola, M.D. Thursday, November 17 9:30 a.m. - 2:30 p.m. Poster Hall The effect of ferric citrate (Auryxia) on serum phosphate control in dialysis patients: Real-world data Pablo Pergola, M.D. Friday, November 18 9:30 a.m. – 2:30 p.m.

Auryxia Return to Patients Doug Jermasek V.P. Marketing and Strategy

Renal Community Support

Three types of patients upon resupply: Patients who have been switched to another binder Patients who have been maintained on Auryxia New patients who could benefit from Auryxia treatment Market research suggests: Physicians and dietitians who have had patients on Auryxia have a willingness to return patients to Auryxia Variability around the timing of Auryxia use Sales force has remained engaged with the renal care teams during the supply interruption and is ready for resupply Commercial Readiness for Auryxia Resupply Goal: Recapture sales momentum as soon as possible and ensure that patients once again have access to the potential benefits of Auryxia
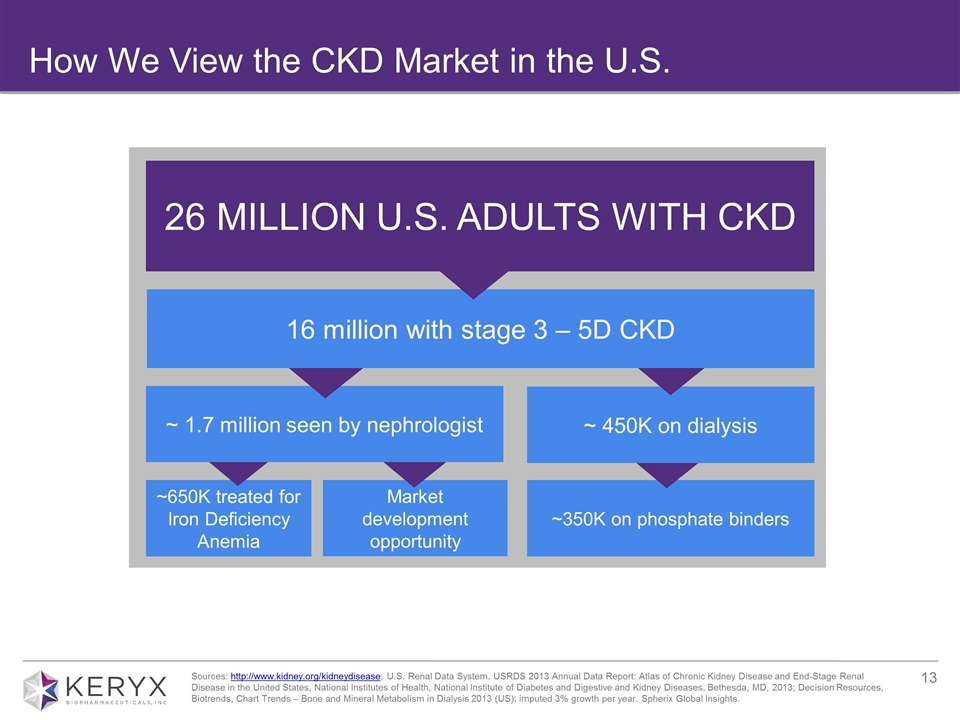
How We View the CKD Market in the U.S. Sources: http://www.kidney.org/kidneydisease; U.S. Renal Data System, USRDS 2013 Annual Data Report: Atlas of Chronic Kidney Disease and End-Stage Renal Disease in the United States, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD, 2013; Decision Resources, Biotrends, Chart Trends – Bone and Mineral Metabolism in Dialysis 2013 (US); imputed 3% growth per year. Spherix Global Insights. 26 MILLION U.S. ADULTS WITH CKD ~650K treated for Iron Deficiency Anemia ~350K on phosphate binders ~ 450K on dialysis Market development opportunity ~ 1.7 million seen by nephrologist 16 million with stage 3 – 5D CKD

Closing Remarks and Q&A

Key Takeaways Supply of Auryxia rebuilt; prepared to make Auryxia available to people on dialysis, pending FDA approval of Patheon Completed sNDA for potential label expansion of Auryxia; ready to submit application pending FDA agreement on pediatric plan Presenting more detailed IDA/CKD Phase 3 data at ASN Kidney Week Developing go-to-market strategy for IDA/NDD-CKD opportunity Auryxia is currently indicated for the control of serum phosphorus levels in CKD patients on dialysis; Ferric citrate is being evaluated for use in non-dialysis dependent CKD patients as a treatment for iron deficiency anemia.

Third Quarter 2016 Financial Results Conference Call November 9, 2016 Keryx Biopharmaceuticals, Inc.
Serious News for Serious Traders! Try StreetInsider.com Premium Free!
You May Also Be Interested In
- Form 8.3 - UK Commercial Property REIT
- Report from Concentric AB's annual general meeting on April 18, 2024
- BeWhere Holdings Inc Reports 2023 Fiscal Year-end Results
Create E-mail Alert Related Categories
SEC FilingsSign up for StreetInsider Free!
Receive full access to all new and archived articles, unlimited portfolio tracking, e-mail alerts, custom newswires and RSS feeds - and more!



 Tweet
Tweet Share
Share